Energy Metabolism Energy metabolism Energy from the food

Energy Metabolism


Energy metabolism Energy from the food we eat is stored in the form of ATP is broken down to liberate the energy used to cause muscle contractions Anabolism— “to build up”; such as the use of amino acids to make proteins, which contribute to muscle mass Catabolism— “to break down”; such as breaking down glycogen to glucose molecules

Energy is stored in food in the form of carbohydrates, Fats and proteins. These basic food components can be broken down in out cells to release the stored energy. Energy production is both time and intensity related. Running at a very high intensity, as in sprinting, means that an athlete can operate effectively for only a very short period. Running at a low intensity, as in gentle jogging, means that an athlete can sustain activity for a long period.

Catabolism and Anabolism Catabolic reactions breakdown complex organic compounds providing energy (exergonic) glycolysis, Krebs cycle and electron transport Anabolic reactions synthesize complex molecules from small molecules requiring energy (endergonic) Exchange of energy requires use of ATP (adenosine triphosphate) molecule.

ATP - Adenosine Triphosphate: a complex chemical compound formed with the energy released from food and stored in all cells, particularly muscles. Only from the energy released by the breakdown of this compound can the cells perform work. The breakdown of ATP produces energy and ADP. CP - Creatine Phosphate: a chemical compound stored in muscle, which when broken down aids in the manufacture of ATP. The combination of ADP and CP produces ATP. LA - Lactic acid: a fatiguing metabolite of the lactic acid system resulting from the incomplete breakdown of glucose. O 2 means aerobic running in which ATP is manufactured from food mainly sugar and fat. This system produces ATP and is the prime energy source during endurance

ATP is the source of energy for muscle contraction Producing enough ATP is essential to performance Adaptations to exercise training involve energy metabolism The metabolic demands of training are important in designing training or exercise prescriptions

ATP IS COMPOSED OF A CARBON NITROGEN BASE CALLED ADENINE, A 5 -CARBON SUGAR CALLED RIBOSE AND THEREE PHOSPHATE.
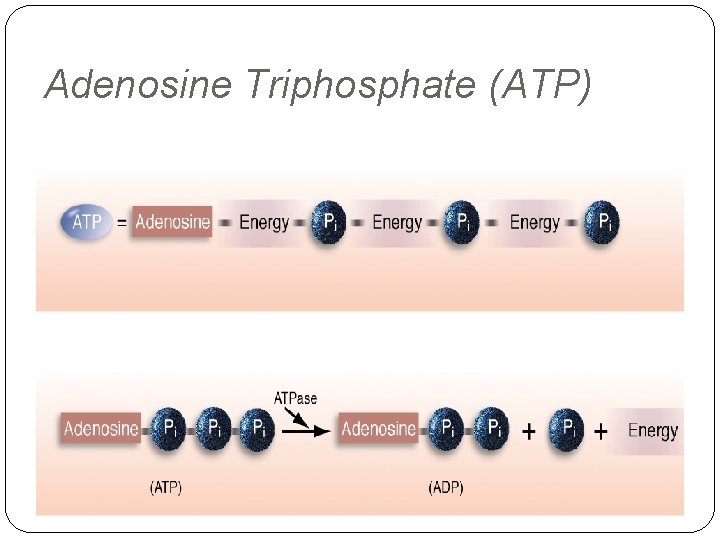
Adenosine Triphosphate (ATP)

The Alactic Anaerobic Energy System This energy system is the dominant source of muscle energy for high intensity explosive exercise that lasts for 10 seconds or less. For example, the alactic anaerobic energy system would be the main energy source for a 100 m sprint, or a short set of a weightlifting exercise. It can provide energy immediately, it does not require any oxygen (that's what "anaerobic" means), and it does not produce any lactic acid (that's what "alactic" means). It is also referred to as the ATP-PCr energy system or the phosphagen energy system.


Definitions of anaerobic and aerobic metabolism Aerobic metabolism is the production of ATP with oxygen. Anaerobic metabolism is the production of ATP without oxygen.

