ENERGY OF REACTIONS Entropy Enthalpy and Gibbs Free

ENERGY OF REACTIONS Entropy, Enthalpy, and Gibb’s Free Energy

ENERGY SO FAR Review: Energy is the ability to do work or produce heat There is potential and kinetic energy Energy can neither be created or destroyed Every compound needs energy to increase temperature or to change from one state of matter to another

ENERGY SO FAR Units of energy: Joule (J) Calorie (cal) 1 cal = 4. 184 J 1 k. J = 1000 J 1 kcal = 1000 cal

REVIEW Convert 255 the following: cal = _______ J 4200 J = _______cal 55 kcal = _______ J 6325 J = _______ k. J 3. 85 k. J = ________kcal

OTHER ASPECTS OF ENERGY Energy is also an important component of chemical reactions Example: 4 Fe(s) In + 3 O 2(g) 2 Fe 2 O 3(s) + 1625 k. J this example, you combine iron and oxygen to produce iron (III) oxide. This reaction also produces 1625 k. J of energy/heat.

ENERGY AND REACTIONS To better explain the energy changes in reactions, chemists have come up with: Enthalpy (H): the heat content of substances under constant pressure For a chemical reaction, we describe the change in enthalpy. This is called: Enthalpy (or heat) of reaction (δHrxn): the change in heat or energy in a chemical reaction

HOW DO WE MEASURE δHrxn To measure the heat produced or used by a reaction, scientists again use calorimeters. (REVIEW: What is a calorimeter? ) To calculate, we have the following formula: δHrxn = Hproducts - Hreactants

PREVIOUS EXAMPLE Previously, we looked at the following: 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) + 1625 k. J According to this equation, we produced (or lost) 1625 k. J of energy to the environment (you feel this as hot) EXOTHERMIC Therefore Hreactants > Hproducts

WHAT THIS MEANS For the reaction: Whenever Hreactants > Hproducts, the convention is to give a negative sign to the δHrxn Therefore, exothermic reactions are always a negative “-” δHrxn On the right side of the arrow

ENDOTHERMIC Since we know that exothermic reactions have a negative heat of reaction: WHAT IS THE SIGN FOR AN ENDOTHERMIC REACTION?

WHAT THIS MEANS Endothermic “+” δHrxn reactions are always a positive On the left side of the arrow You have to add energy to the reactant side to equal the products Therefore we always have to add energy to an endothermic reaction: Example: 27 k. J + NH 4 NO 3 (s) NH 4+(aq) + NO 3 -(aq)

CALCULATING HEATS OF REACTION Chemists have measured different heats of reaction for combustion reactions. In each case, they do this under a condition of standard pressure and temperature. They call these heats of combustion: δHcomb

REVIEW What is the general format for a combustion reaction?

COMBUSTION REACTION + O 2 CO 2 + H 2 O Therefore, these are values for the combustion of 1 MOLE of the molecule Example: Molecule We will look at the combustion of 1 mole of glucose (sugar) C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + 2808 k. J δHcomb = -2808 k. J/mole
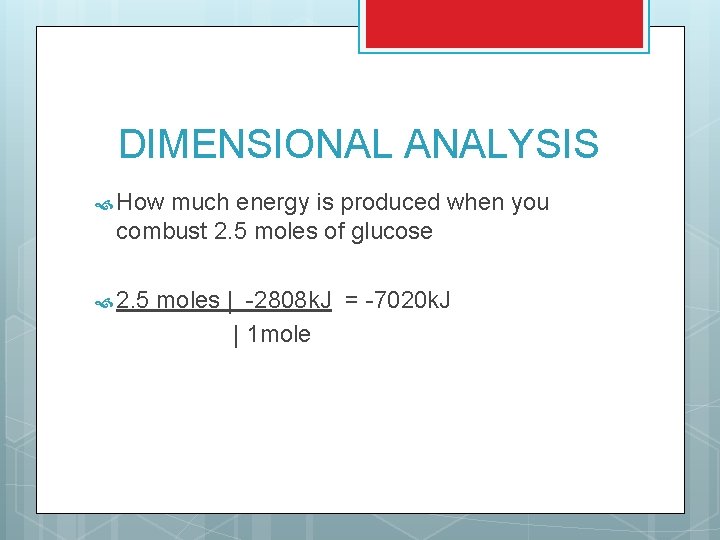
DIMENSIONAL ANALYSIS How much energy is produced when you combust 2. 5 moles of glucose 2. 5 moles | -2808 k. J = -7020 k. J | 1 mole

