ENERGY Chapter 10 ENERGY Ability to do work

ENERGY Chapter 10

ENERGY • Ability to do work or produce heat. • Potential energy – energy due to position or composition • water in a dam • attractive and repulsive forces • Kinetic energy – energy due to motion of the object, depends on mass and velocity of object KE = ½ mv 2 • Law of conservation of energy – energy can be converted from one form to another but cannot be created or destroyed. • Work – force acting over a distance • Frictional heating – transfer of energy to another surface as heat • Total energy is constant, heat and work depend on a pathway (how it occurs, specific conditions)

TEMPERATURE AND HEAT • Temperature – measure of the random motions of the components of a substance • Heat – flow of energy due to temperature difference. • Thermal energy – random motion of components in an object. • If you mix two different temperature liquids, they will transfer energy until they are both at the same energy level (temperature). • Tfinal = (Thot + Tcold)/2 • Class Demo • 250 ml of hot water and 250 ml of cold water • Use thermometer to determine temps. • Place thermodynamic fan into both. This works if masses of water are equal.

EXOTHERMIC AND ENDOTHERMIC • System – part of the universe which we wish to focus on • Surrounding – everything else that isn’t relevant to what we need • Exothermic – energy flows out of the system in chemical reaction • Endothermic – energy flows into a system during a chemical reaction • Energy gained by surroundings must equal energy lost by system. • Change in energy is from potential energy transformed and released.
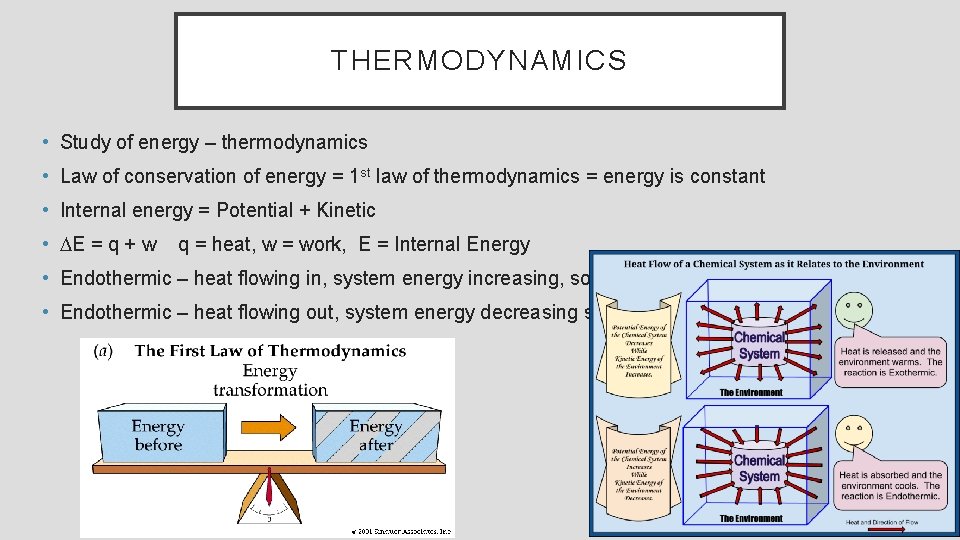
THERMODYNAMICS • Study of energy – thermodynamics • Law of conservation of energy = 1 st law of thermodynamics = energy is constant • Internal energy = Potential + Kinetic • DE = q + w q = heat, w = work, E = Internal Energy • Endothermic – heat flowing in, system energy increasing, so +q • Endothermic – heat flowing out, system energy decreasing so -q

MEASURING ENERGY CHANGES • Units of energy – calorie and joule • 1 calorie = amount of energy (heat) needed to raise the temperature of 1 gram of water by 1 o. C. • Joule – Metric unit 1 calorie = 4. 184 Joule 1 cal = 4. 184 J • Determine number of calories in 28. 4 J? • x cal = 1 cal x 28. 4 J = 6. 78 cal • 4. 184 J • How much energy (in J) would be needed to raise 1 gram of water 5 degrees? • ? J = 4. 184 J x 1 gram x 5 o. C = 20. 92 J 1 g 1 o. C

PICK A CARD, ANY CARD… X ? P

CLASS PROBLEMS • How much energy (in J) is needed to heat 7. 4 g of water from 29 o. C to 46 o. C? • ? J = 4. 184 J x 7. 4 g x (46 -29) o. C = 526 J 1 g 1 o. C • How much energy (in J) is needed heat 62 g of water from 35 o. C to 81 o. C? • ? J = 4. 184 J x 62 g x (81 -35) o. C = 11933 J 1 g 1 o. C • How much energy (in J) is needed heat 450 g of water from 5. 4 o. C to 98. 6 o. C? • ? J = 4. 184 J x 450 g x (98. 6 -5. 4) o. C = 175477 J 1 g 1 o. C

