Energy Fundamentals Energy fundamentals Energy an intangible phenomenon

Energy Fundamentals

Energy fundamentals Energy: an intangible phenomenon that can change the position, physical composition, temperature of matter Involved in biological, chemical, physical processes Potential energy: energy of position Kinetic energy: energy of motion Chemical energy: potential energy held in the bonds between atoms Changing potential into kinetic energy Releases energy Produces motion, action, or heat

Potential vs. kinetic energy Potential energy stored in our food becomes kinetic energy when we exercise and releases carbon dioxide, water, and heat as by-products Insert Figure 2. 11

Energy is conserved but changes in quality First law of thermodynamics: energy can change form but cannot be created or destroyed Second law of thermodynamics: energy changes from a more-ordered to a less-ordered state Entropy: an increasing state of disorder Living organisms resist entropy by getting energy from food and photosynthesis Dead organisms get no energy and through decomposition lose their organized structure

The sun’s energy powers living systems Energy that powers Earth’s ecological systems comes mainly from the sun The sun releases radiation from the electromagnetic spectrum Some is visible light
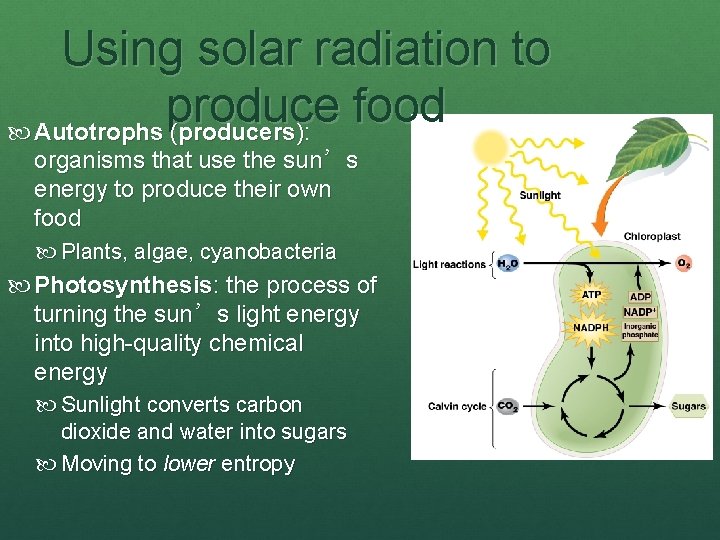
Using solar radiation to produce food Autotrophs (producers): organisms that use the sun’s energy to produce their own food Plants, algae, cyanobacteria Photosynthesis: the process of turning the sun’s light energy into high-quality chemical energy Sunlight converts carbon dioxide and water into sugars Moving to lower entropy

Photosynthesis produces food Chloroplasts: organelles where photosynthesis occurs Contain chlorophyll: a light-absorbing pigment Light reaction: solar energy splits water and creates highenergy molecules that fuel the … Calvin cycle: links carbon atoms from carbon dioxide into sugar (glucose) 6 CO 2 + 6 H 2 O + sun’s energy C 6 H 12 O 6 (sugar) + 6 O 2
Light and pigments Animation: Light And Pigments Right-click / Select “Play”

Cellular respiration releases It occurs in all living things (plants, animals, etc. ) energy Organisms use chemical energy created by photosynthesis Oxygen breaks the high-energy chemical glucose bonds The energy is used to make other chemical bonds or tasks Heterotrophs: organisms that gain energy by feeding on others Animals, fungi, microbes The energy is used for cellular tasks C 6 H 12 O 6 (sugar) + 6 O 2 6 CO 2 + 6 H 2 O + energy
- Slides: 9