CALCULATING HEAT OF COMBUSTION Example: You start with 3. 55 x 103 g of glucose (C 6 H 12 O 6). How much energy is released when glucose goes through a complete combustion reaction? STEPS: Convert to moles Use the δHcomb for glucose to calculate energy δHcomb = -2808 k. J/mole
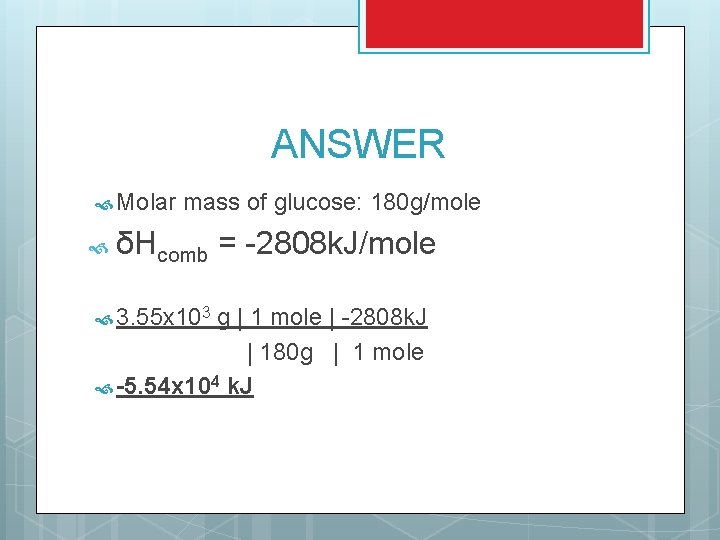
ANSWER Molar mass of glucose: 180 g/mole δHcomb = -2808 k. J/mole 3. 55 x 103 g | 1 mole | -2808 k. J | 180 g | 1 mole -5. 54 x 104 k. J

TRY THESE 1. 2. The heat of combustion for octane (C 8 H 18) is -5471 k. J/mole. If you start with 1550 g of octane, how much energy is released? Sucrose (C 12 H 22 O 11), or table sugar, has a heat of combustion of -5644 k. J/mole. If you add 2. 5 g of sucrose to your cereal, how much energy will be added to your cereal?
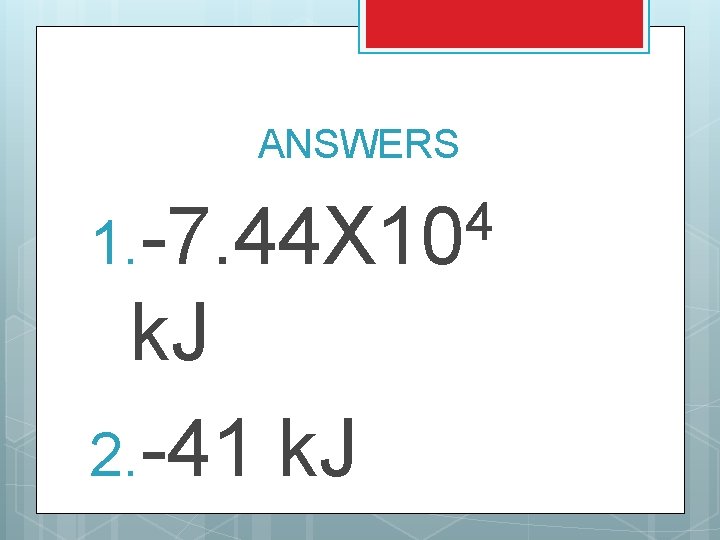
ANSWERS 4 1. -7. 44 X 10 k. J 2. -41 k. J

TRY THIS You have a cup filled with 125 m. L of glucose (C 6 H 12 O 6). If the density of glucose is 1. 54 g/m. L, how many moles of glucose do you have? If the heat of combustion for glucose is 2805 k. J/mole, what is the heat produced from the cup of glucose?
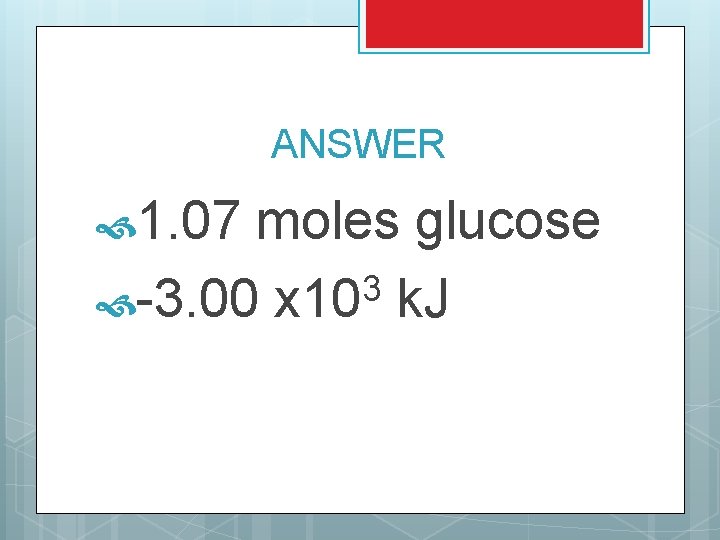
ANSWER 1. 07 moles glucose 3 -3. 00 x 10 k. J