PICK A CARD, ANY CARD… X ? P

MELTING BLOCKS • Break into groups. • Is there a temperature difference when you touch the blocks? Note any observations. • Place 1 rubber ring on each block. • Add a few ice cubes to each block inside the ring. • Note your observations. • Feel the blocks again, note any observations.
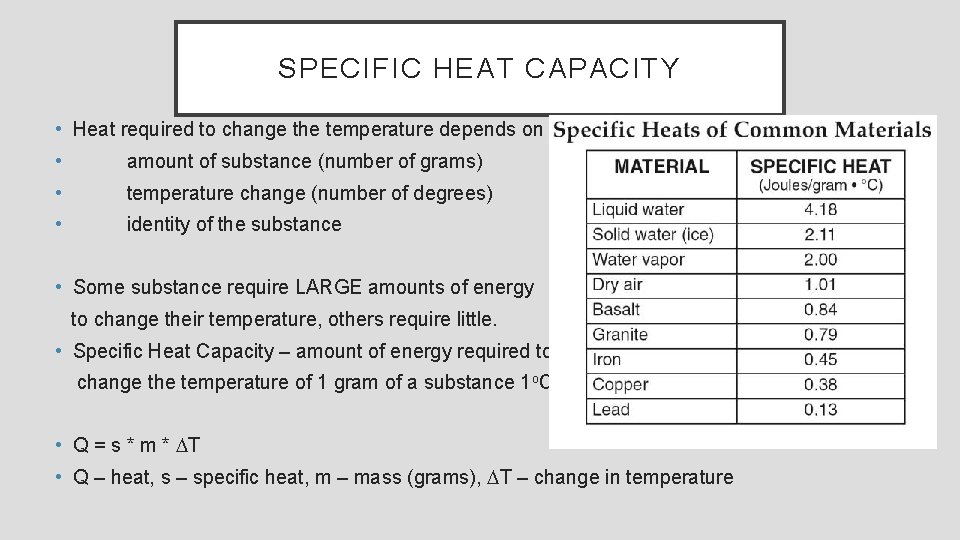
SPECIFIC HEAT CAPACITY • Heat required to change the temperature depends on • amount of substance (number of grams) • temperature change (number of degrees) • identity of the substance • Some substance require LARGE amounts of energy to change their temperature, others require little. • Specific Heat Capacity – amount of energy required to change the temperature of 1 gram of a substance 1 o. C. • Q = s * m * DT • Q – heat, s – specific heat, m – mass (grams), DT – change in temperature

USING WHAT WE KNOW • How much energy (in J) is needed to heat a piece of iron weighing 1. 3 g from 25 o. C to 46 o. C? • So what do we know… Q = s * m * DT • s = specific heat of iron. 45 J/g * o. C • m = 1. 3 grams of iron • DT = 46 -25 = 21 degree temperature increase • • So what do we do…. Q = s * m * DT • Q = ? J =. 45 J x 1. 3 g x 21 o. C = 12. 3 J g o. C • What is this in calories? • ? cal = 1 cal x 12. 3 J = 2. 94 cal 4. 184 J

PICK A CARD, ANY CARD… X ? P

CLASS PROBLEMS Using Q = s * m * DT, find how much energy (in J) is required? • • A 5. 63 gram sample of solid gold is heated from 21 o. C to 32 o. C. • Q =. 129 J x 5. 63 g x (32 -21 o. C) = 8 J g o. C • A 1. 6 gram sample of iron is heated fro 23 o. C to 41 o. C. • Q =. 449 J x 1. 6 g x (41 -23 o. C) = 12. 9 J g o. C • A 42. 7 gram block of aluminum is heated by 15. 2 o. C. • Q =. 987 J x 42. 7 g x (15. 2 o. C) = 640 J g o. C

ENTHALPY • Enthalpy (H) – for reactions occurring under constant pressure, the change in enthalpy (DHp) equals the energy in the form of heat. • When 1 mole of methane, CH 4, is burned at constant pressure, 890 k. J of energy is released as heat. Calculate DHp for a process where 5. 8 g of methane is burned under constant pressure. • Heat released DHp = -890 k. J Negative because heat leaves the system • Use molar mass to help calculate amount of heat released for our sample size. • DHp = -890 k. J x 1 mole CH 4 x 5. 8 g CH 4 = -321 k. J 1 mole CH 4 16. 042 g CH 4