ATP production ATP can be produced aerobically or anaerobically Most physical activities involve both aerobic and anaerobic metabolism
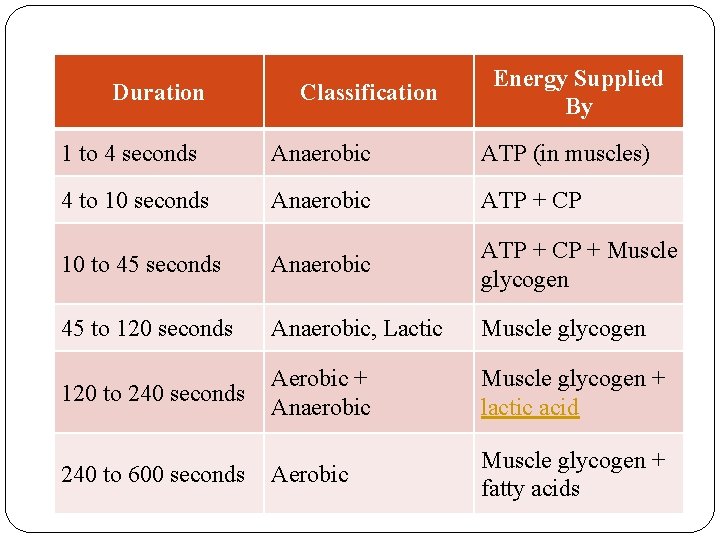
Duration Classification Energy Supplied By 1 to 4 seconds Anaerobic ATP (in muscles) 4 to 10 seconds Anaerobic ATP + CP 10 to 45 seconds Anaerobic ATP + CP + Muscle glycogen 45 to 120 seconds Anaerobic, Lactic Muscle glycogen 120 to 240 seconds Aerobic + Anaerobic Muscle glycogen + lactic acid Aerobic Muscle glycogen + fatty acids 240 to 600 seconds
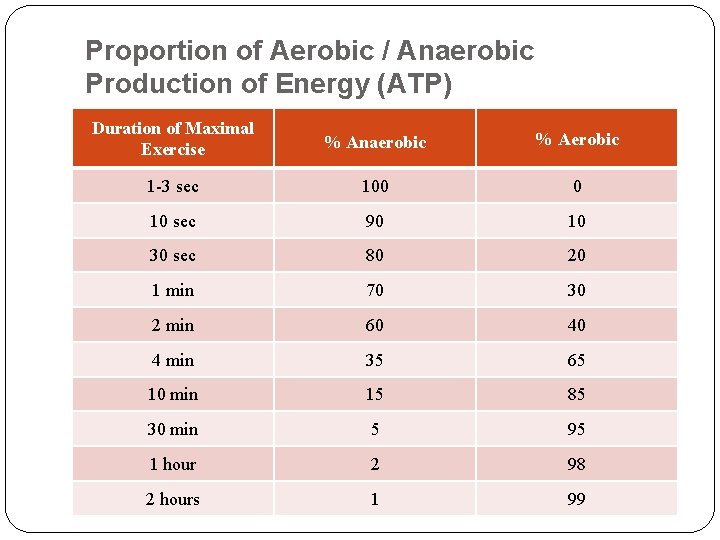
Proportion of Aerobic / Anaerobic Production of Energy (ATP) Duration of Maximal Exercise % Anaerobic % Aerobic 1 -3 sec 100 0 10 sec 90 10 30 sec 80 20 1 min 70 30 2 min 60 40 4 min 35 65 10 min 15 85 30 min 5 95 1 hour 2 98 2 hours 1 99