TRY THIS A combustion reaction with octane (C 8 H 18) releases a total of -5. 55 x 104 k. J of energy. If the heat of combustion for octane is -250 k. J/mole, how many grams of octane did you start with?
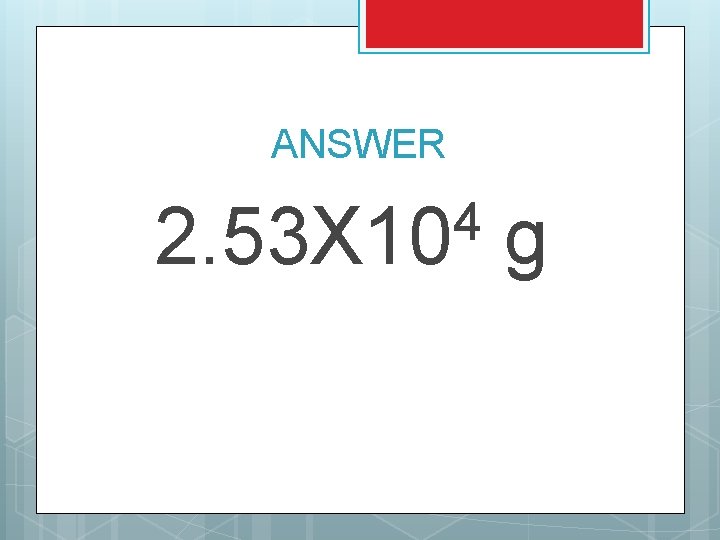
ANSWER 4 2. 53 X 10 g

HEATS OF REACTION As we said earlier, most heats of reaction or heats of combustion are measured using a calorimeter. Sometimes (because some reactions are toxic or unstable), we cannot use a calorimeter and must find an alternative way to measure heats of reaction.

HEATS OF REACTION Hess’s law: If you add two or more thermochemical equations to produce a final equation for a reaction, then the sum of the enthalpy changes for the individual reactions is the enthalpy change for the final reaction

EXAMPLE: FIND THE HEAT OF REACTION 2 S + 3 O 2 2 SO 3 This reaction can occurs in 2 steps: S + O 2 SO 2 ; 2 SO 3 2 SO 2 + O 2 ; How δH = -297 k. J δH = 198 k. J do these reactions go together to form the final reaction?
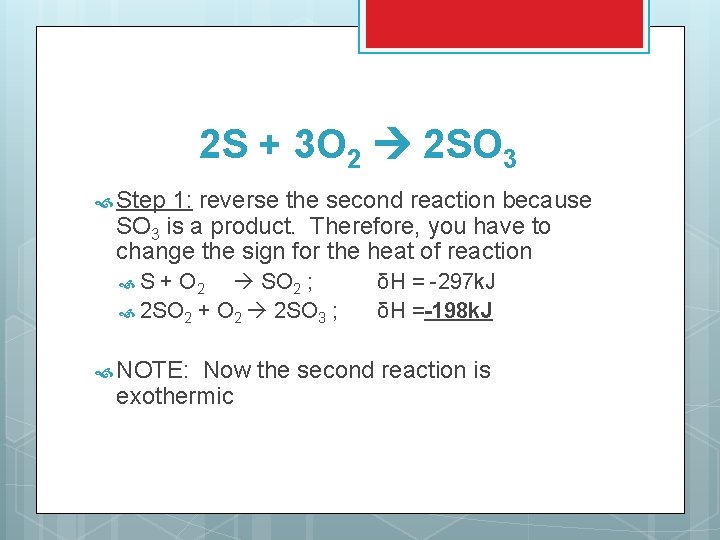
2 S + 3 O 2 2 SO 3 Step 1: reverse the second reaction because SO 3 is a product. Therefore, you have to change the sign for the heat of reaction S + O 2 SO 2 ; 2 SO 2 + O 2 2 SO 3 ; NOTE: δH = -297 k. J δH =-198 k. J Now the second reaction is exothermic

