Energy Levels Orbitals The Hydrogen Orbitals Orbitals do

Energy Levels & Orbitals
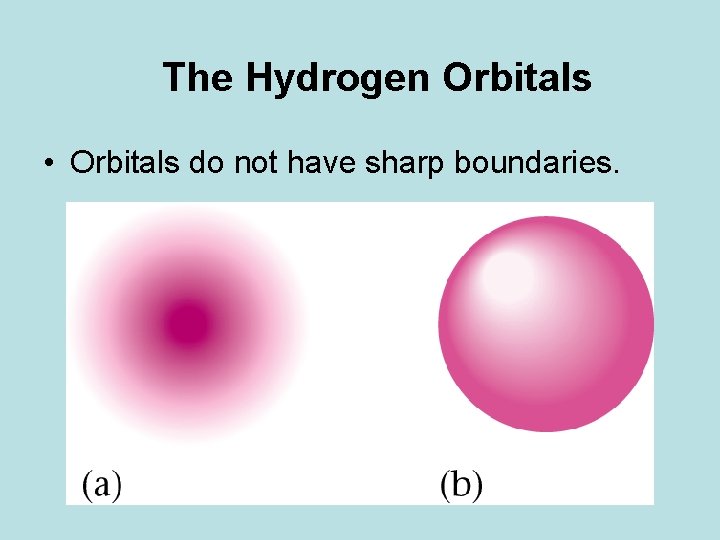
The Hydrogen Orbitals • Orbitals do not have sharp boundaries.

The Hydrogen Orbitals • Hydrogen has discrete energy levels. – Called principal energy levels – Labeled with whole numbers photon - An elementary particle, the quantum of the electromagnetic interaction and the basic unit of light. In other words a photon is a little packet of energy which can carry light.
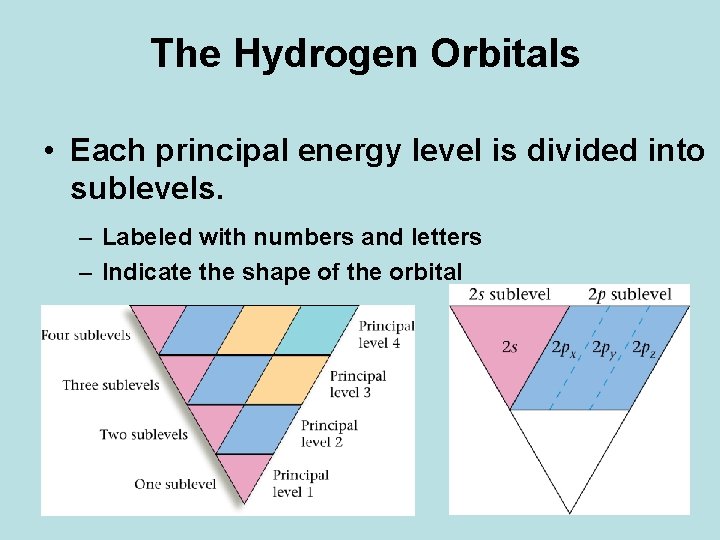
The Hydrogen Orbitals • Each principal energy level is divided into sublevels. – Labeled with numbers and letters – Indicate the shape of the orbital
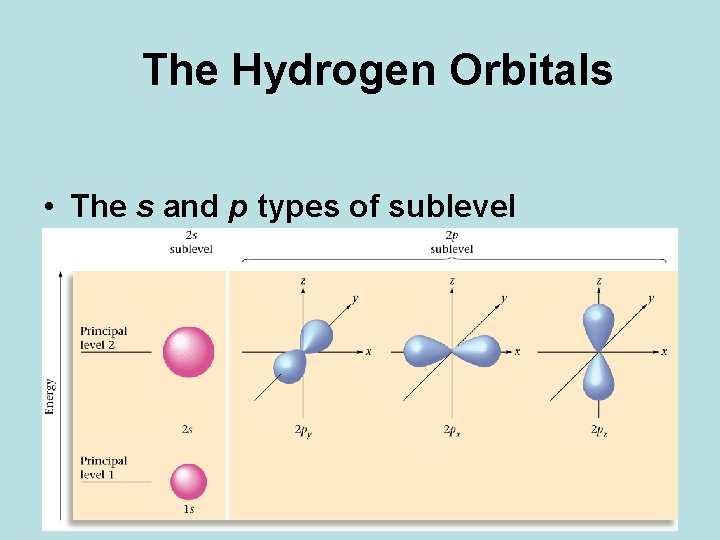
The Hydrogen Orbitals • The s and p types of sublevel

The Hydrogen Orbitals • Why does an H atom have so many orbitals and only 1 electron? – An orbital is a potential space for an electron. – Atoms can have many potential orbitals.