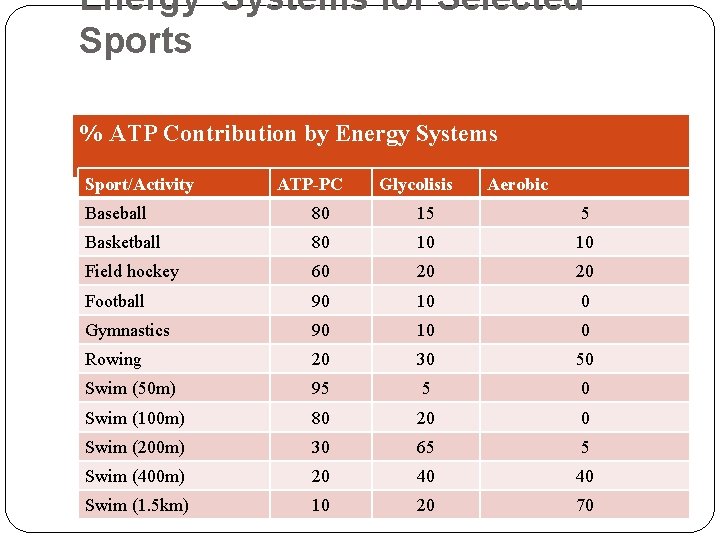
Energy Systems for Selected Sports % ATP Contribution by Energy Systems Sport/Activity ATP-PC Glycolisis Aerobic Baseball 80 15 5 Basketball 80 10 10 Field hockey 60 20 20 Football 90 10 0 Gymnastics 90 10 0 Rowing 20 30 50 Swim (50 m) 95 5 0 Swim (100 m) 80 20 0 Swim (200 m) 30 65 5 Swim (400 m) 20 40 40 Swim (1. 5 km) 10 20 70
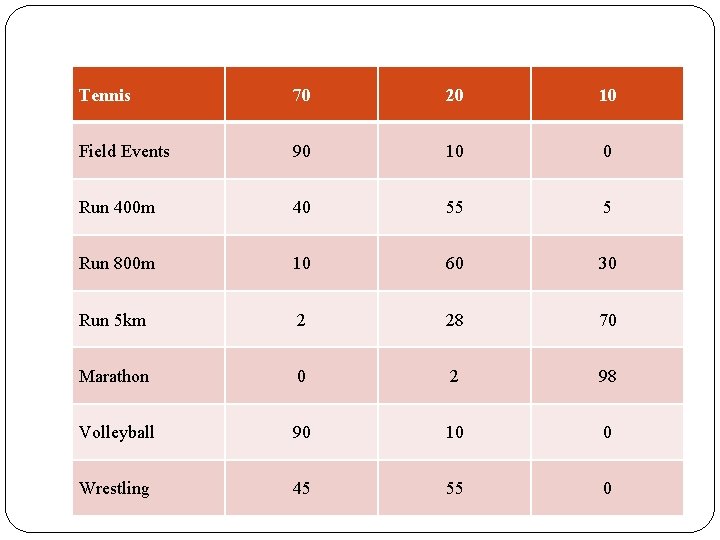
Tennis continued 70 20 10 Field Events 90 10 0 Run 400 m 40 55 5 Run 800 m 10 60 30 Run 5 km 2 28 70 Marathon 0 2 98 Volleyball 90 10 0 Wrestling 45 55 0
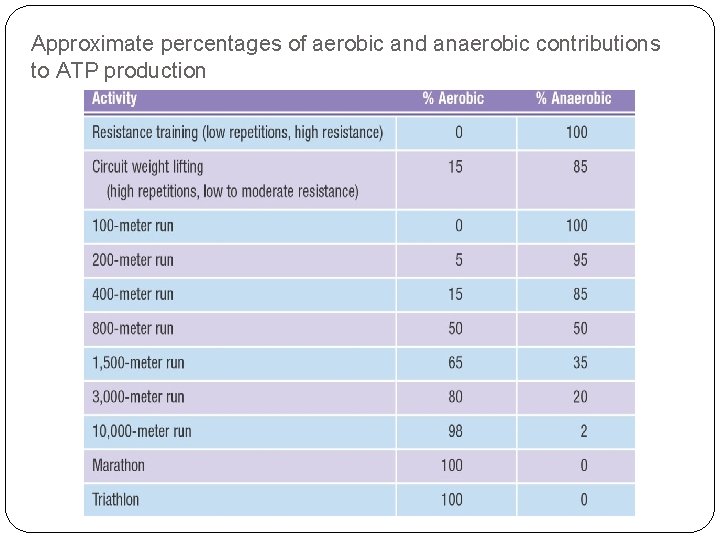
Approximate percentages of aerobic and anaerobic contributions to ATP production
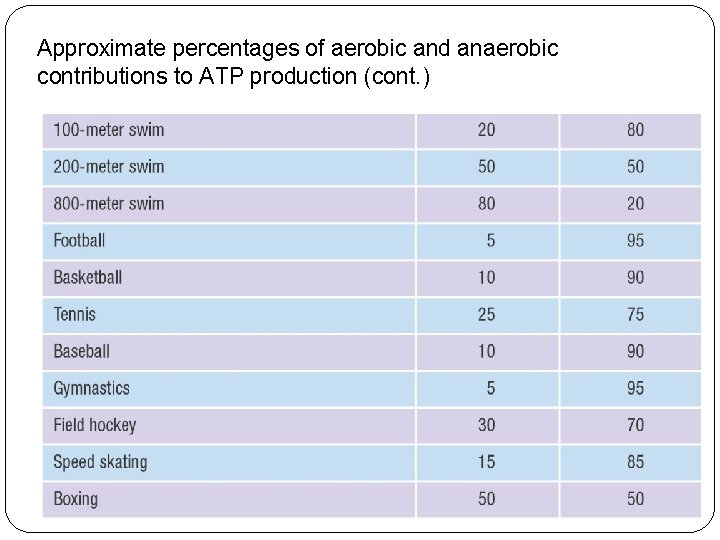
Approximate percentages of aerobic and anaerobic contributions to ATP production (cont. )