2 S + 3 O 2 2 SO 3 Step 2: there are 2 moles of S in the first reaction. This means we have to double everything in the first reaction (including the heat of reaction) 2(S 2 S + O 2 SO 2 ; + 2 O 2 2 SO 2; 2 SO 2 + O 2 2 SO 3 ; δH = -297 k. J) δH = -594 k. J δH =-198 k. J

2 S + 3 O 2 2 SO 3 Step 3: you add the two reactions together to get the final product 2 S + 2 O 2 2 SO 2; 2 SO 2 + O 2 2 SO 3 ; 2 S + 3 O 2 2 SO 3; NOTE: δH = -594 k. J δH = -198 k. J__ δH =-792 k. J__ If a compound is on opposite sides of a reaction, they cancel out

SUMMARY 1. 2. 3. 4. 5. You need to have known chemical reactions that you can combine to form the final chemical reaction You need the δH for each reaction If you need to reverse a reaction, you must also change the sign of δH If you need to multiply a reaction by a number, you must also multiply the δH When all reactions are completed, you then add them together to get the final δH

TRY THE FOLLOWING Combine the two reactions to find the heat of reaction when hydrogen peroxide (H 2 O 2) breaks apart to form water and oxygen) 2 H 2 O 2 2 H 2 O + O 2; + O 2 2 H 2 O; H 2 + O 2 H 2 O 2; ΔH = ? δH = -572 k. J δH = -188 k. J

2 H 2 O 2 2 H 2 O + O 2 Step 1: We need to reverse the second reaction 2 H 2 + O 2 2 H 2 O; H 2 O 2 H 2 + O 2; Step δH = -572 k. J δH = 188 k. J 2: You need 2 moles of H 2 O 2 2 H 2 + O 2 2 H 2 O; 2 H 2 O 2 2 H 2 + 2 O 2; δH = -572 k. J δH = 376 k. J

2 H 2 O 2 2 H 2 O + O 2 Step 3: Combine the reactions 2 H 2 + O 2 2 H 2 O; δH = -572 k. J 2 H 2 O 2 2 H 2 + 2 O 2; δH = 376 k. J 2 H 2 O 2 2 H 2 O + O 2 NOTE: δH = -196 k. J When you cancel out reactants and products, 2 H 2 cancels out 2 H 2, but only 1 O 2 is cancelled out in the products

TRY THIS (EXTRA CREDIT) 2 Ca + 2 C + 3 O 2 2 Ca. CO 3 Ca + 2 C Ca. C 2; d. Hrxn = -62. 8 k. J CO 2 C + O 2; 2 Ca. CO 3 + 2 CO 2 2 Ca. C 2 + 5 O 2; d. Hrxn = +1538 k. J d. Hrxn = -393 k. J