The Wave Mechanical Model: Further Development Atoms Beyond Hydrogen • The Bohr model was discarded because it does not apply to all atoms.

The Wave Mechanical Model: Further Development • Pauli Exclusion Principle - an atomic orbital can hold a maximum of 2 electrons and those 2 electrons must have opposite spins

General Rules • Pauli Exclusion Principle – Each orbital can hold TWO electrons with opposite spins.

General Rules • Hund’s Rule – Within a sublevel, place one e- per orbital before pairing them. – “Empty Bus Seat Rule” WRONG RIGHT
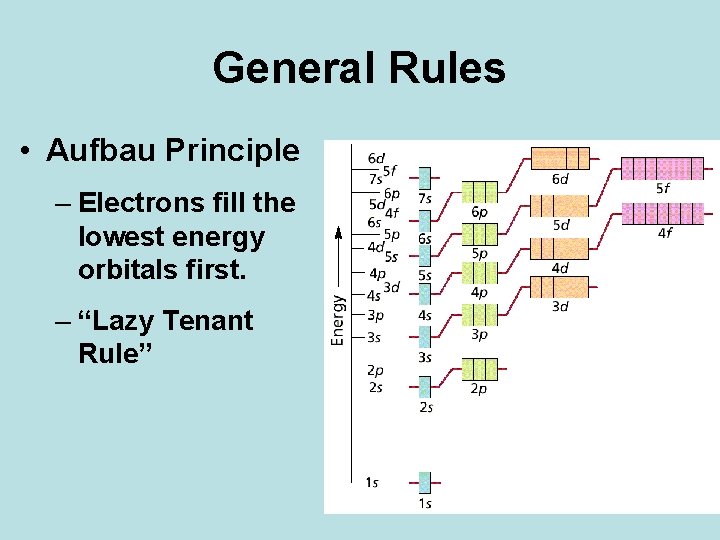
General Rules • Aufbau Principle – Electrons fill the lowest energy orbitals first. – “Lazy Tenant Rule”
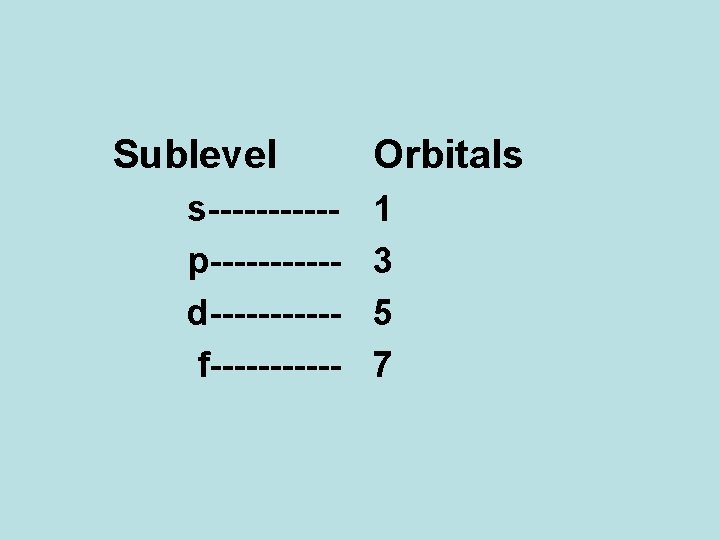
Sublevel s-----p-----d-----f------ Orbitals 1 3 5 7