The three characteristics of enzymes Speed up or catalyze a reaction Are not changed by the reaction they cause Do not change the result of the reaction

Lactic Anaerobic Energy System This system is the dominant source of muscle energy for high intensity exercise activities that last up to approximately 90 seconds to 2 minutes. For example, 800 m sprint, 400 m. Essentially, this system is dominant when your alactic anaerobic energy system is depleted but you continue to exercise at an intensity that is too demanding for your aerobic energy system to handle. this system is also anaerobic and so it does not require any oxygen. However, this system produce lactic acid. It is also referred to as the lactic acid system or the anaerobic glycolytic system.

Summary of aerobic metabolism Of carbohydrates Anaerobic glycolysis precedes aerobic phases of ATP production Of fats (fatty acid oxidation) 1. Fatty acids are liberated from storage as a part of triglycerides 2. Long carbon chain fatty acids are metabolized through beta oxidation into two carbon acetyl coenzyme A molecules 3. These enter the Krebs cycle and go through the ETS for ATP production Of protein 1. Amino acids are converted into keto acids by the liver or muscle 2. Keto acids form substances that produce ATP through the Krebs cycle and ETS
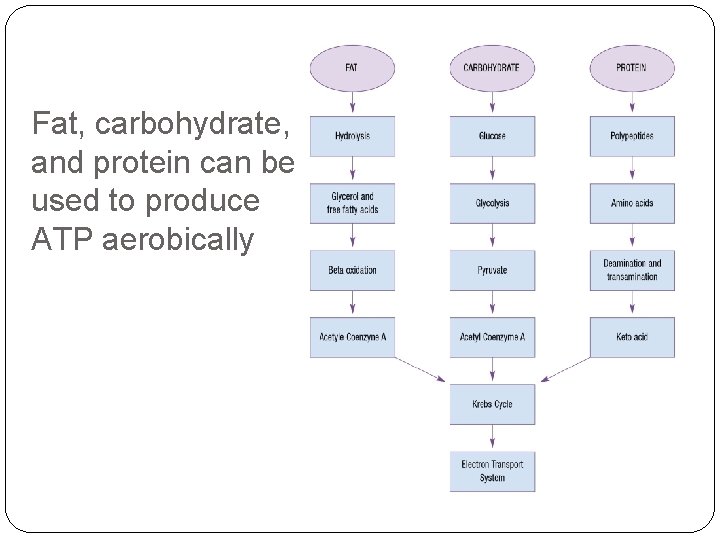
Fat, carbohydrate, and protein can be used to produce ATP aerobically
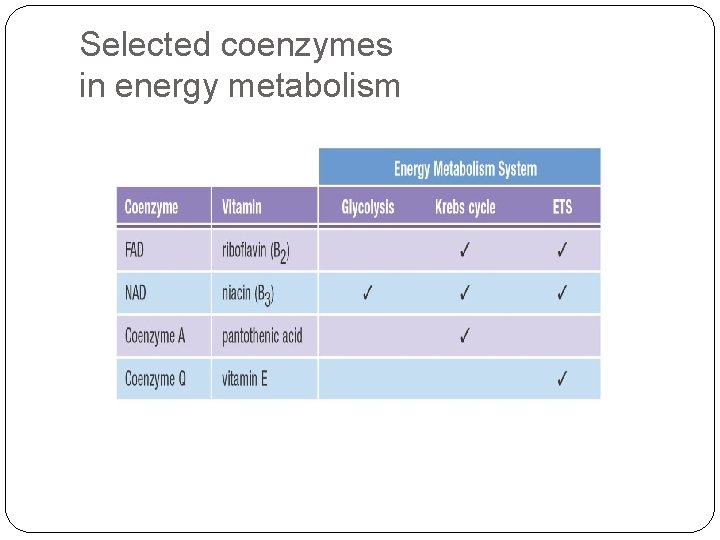
Selected coenzymes in energy metabolism

Anaerobic ATP production ATP can be produced anaerobically through two pathways: ATP-PC system Anaerobic glycolysis

The three primary enzymatic reactions that occur in the ATP-PC system Myosin 1. ATP ADP + inorganic ATPase phosphate (Pi) + energy Creatine 2. PC + ADP ATP + C Kinase (CK) Adenylate 3. 2 ADP ATP + AMP Kinase (AK)

Anaerobic glycolysis The primary system for ATP production for activities that last from approximately 20– 30 seconds to two to three minutes The breakdown of glucose to lactate without the use of oxygen



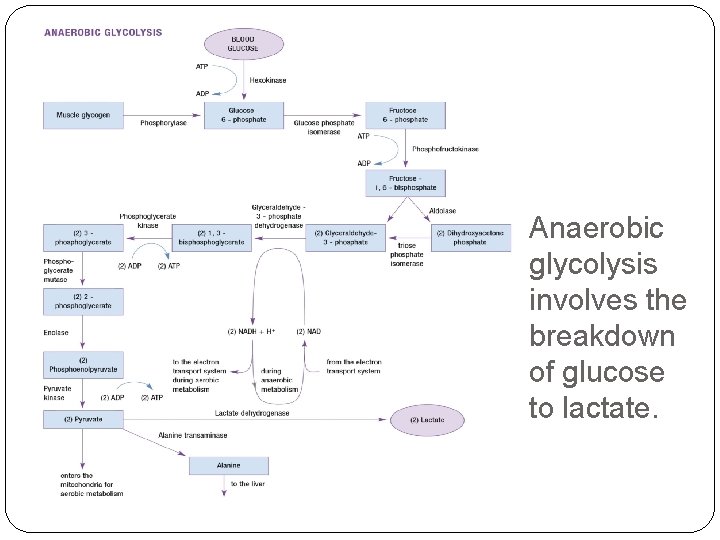
Anaerobic glycolysis involves the breakdown of glucose to lactate.
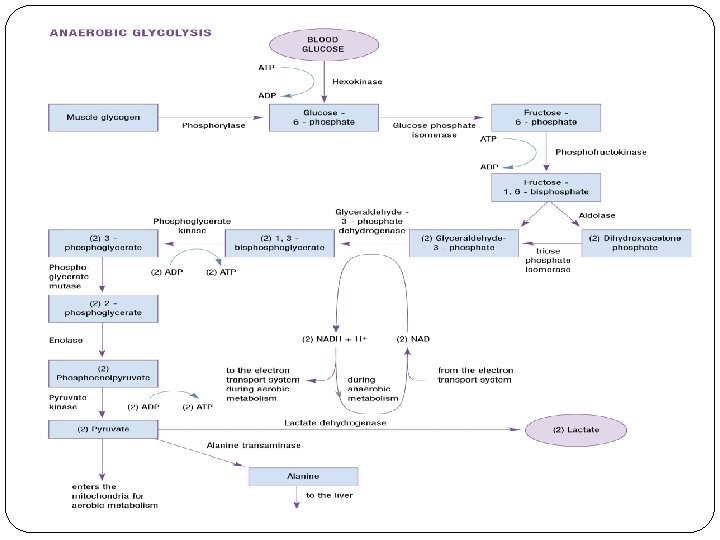