OTHER PRACTICE 4 NH 3 + 5 O 2 4 NO + 6 H 2 O N 2 + O 2 2 NO ∆H = -180. 5 k. J N 2 + 3 H 2 2 NH 3 ∆H = -91. 8 k. J 2 H 2 + O 2 2 H 2 O ∆H = -483. 6 k. J
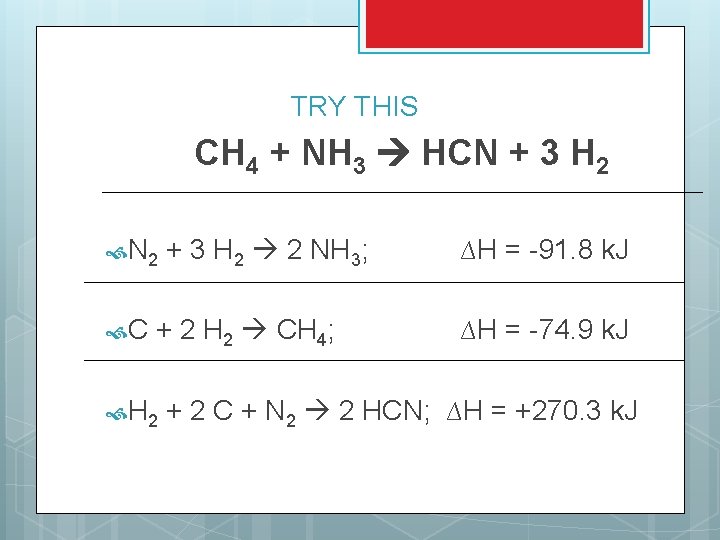
TRY THIS CH 4 + NH 3 HCN + 3 H 2 N 2 C + 3 H 2 2 NH 3; + 2 H 2 CH 4; H 2 ∆H = -91. 8 k. J ∆H = -74. 9 k. J + 2 C + N 2 2 HCN; ∆H = +270. 3 k. J
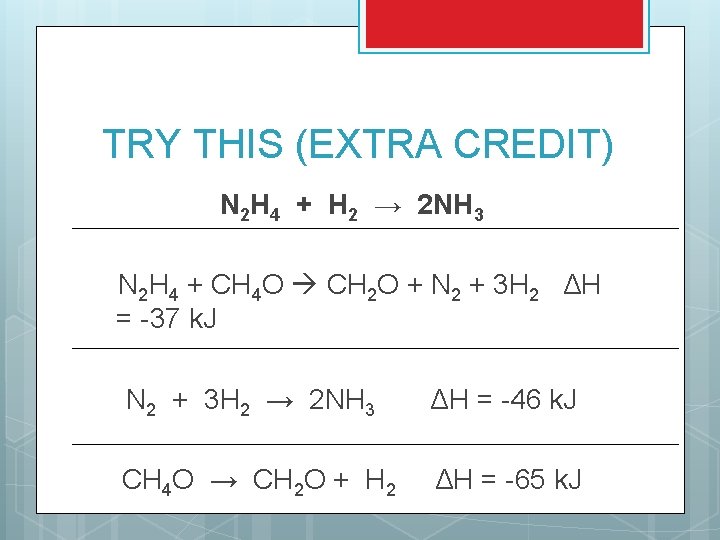
TRY THIS (EXTRA CREDIT) N 2 H 4 + H 2 → 2 NH 3 N 2 H 4 + CH 4 O CH 2 O + N 2 + 3 H 2 ΔH = -37 k. J N 2 + 3 H 2 → 2 NH 3 ΔH = -46 k. J CH 4 O → CH 2 O + H 2 ΔH = -65 k. J
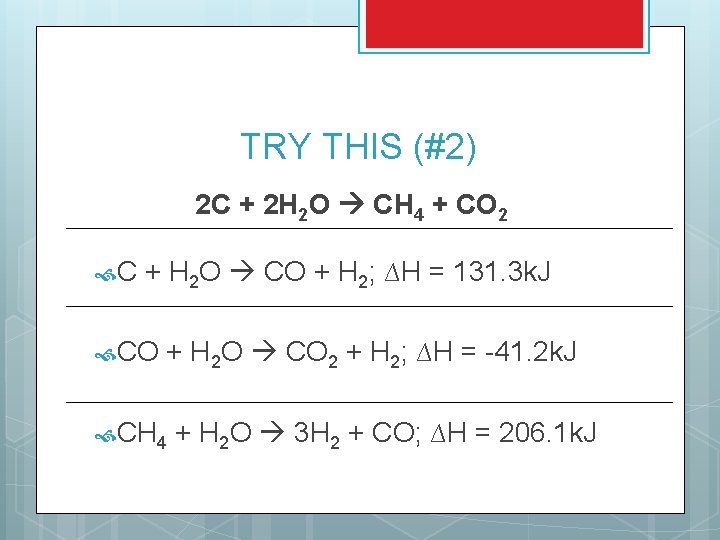
TRY THIS (#2) 2 C + 2 H 2 O CH 4 + CO 2 C + H 2 O CO + H 2; ∆H = 131. 3 k. J CO + H 2 O CO 2 + H 2; ∆H = -41. 2 k. J CH 4 + H 2 O 3 H 2 + CO; ∆H = 206. 1 k. J

TRY THIS (#1) 5 C + 6 H 2 C 5 H 12 C + O 2 CO 2 ΔH = -393. 5 k. J 2 H 2 + O 2 2 H 2 O ΔH = -571. 8 k. J C 5 H 12 +8 O 2 5 CO 2 + 6 H 2 O ΔH = -3536 k. J

PREDICTING SPONTANEOUS CHEMICAL REACTIONS Energy is an essential ingredient for a chemical reaction Not all chemical reactions happen spontaneously To determine if a reaction occurs spontaneously, we need to look at another concept: ENTROPY

ENTROPY Entropy (S): a measure of the number of possible ways that the energy of a system can be distributed In other words, entropy is the tendency for molecules to spread out as far as possible from each other Since molecules spreading out is dependent on temperature, the unit for entropy is (J/K) – Joules/Kelvin

KELVIN TEMPERATURE Before we go any further, we need to review Kelvins: Kelvin (K) A temperature scale based on absolute 0 (the coldest possible temperature) The Celsius temperature converts to Kelvin: K = °C + 273

THERMODYNAMICS Thermodynamics: The laws of thermodynamics, in principle, describe the specifics for the transport of heat and work. The only law we are interested in is the second law of thermodynamics

SECOND LAW OF THERMODYNAMICS Second Law of Thermodynamics: spontaneous processes always proceed in such a way that the entropy of the universe increases.