MORE ENTHALPY • Given the following equation: • 4 Fe (s) + 3 O 2 (g) = 2 Fe 2 O 3 (s) DHp = -1652 k. J • How much heat is released when 4 moles of Fe is reacted? • -1652 k. J • How much heat is released when 1 mole of Fe 2 O 3 is produced? • DHp = -1652 k. J x 1 mole Fe 2 O 3 = -826 k. J 2 moles Fe 2 O 3 • How much heat is released when 1. 00 g of iron is reacted with excess O 2? • DHp = -1652 k. J x 1 mole Fe x 1. 00 g Fe = -7. 39 k. J 4 moles Fe 55. 845 g Fe • How much heat is released when 10 g of Fe and 2 g O 2 react? • ? moles Fe = 1 mole/55. 845 g x 10 g =. 179 Lim Factor. • DHp = -1652 k. J x 1 mole O 2 x 2 g O 2 = -34. 42 k. J 3 moles O 2 32 g O 2 ? moles O 2 = 1 mole/32 g x 2 g =. 0625

PICK A CARD, ANY CARD… X ? P

HESS’S LAW • Hess’s law states that the total enthalpy of a reaction is the same if it occurs in one step or a series of steps. • Allow us to determine heats of reaction that cannot be measured with calorimeter. • Two Characteristics of Hess’s Law • 1. If the reaction is reversed, the sign of DH is reversed. • 2. Magnitude of DH is directly proportional to quantities of reactants and products. • Xe + 2 F 2 = Xe. F 4 DH = -251 k. J • 2 Xe + 4 F 2 = 2 Xe. F 4 DH = -502 k. J Xe. F 4 = Xe + 2 F 2 DH = 251 k. J

USING HESS’S LAW • Given the following information: • C (s) + O 2 (g) = CO 2 (g) DH = -393 k. J • 2 CO (g) + O 2 (g) = 2 CO 2 (g) DH = -566 k. J • Determine DH for 2 C (s) + O 2 (g) = CO (g) • Double the first reaction (red) • Reverse the second reaction. • Cross off things on both sided of the equation. • Add the DH • 2 C (s) + 2 O 2 (g) = 2 CO 2 (g) DH = -786 k. J • DH = 566 k. J • 2 CO 2 (g) = 2 CO (g) + O 2 (g) 2 C (s) + O 2 (g) = 2 CO (g) DH = -220 k. J