The reactants, enzymes, and products for the two steps in glycolysis where ATP is produced Step 1 Reactant: 1, 3 -bisphoglycerate Enzyme: phosphoglycerate kinase (PGK) Product: 3 -phosphoglycerate Step 2 Reactant: phosphoenolpyruvate Enzyme: pyruvate kinase (PK) Product: pyruvate

for the two steps in glycolysis where ATP is used Step 1 Reactant: blood glucose Enzyme: hexokinase (HK) Product: glucose-6 -phosphate Step 2 Reactant: fructose-6 -phosphate Enzyme: phosphofructokinase (PFK) Product: fructose-1, 6 -bisphosphate

The reactant, enzyme, and product for the step in glycolysis where NAD is reduced Reactant: glyceraldehyde 3 -phosphate Enzyme: glyceraldehyde 3 -phosphate dehydrogenase Product: 1, 3 -bisphoglycerate

The reactant, enzyme, and product for the step in glycolysis where NAD is oxidized Reactant: pyruvate Enzyme: lactate dehydrogenase Product: Lactate

The role of Phosphofructokinase (PFK) It is the rate-limiting enzyme in glycolysis It is the weak link—the rate of conversion of the reactant to product through enzymatic steps can proceed no faster than the rate-limiting enzyme will allow.

Aerobic metabolism of carbohydrates In the presence of sufficient oxygen, pyruvate from glycolysis enters muscle fiber mitochondria There, ATP is produced in the Krebs cycle and ETS Produces 38 molecules of ATP per molecule of glucose

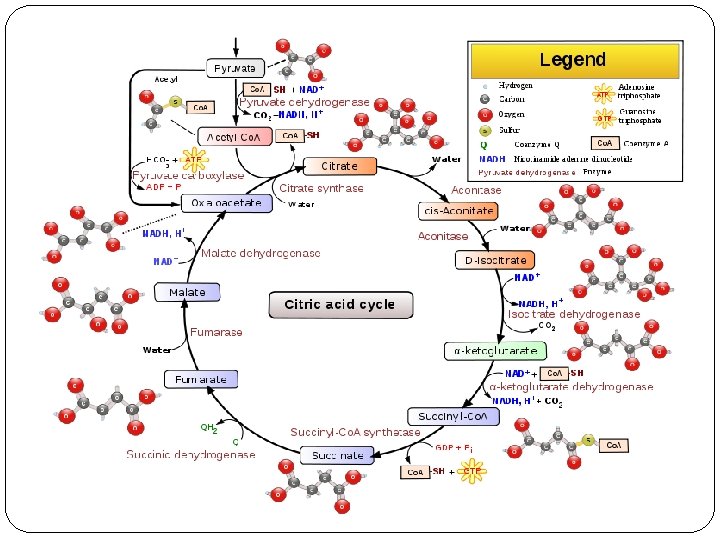
The Krebs cycle occurs within the mitochondria of the muscle fiber The Krebs cycle is a series of reactions which occurs in the mitochondria and results in the formation of ATP. The pyruvic acid molecules from glycolysis undergo oxidation in the mitochondrion to produce acetyl coenzyme A and then the Krebs cycle begins.

Three major events occur during the Krebs cycle. One guanosine triphosphate (GTP) is produced which donates a phosphate group to ADP to form one ATP; three molecules of Nicotinamide adenine dinucleotide (NAD) and one molecule of flavin adenine dinucleotide (FAD) are reduced. Although one molecule of GTP leads to the production of one ATP, the production of the reduced NAD and FAD are far more significant in the cell's energy generating process because they donate their electrons to an electron transport system that generates large amounts ATP.
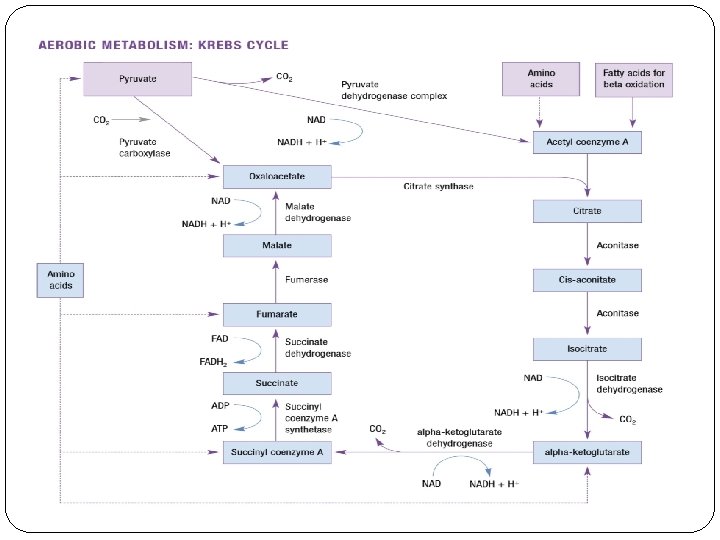