PREDICTING CHANGES IN ENTROPY Reminder: δHrxn We = Hreactants – Hproducts have a similar equation for entropy δSrxn = Sproducts - Sreactants If the entropy increases during a reaction, then Sproducts > Sreactants and δSrxn is positive If the entropy decreases during a reaction, then Sproducts < Sreactants and δSrxn is negative

PREDICTING CHANGES IN ENTROPY Changing states of matter: entropy changes when you go between solid, liquid or gas H 2 O(l) H 2 O (g); δSrxn > 0 Dissolving a gas in a solid or liquid always decreases entropy When you increase the number of gas particles in a reaction, entropy tends to increase Zn(s) + HCl(aq) Zn. Cl 2(aq) + H 2(g); δSrxn > 0

PREDICTING CHANGES IN ENTROPY With some exceptions, entropy increases when a solid dissolves in a liquid The solid tends to break apart Random motion of particles increases as the temperature increases As you increase temperature, the entropy increases

PREDICT THE FOLLOWING Try to determine whether entropy increases or decreases for the following reactions: 1. 2. 3. 4. CF(g) + F 2(g) CF 3 (g) NH 3 (g) NH 3 (aq) C 10 H 8 (s) C 10 H 8 (l) H 2 O (l) H 2 (g) + O 2 (g)
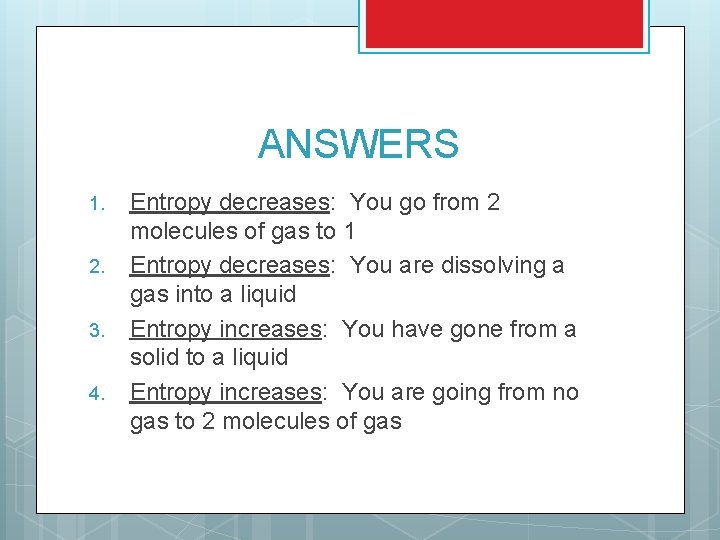
ANSWERS 1. 2. 3. 4. Entropy decreases: You go from 2 molecules of gas to 1 Entropy decreases: You are dissolving a gas into a liquid Entropy increases: You have gone from a solid to a liquid Entropy increases: You are going from no gas to 2 molecules of gas

PUTTING IT ALL TOGETHER Generally, exothermic reactions are spontaneous (you do not need to add energy to make them work) Generally, spontaneous reactions will increase in entropy Yet, there is a way to know for sure if a reaction will occur spontaneously or not

GIBB’S FREE ENERGY William Gibbs (an American) combined enthalpy and entropy to determine if a reaction was spontaneous He called his equation Gibbs Free Energy equation Free energy: the energy that is available to do work (δG) The energy that holds a molecule together is called BOND ENERGY Reactions are either making or breaking these bonds by releasing or adding energy

SPONTANEOUS? Like entropy or enthalpy, Gibb’s free energy can be positive or negative If positive (+): if the Gibb’s free energy is positive, the reaction is not spontaneous (you would need to add energy to make the reaction work) - ENDERGONIC If negative (-): if the Gibb’s free energy is negative, the reaction is spontaneous EXERGONIC
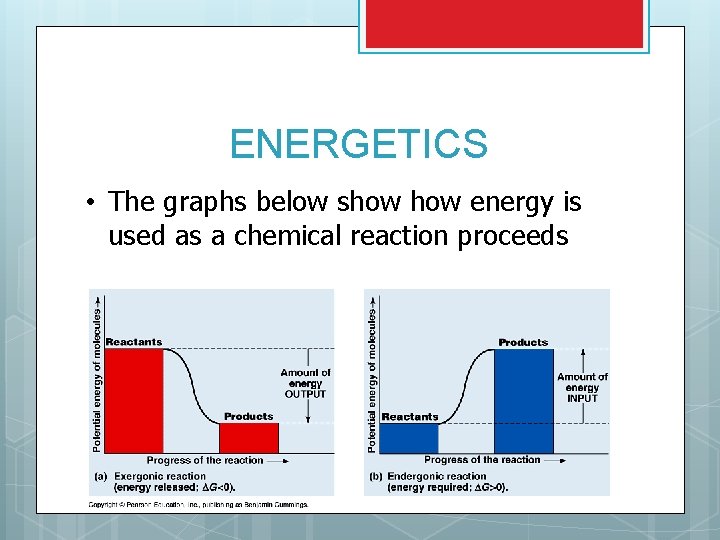
ENERGETICS • The graphs below show energy is used as a chemical reaction proceeds