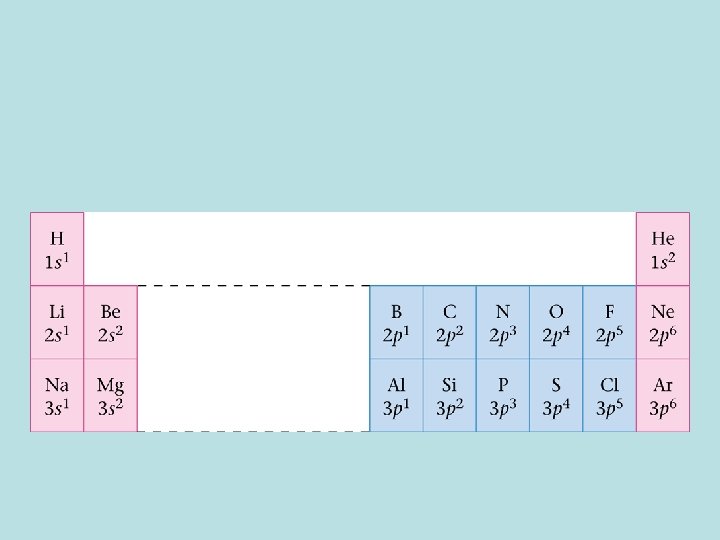
Electron Arrangements in the First 18 Atoms on the Periodic Table Classifying Electrons • Valence electrons – electrons in the outermost (highest) principal energy level of an atom • Core electrons – inner electrons • Elements with the same valence electron arrangement show very similar chemical behavior.
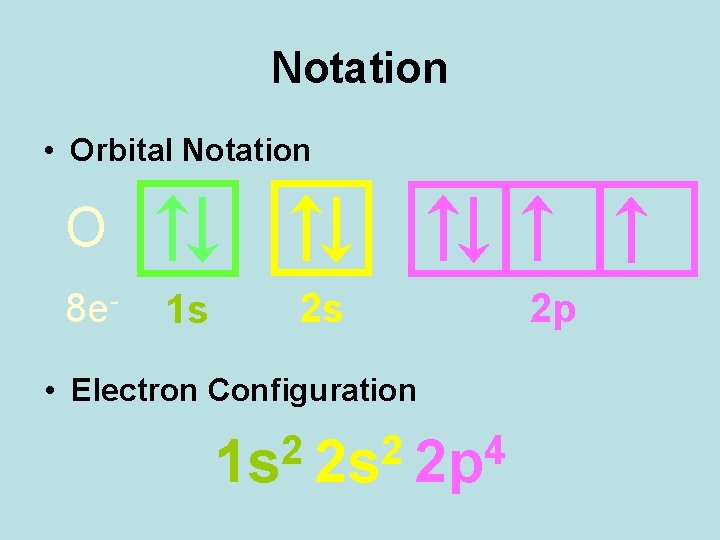
Notation • Orbital Notation O 8 e- 1 s 2 s • Electron Configuration 2 2 4 1 s 2 s 2 p 2 p
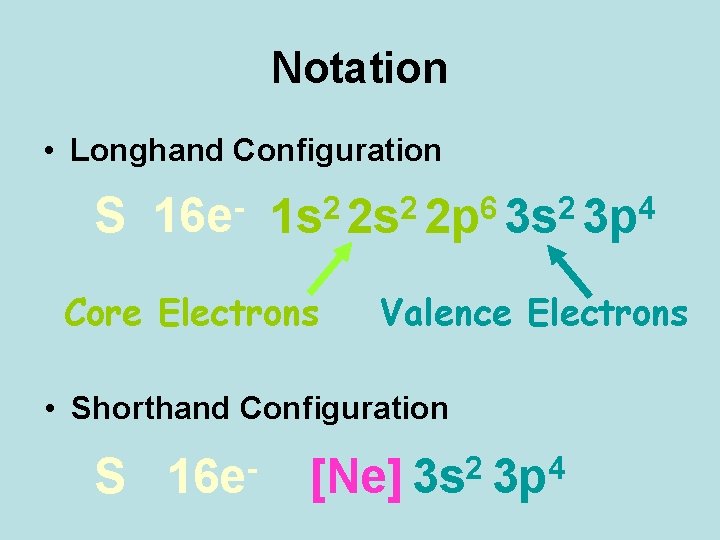
Notation • Longhand Configuration S 16 e 6 2 2 2 4 1 s 2 s 2 p 3 s 3 p Core Electrons Valence Electrons • Shorthand Configuration S 16 e 2 4 [Ne] 3 s 3 p