The four steps where NAD is reduced during the aerobic metabolization of carbohydrates Step 1 Reactant: pyruvate Enzyme: pyruvate dehydrogenase complex Product: acetyl coenzyme A Step 2 Reactant: isocitrate Enzyme: isocitrate dehydrogenase Product: alphaketoglutarate Step 3 Reactant: alpha- ketoglutarate Enzyme: alphaketoglutarate Product: succinyl coenzyme A Step 4 Reactant: malate Enzyme: malate dehydrogenase Product: oxaloacetate

The reactant, enzyme, and product for the step in the Kreb’s cycle where FAD is reduced Reactant: succinate Enzyme: succinate dehydrogenese Product: fumarate

The reactant, enzyme, and product in the Kreb’s Cycle where ATP is produced Reactant: succinyl coenzyme A Enzyme: succinyl coenzyme A synthetase Product: succinate

The electron transport system The part of aerobic metabolism where 34 of the 38 ATP are produced

What is the respiratory chain? The Krebs cycle and the electron transport system (ETS), where ATP is produced and oxygen is utilized.
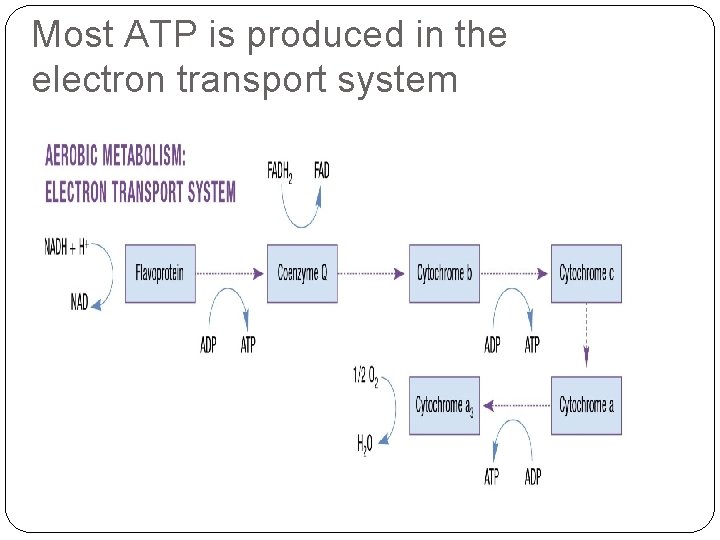
Most ATP is produced in the electron transport system

Anaerobic breakdown of glucose results in the net production of only 2 ATP, while aerobic metabolism nets 38 ATP
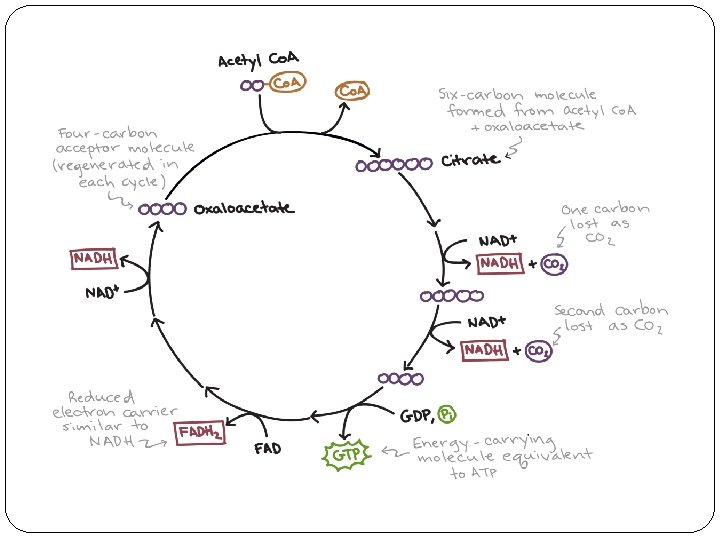

Steps of the citric acid cycle Step 1. In the first step of the citric acid cycle, acetyl Co. A joins with a four-carbon molecule, oxaloacetate, releasing the Co. A, group and forming a six-carbon molecule called citrate. Step 2. In the second step, citrate is converted into its isomer, isocitrate. This is actually a two-step process, involving first the removal and then the addition of a water molecule, which is why the citric acid cycle is sometimes described as having nine steps—rather than the eight listed here

Step 3. In the third step, isocitrate is oxidized and releases a molecule of carbon dioxide, leaving behind a five-carbon molecule—α-ketoglutarate. During this step, NAD+ is reduced to form NADHN. The enzyme catalyzing this step, isocitrate dehydrogenase, is important in regulating the speed of the citric acid cycle.