GIBB’S FREE ENERGY Gibbs Free Energy Equation: δGrxn δG = δHrxn – TδSrxn (k. J): represents free energy δH (k. J): represents change in enthalpy T (K): temperature in kelvins δS (J/K): represents change in entropy

EXAMPLE N 2(g) + 3 H 2(g) 2 NH 3(g) 1. 2. 3. δHrxn = -91. 8 k. J δSrxn = -197 J/K T = 25°C Why does the entropy decrease in this reaction? What is the Gibb’s free energy? Is this reaction spontaneous?

STEP 1: WRITE WHAT YOU KNOW δG =? δH = -91. 8 k. J T = 25°C +273 = 298 K δS = -197 J/K = -0. 197 k. J/K NOTE: Temperature must be in Kelvin and entropy is converted to k. J so that it matches the units for δG and δH.

STEP 2: PUT THE NUMBERS IN THE FORMULA δGrxn = δHrxn – TδSrxn δGrxn = -91. 8 k. J – (298 K)(-0. 197 k. J/K) δGrxn = -91. 8 k. J – (-58. 7 k. J) δGrxn = -91. 8 k. J + 58. 7 k. J δGrxn = -33. 1 k. J

STEP 3: DETERMINE IF IT IS SPONTANEOUS 1. 2. 3. The reason why the entropy of this reaction decreased is that you are going from 4 moles of gas to 2 moles of gas. The amount of gas is reduced and lowers the entropy. δGrxn = -33. 1 k. J Since the Gibb’s free energy is negative, this is a spontaneous reaction.

TRY THIS A Chemical reaction has the following information: 1. 2. δHrxn = 145 k. J δSrxn = 322 J/K T = 109°C What is the Gibb’s free energy? Is this reaction spontaneous?

ANSWER 1. 2. δG = +22 k. J Since the Gibb’s free energy is positive, this reaction is NOT spontaneous Challenge: At what temperature would this reaction become spontaneous?

ANSWER We δHrxn = 145 k. J δSrxn = 322 J/K T = 109°C need to find what temperature δG<0 0 k. J = 145 k. J – T(0. 322 k. J/K) = 145 k. J T = 450 K the reaction has to be at least 450 K to be spontaneous

TRY THIS ΔH = 55 k. J ΔS = 225 J/K At what temperature does this reaction become spontaneous?
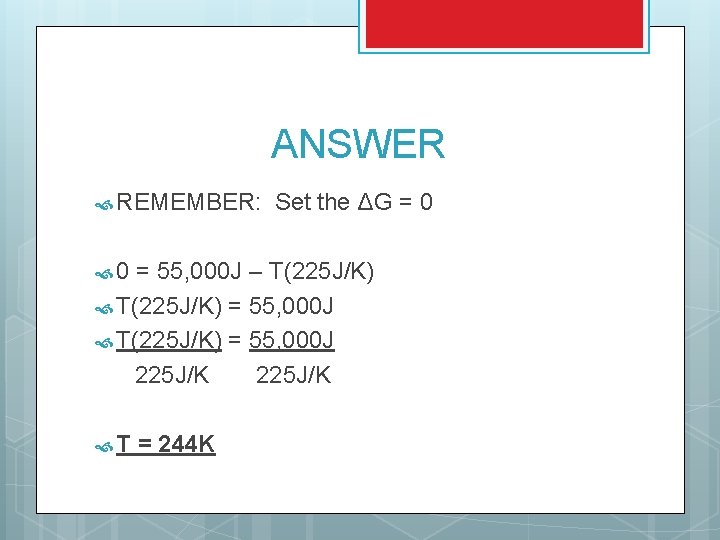
ANSWER REMEMBER: 0 Set the ΔG = 0 = 55, 000 J – T(225 J/K) = 55, 000 J 225 J/K T = 244 K

WHEN ARE REACTIONS SPONTANEOUS? Reactions are spontaneous when: δHrxn < 0; δSrxn > 0 δHrxn < 0; δSrxn < 0 (spontaneous at lower temperatures only) δHrxn > 0; δSrxn > 0 (spontaneous at higher temperatures only Reactions δHrxn are not spontaneous when: > 0; δSrxn < 0