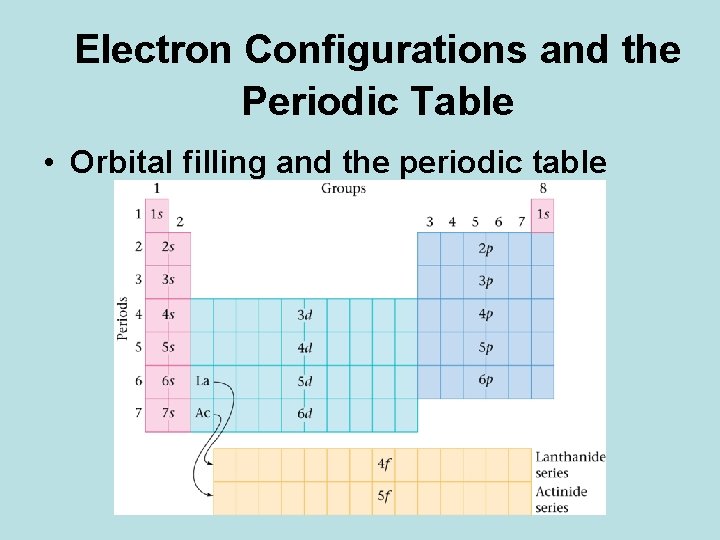
Electron Configurations and the Periodic Table • Orbital filling and the periodic table
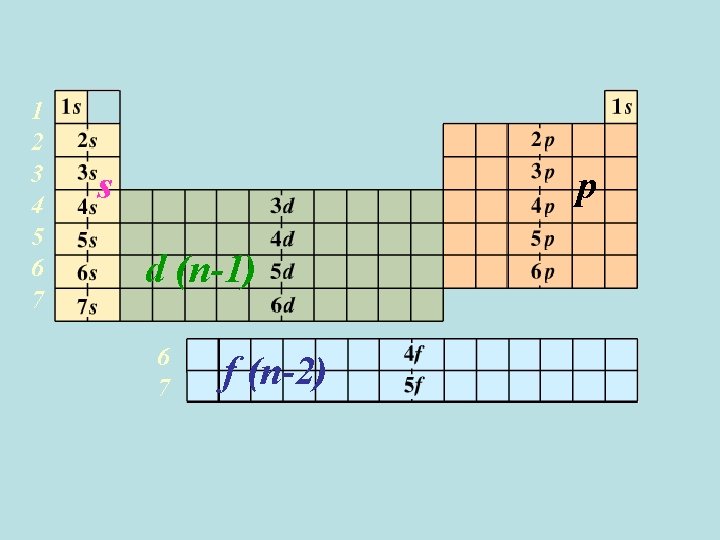
1 2 3 4 5 6 7 s p d (n-1) 6 7 f (n-2)
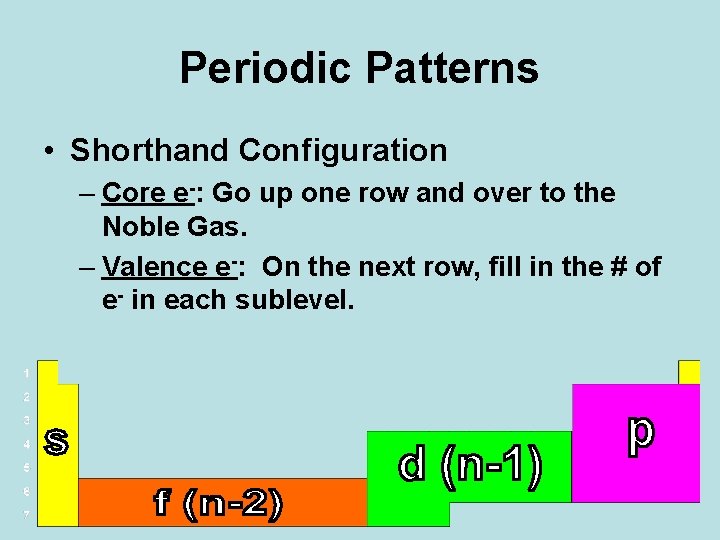
Periodic Patterns • Shorthand Configuration – Core e-: Go up one row and over to the Noble Gas. – Valence e-: On the next row, fill in the # of e- in each sublevel.
![Periodic Patterns • Example - Germanium [Ar] 2 10 2 4 s 3 d Periodic Patterns • Example - Germanium [Ar] 2 10 2 4 s 3 d](http://slidetodoc.com/presentation_image_h2/b7aa26197b709fb67b7c012dcfeb5cc2/image-21.jpg)
Periodic Patterns • Example - Germanium [Ar] 2 10 2 4 s 3 d 4 p