Step 4. The fourth step is similar to the third. In this case, it’s α-ketoglutarate that’s oxidized, reducing NAD+to NADH and releasing a molecule of carbon dioxide in the process. The remaining fourcarbon molecule picks up Coenzyme A, forming the unstable compound succinyl. Co. A. The enzyme catalyzing this step, α-ketoglutarate dehydrogenase, is also important in regulation of the citric acid cycle.

Step 5. In step five, Co. A of succinyl Co. A is replaced by a phosphate group, which is then transferred to ADP to make ATP. In some cells, GDP, guanine diphosphate—is used instead of ADP, forming GTP— guanine triphosphate—as a product. The four-carbon molecule produced in this step is called suc Step 6. In step six, succinate is oxidized, forming another four-carbon molecule called fumarate. In this reaction, two hydrogen atoms—with their electrons— are transferred to FAD , producing FADH 2. The enzyme that carries out this step is embedded in the inner membrane of the mitochondrion, so FADH 2 can transfer its electrons directly into the electron transport chain.

Step 7. In step seven, water is added to the four-carbon molecule fumarate, converting it into another fourcarbon molecule called malate. Step 8. In the last step of the citric acid cycle, oxaloacetate—the starting four-carbon compound—is regenerated by oxidation of malate. Another molecule of NAD+ is reduced to NADH in the process.

Products of the citric acid cycle In a single turn of the cycle, two carbons enter from acetyl Co. A and two molecules of carbon dioxide are released. three molecules of NADH and one molecule of FADH 2 are generated; and one molecule of ATP or GTP is produced. These figures are for one turn of the cycle, corresponding to one molecule of acetyl Co. A. Each glucose produces two acetyl Co. A molecules, so we need to multiply these numbers by 2 if we want the per -glucose yield.

10 NADH and 2 FADH 2 participates in ETC. Each NADH produced 3 ATP and each FADH 2 produced 2 ATP
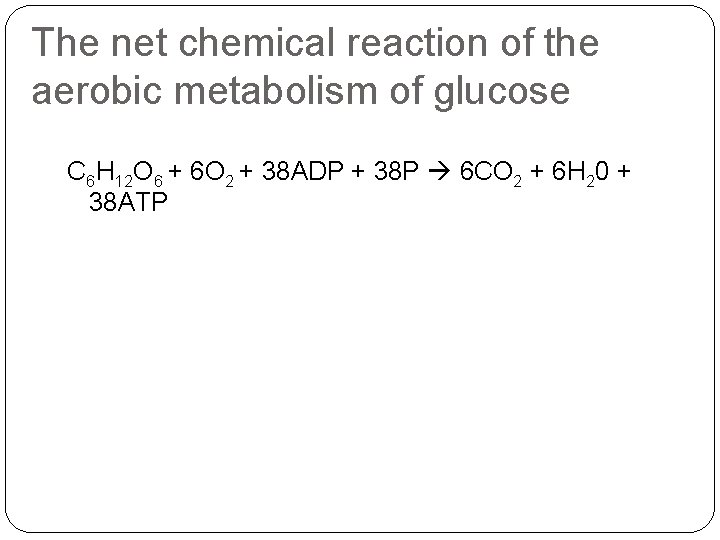
The net chemical reaction of the aerobic metabolism of glucose C 6 H 12 O 6 + 6 O 2 + 38 ADP + 38 P 6 CO 2 + 6 H 20 + 38 ATP

Comparison of the power and capacity of the various energy production systems The ATP-PC system has low capacity because there is a limited store of phosphagens available. Carbohydrate oxidation is limited by glycogen depletion. Fatty acid metabolism has the greatest capacity because, under normal conditions, each person has an inexhaustible supply of energy-rich fats.

Fat Metabolism Fat is found in many common foods. Fat, in the form of triglyceride (sometimes known as triacylglycerol), is the major storage form of energy in humans. Some triglyceride is stored within muscle cells (Figure 2. 13), but the vast majority is deposited in adipose cells which comprise at least 10– 15% of the body weight of average young males and 20– 25% of the body weight of average young females.

Fat is an excellent storage fuel for several reasons. First, fat is an energy-dense fuel yielding 9. 13 kcal. g− 1; both carbohydrate and protein yield slightly less than 4 kcal. g− 1. It is easy to appreciate the difference by looking at the chemical composition of the free fatty acid palmitate, which is C 16 H 32 O 2. This fatty acid has almost three times as much C and H, but only a third as much O as glucose (C 6 H 12 O 6). Remember that it is H that donates the electrons (e−) and protons (H+) used during electron transport and oxidative phosphorylation.