ENERGETICS AND REACTIONS Even if spontaneous, reactions don’t just occur They need energy to start (Energy of activation) Many times this energy cost is very high and needs to be reduced to make the reaction work A catalyst is a substance that lowers the amount of energy needed to start a chemical reaction SEE GRAPH
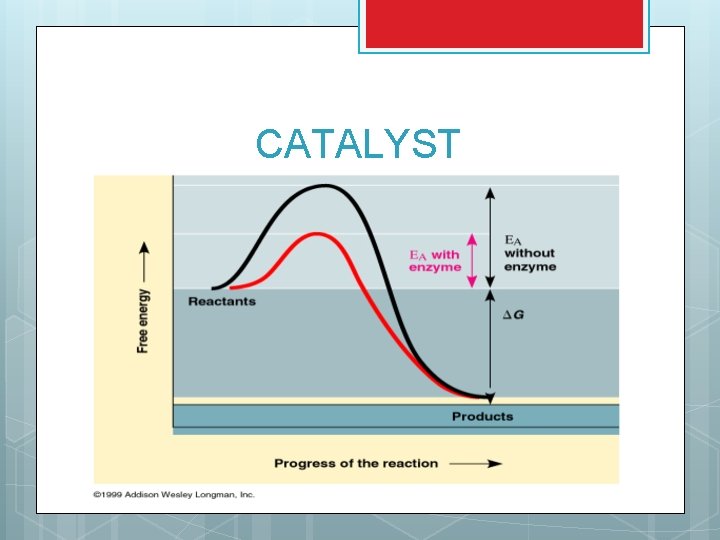
CATALYST

HOW CATALYSTS WORK Action of catalysts When reactions occur it is because molecules are smashing into each other Yet, they are like puzzle pieces and have to hit each other at an exact angle – EFFECTIVE COLLISION This takes energy to move the molecules around enough to hit at this angle A catalyst is a substance that “holds” molecules together so that it is easier to put them together

REACTION RATES Just because a reaction is spontaneous, it doesn’t mean that you’ll be able to see anything happening If a reaction is too slow, you won’t be able to see change for a long time A measure of how fast a reaction goes is called the REACTION RATE There are several factors that affect how fast a reaction goes and WHICH DIRECTION it goes

HOW DO REACTIONS WORK REMEMBER: When we talked about reactions we mentioned that a reaction can go either direction: Fe +O 2 Fe 2 O 3 The reaction can either go to the left or the right Actually, it is continually going left AND right

HOW DO REACTIONS WORK Fe +O 2 Fe 2 O 3 In this reaction iron and oxygen are making iron (III) oxide, but iron (III) oxide is also breaking apart at the SAME TIME The speed that this happens and which things are made or broken apart depends on many things The fact that they are constantly doing this is called DYNAMIC EQUILIBRIUM or CHEMICAL EQUILIBRIUM

CHEMICAL/DNYAMIC EQUILIBRIUM Reactions go to a certain ending that gives you a certain concentration of product At that point you won’t get a higher concentration, but the chemicals are forming or breaking apart at a rate that makes sure that the concentration is the same The reaction is still proceeding, but the concentrations are stay the same

HOW DO YOU KNOW WHICH WAY A REACTION GOES Le Chatelier was a scientist that studies reaction rates and which direction reactions go The things he found are called Le Chatelier’s Principles

EFFECTS OF CONCENTRATION PCl 5 PCl 3 + Cl 2 If you add more reactant (PCl 5), the reaction shifts to the right If you add more product (PCl 3 OR Cl 2), the reaction shifts to the left SUMMARY: If you add a higher concentration to one side of the reaction it shifts the reaction to make more of the other side

PRESSURE/VOLUME N 2(g) + H 2(g) NH 3(g) If you increase the pressure OR decrease the volume, the reaction shifts to the side with the fewest molecules (makes NH 3) If you decrease the pressure OR increase the volume, the reaction shifts to the side with the most molecules (N 2 and H 2)

TEMPERATURE 4 Fe(s) + 3 O 2(g) 2 Fe 2 O 3(s) + heat EXOTHERMIC REACTION: If the temperature is increased, the reaction will shift to the left (Fe and O 2) If the temperature is decreased, the reaction will shift to the right (Fe 2 O 3) THE OPPOSITE IS TRUE OF ENDOTHERMIC REACTIONS

EFFECTS OF CATLYST A catalyst will not shift the reaction either way It will just make sure that the reaction reaches equilibrium much faster Increased reaction rate
- Slides: 76