• H atom – Electron configuration – electron arrangement – 1 s 1 – Orbital diagram – orbital is a box grouped by sublevel containing arrow(s) to represent electrons

• He atom – Electron configuration– 1 s 2 – Orbital diagram
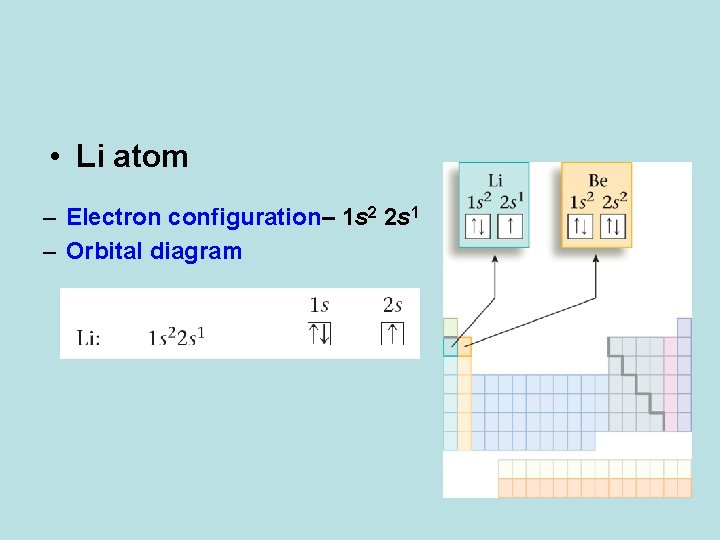
• Li atom – Electron configuration– 1 s 2 2 s 1 – Orbital diagram
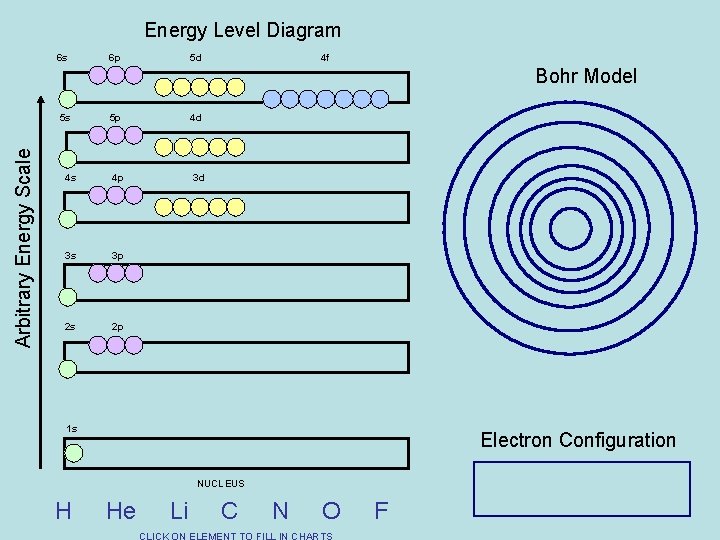
Energy Level Diagram 6 s 6 p 5 d 4 f Bohr Model Arbitrary Energy Scale 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N O F
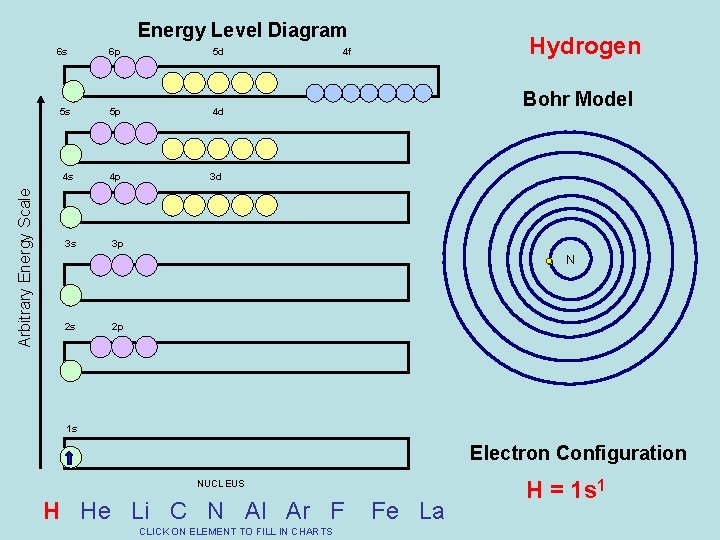
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Hydrogen 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La H = 1 s 1
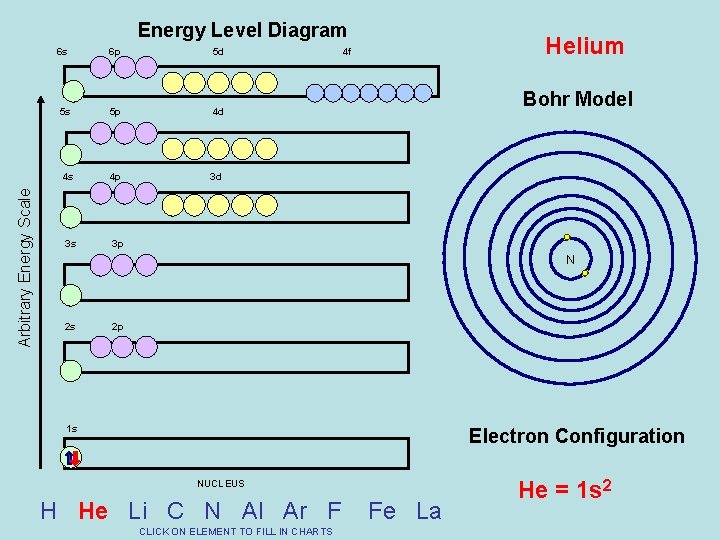
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Helium 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La He = 1 s 2
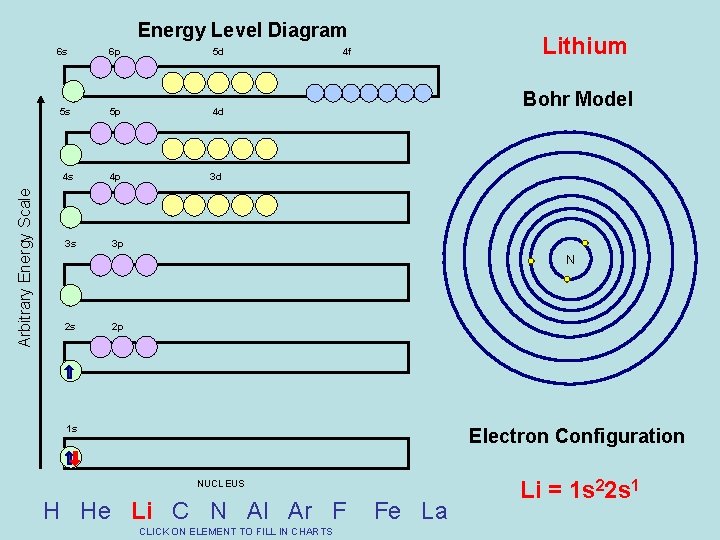