Fatty acids may be saturated, unsaturated, or Polyunsaturated. A saturated fatty acid has a chemical bonding arrangement that allows it to hold as many hydrogen atoms as possible. Thus, the term “saturated” means “saturated with hydrogen. ” Unsaturated fatty acids have a chemical bonding arrangement with a reduced- hydrogen binding potential and therefore are unsaturated with respect to hydrogen atoms. Polyunsaturated means several bonds are without hydrogen.

The breakdown of triglycerides into glycerol and fatty acids is catalyzed by the enzyme hormone-sensitive lipase. The glycerol is soluble in blood, but the free fatty acids (FFA) are not. Glycerol can enter glycolysis in the cytoplasm (as 3 -phosphoglycerate, the product of step 7 in Figure 2. 6), but it is not typically utilized by muscle cells in this way. The direct role of glycerol as a fuel in the muscle cells during exercise is so minor that it need not be considered. However, glycerol can be converted to glucose by the liver.

FFA must be transported in the blood bound to albumin. Specifi c receptor sites on the muscle cell membrane take up the FFA into the cell. The FFA must then be translocated or transported from the cytoplasm into the mitochondria. Once in the mitochondrial matrix, the FFA undergoes the process of beta-oxidation.

Beta-oxidation is a cyclic series of steps that breaks off successive pairs of carbon atoms from FFA, which are then used to form acetyl Co. A. Remember that acetyl Co. A is the common intermediate by which all foodstuffs enter the Krebs cycle and ETS/OP stage. The number of cycles depends upon the number of carbon atoms; most fatty acids have 14– 24 carbons. When there is an adequate supply of oxaloacetate to combine with, the fat-derived acetyl Co. A enters the Krebs cycle and proceeds through electron transport and oxidative phosphorylation.

As with glycolysis, ATP is used for activation; but unlike glycolysis, beta-oxidation produces no ATP directly by substrate phosphorylation.

The Steps of Beta-oxidation Step 1. The fatty acid molecule is activated by the breakdown of 1 ATP to AMP, releasing the energy equivalent of 2 ATP if broken down as it is normally to ADP. Concurrently, coenzyme A is added. Step 2. FAD is reduced to FADH 2. The FADH 2 enters the electron transport chain and produces 1. 5 ATP (actual count).

Step 3. A molecule of water is added, and NAD+ is reduced to NADH + H+. The NADH + H+ will enter the electron transport chain and produce 2. 5 ATP (actual count). Steps 2 and 3, with the removal of the hydrogen atoms, account for the oxidation portion of the name for this process. Step 4. The bond between the alpha (a) carbon (C 2) and the beta (b) carbon (C 3) is broken, resulting in the removal of two carbon atoms (C 1 and C 2), which are then used to form acetyl coenzyme A. The cleavage of carbons at the site of the beta carbon explains why the process is called beta-oxidation.

Step 5. Steps 1– 4 are repeated for each pair of carbons except the last, since the last unit formed is acetyl Co. A itself. Therefore, the number of cycles that must be completed to oxidize the fat can be computed using the formula n/2– 1, where n is the number of carbons. The acetyl Co. A can enter the Krebs cycle and electron transport system
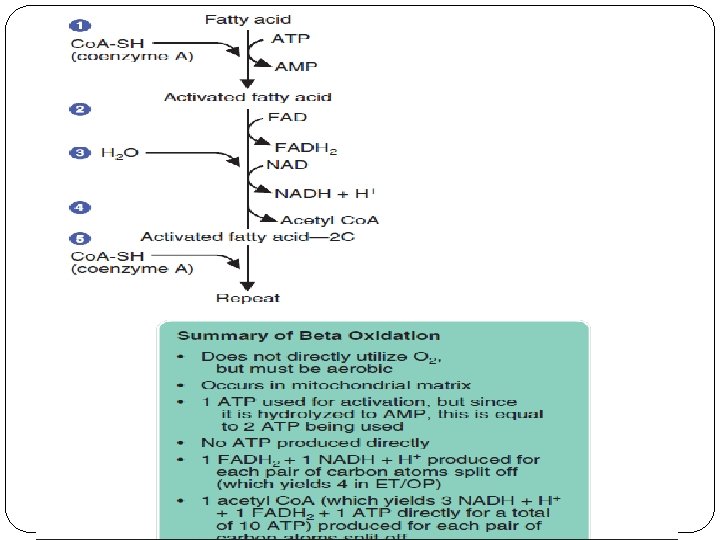

ATP Production from Fatty Acids The number of ATP produced from the breakdown of fat depends on the fatty acid utilized. The following example shows a calculation of ATP production by actual count for palmitate. As with carbohydrate, the actual number of ATP molecules produced is lower than theoretical number would be.


http: //highered. mheducation. com/sites/0072507470/st udent_view 0/chapter 25/animation__how_the_krebs_c ycle_works__quiz_1_. html https: //www. khanacademy. org/test- prep/mcat/biomolecules/krebs-citric-acid-cycle-andoxidative-phosphorylation/v/electron-transport-chain http: //www. science. smith. edu/departments/Biology/Bi o 231/etc. html
- Slides: 73