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Lithium 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Li = 1 s 22 s 1
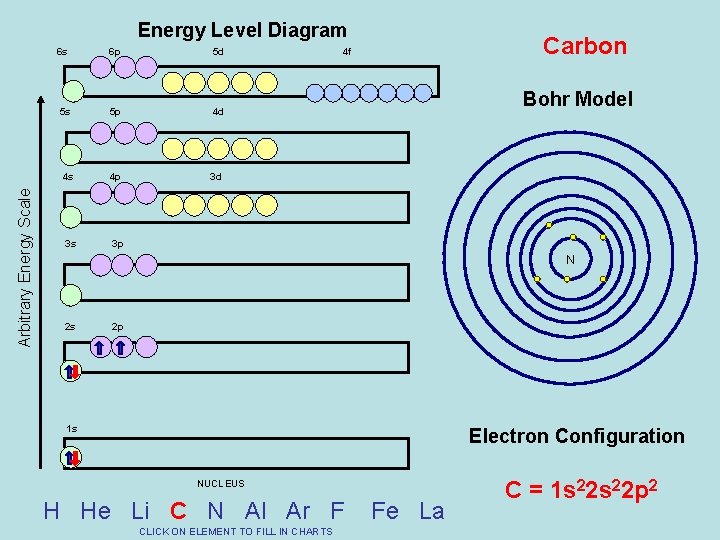
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Carbon 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La C = 1 s 22 p 2
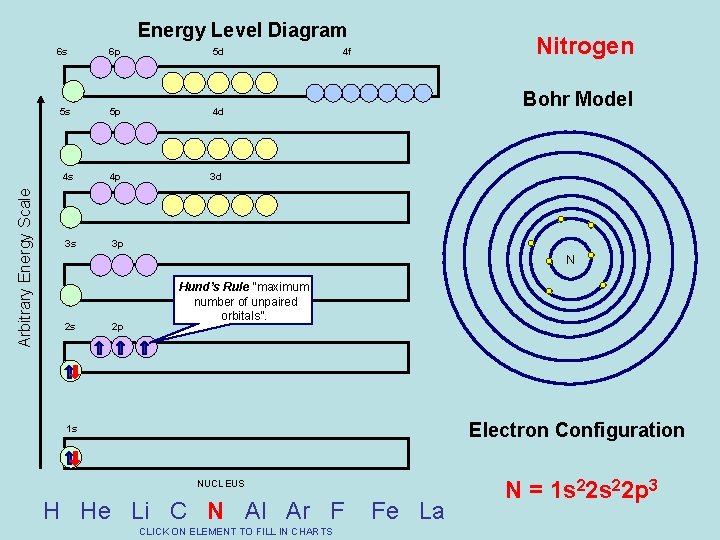
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Nitrogen 4 f Bohr Model N 2 s 2 p Hund’s Rule “maximum number of unpaired orbitals”. Electron Configuration 1 s NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La N = 1 s 22 p 3
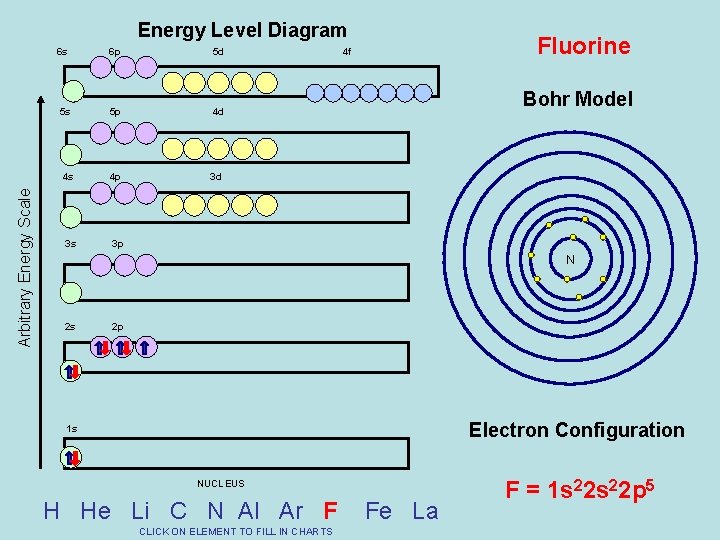
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Fluorine 4 f Bohr Model N 2 s 2 p Electron Configuration 1 s NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La F = 1 s 22 p 5
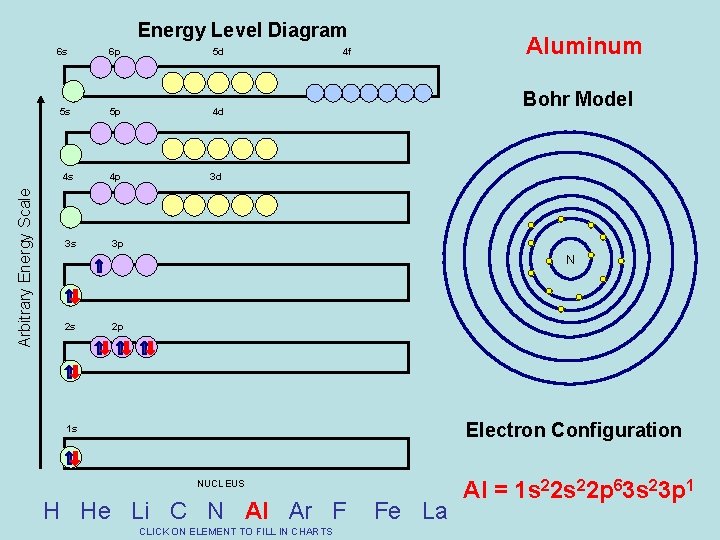
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Aluminum 4 f Bohr Model N 2 s 2 p Electron Configuration 1 s NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Al = 1 s 22 p 63 s 23 p 1
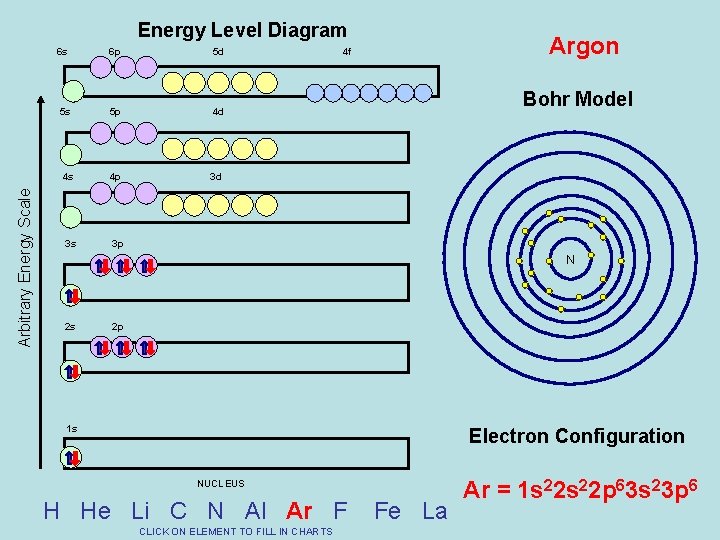
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p Argon 4 f Bohr Model N 2 s 2 p 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe La Ar = 1 s 22 p 63 s 23 p 6
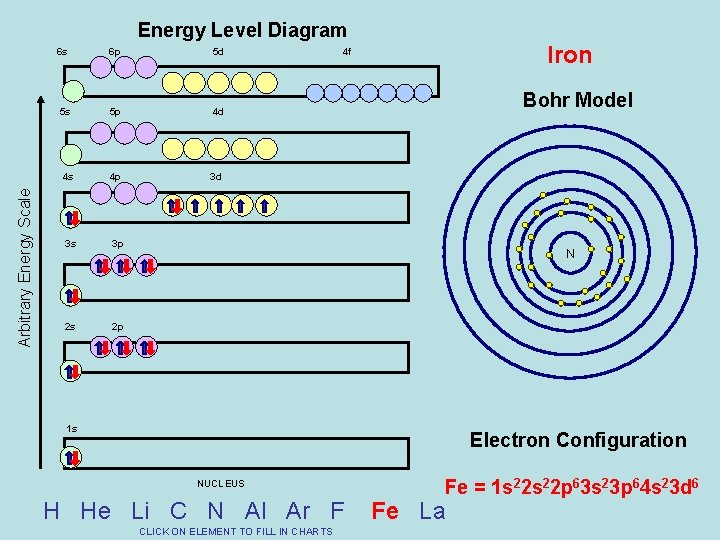
Arbitrary Energy Scale Energy Level Diagram 6 s 6 p 5 d 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p 2 s 2 p Iron 4 f Bohr Model N 1 s Electron Configuration NUCLEUS H He Li C N Al Ar F CLICK ON ELEMENT TO FILL IN CHARTS Fe = 1 s 22 p 63 s 23 p 64 s 23 d 6 Fe La
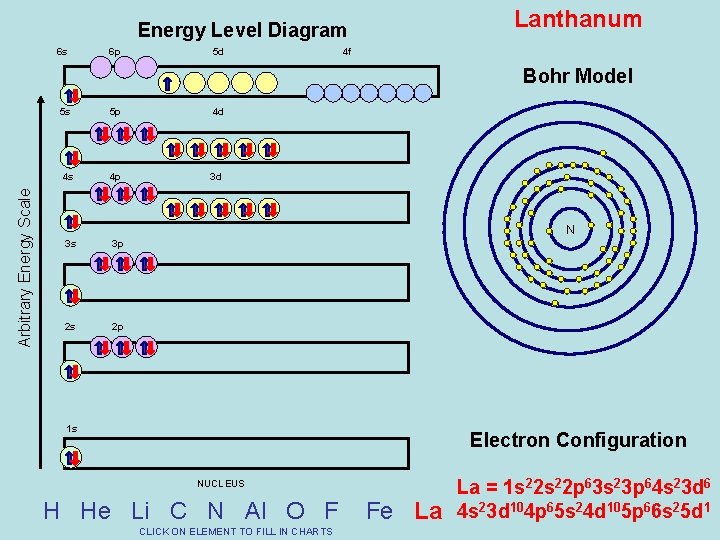
Lanthanum Energy Level Diagram 6 s 6 p 5 d 4 f Bohr Model Arbitrary Energy Scale 5 s 5 p 4 d 4 s 4 p 3 d 3 s 3 p 2 s 2 p N 1 s Electron Configuration NUCLEUS H He Li C N Al O F CLICK ON ELEMENT TO FILL IN CHARTS Fe La La = 1 s 22 p 63 s 23 p 64 s 23 d 6 4 s 23 d 104 p 65 s 24 d 105 p 66 s 25 d 1

B. Electron Configurations and the Periodic Table • Look at electron configurations for K through Kr

Electron Configurations and the Periodic Table

C. Periodic Patterns • Period # – energy level (subtract for d & f) • A/B Group # – total # of valence e- • Column within sublevel block – # of e- in sublevel
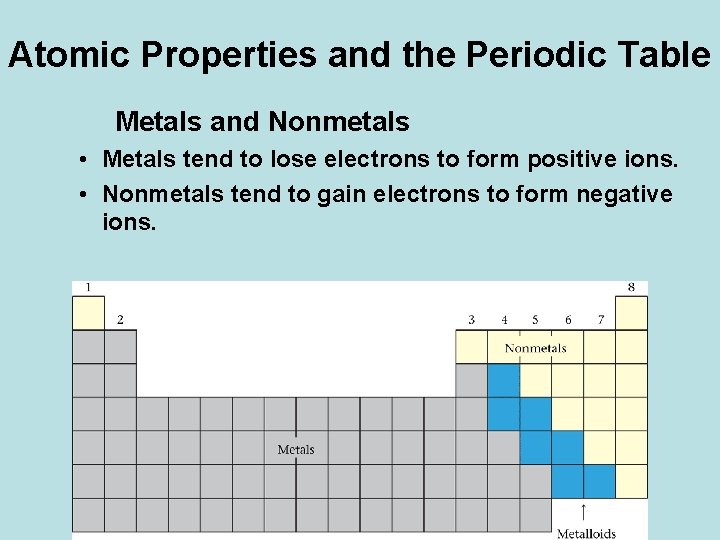
Atomic Properties and the Periodic Table Metals and Nonmetals • Metals tend to lose electrons to form positive ions. • Nonmetals tend to gain electrons to form negative ions.

Atomic Properties and the Periodic Table Atomic Size • Size tends to increase down a column. • Size tends to decrease across a row.

Atomic Properties and the Periodic Table Ionization Energies • Ionization Energy – energy required to remove an electron from an individual atom (gas) – Tends to decrease down a column – Tends to increase across a row

- Slides: 42