PICK A CARD, ANY CARD… X ? P
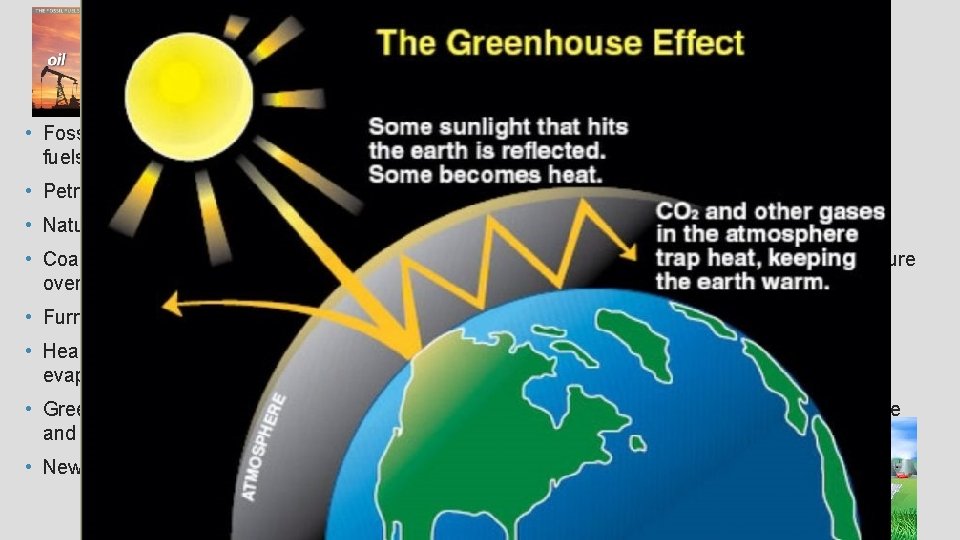
ENERGY IN OUR WORLD • Fossil Fuels – plant or decay products that have been converted over millions of years to usable fuels such as woody plants, coal, petroleum and natural gas. • Petroleum – thick, dark liquid make of carbon and hydrogens (hydrocarbons) • Natural Gas – consists mostly of methane, but consists of ethane, propane, and butane. • Coal – formed from the remains of plants that were buried and subjected to high heat and pressure over time. • Furnishes 20% of US energy but hard, dangerous to mine and produces air pollution/acid rain. • Heat radiation from the sun (infrared radiation) absorbed by plants for photosynthesis, used to evaporate water in the ocean, warms the earth’s surface, 30% reflected back. • Greenhouse effect – where heat trapped by H 2 O and CO 2 is not allowed to pass back into space and continues to warm the earth. • New energy sources – solar, biomass, hydroelectric, wave
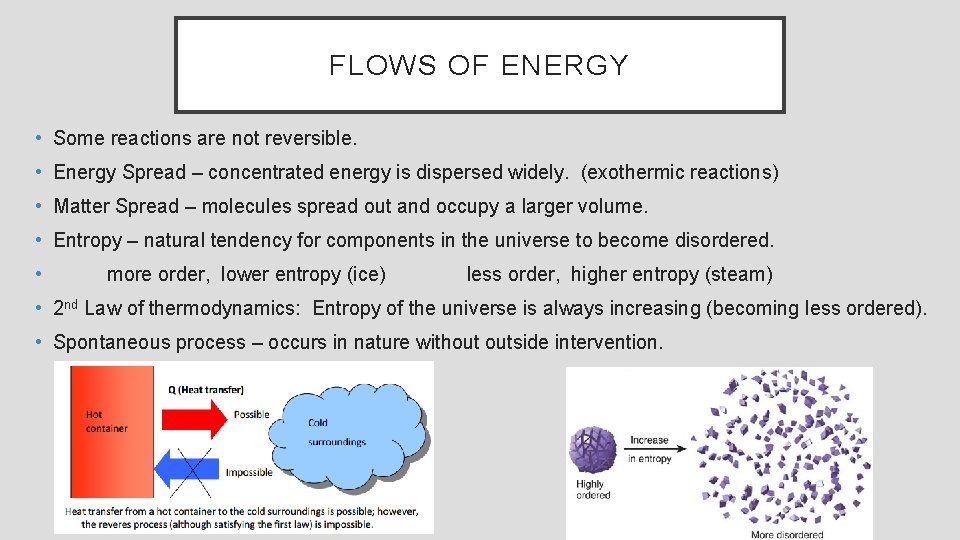
FLOWS OF ENERGY • Some reactions are not reversible. • Energy Spread – concentrated energy is dispersed widely. (exothermic reactions) • Matter Spread – molecules spread out and occupy a larger volume. • Entropy – natural tendency for components in the universe to become disordered. • more order, lower entropy (ice) less order, higher entropy (steam) • 2 nd Law of thermodynamics: Entropy of the universe is always increasing (becoming less ordered). • Spontaneous process – occurs in nature without outside intervention.

WORKS CITED • https: //encryptedtbn 0. gstatic. com/images? q=tbn: ANd 9 Gc. Sntgvo 4 by. IGAuz 7 TUlet 8 Tr. Yrl. Vh. V 6 he. Zro. FNx 6 Zs. P 6 Dd 9 d. EY 8 QQ • https: //www. arborsci. com/reversible-thermoelectric-demonstrator. html? ff=4&fp=619 • http: //www. nonprofitpro. com/thumb/? src=/wp-content/uploads/sites/8/2013/12/FS 1213 -match 50036134. jpg&h=400 • https: //emergencywater 101. com/wp-content/uploads/2017/07/Boiling-water. jpg • https: //1. bp. blogspot. com/W_h. Ufy 4 ki. LU/V_sg. FCFRly. I/AAAACn. M/__u. ECS 8 APVIW 7 i. Fnjy 4 l. QLPl. Sg. Be. KWNcg. CLc. B/s 1600/first_law_ of_thermodynamics. jpg • http: //4. bp. blogspot. com/o. PZmpi. Rnvv 8/UWH 3 HKb 67 RI/AAAAAz. Y/k. Nmw. MOKd. ICY/s 1600/Chemical+Heat+Flow. png • https: //www. arborsci. com/ice-melting-blocks-thermal-conductivity. html? ff=4&fp=117 • http: //regentse. dot 5 hosting. com/Illustrated%20 ESRT/Page%201%20(Prop%20 of%20 H 2 O%20&%20 Sp. %20 He at)/Specific%20 Heats. jpg • http: //slideplayer. com/8468950/26/images/21/16. 1+Energy+Table+I%3 A+Specific+Heats+of+Common+Substa nces+at+298+K+%2825+%CB%9 AC%29+Substance. +Specific+heat+J%2 F%28 g%CB%9 AC%29. jpg

WORKS CITED 2 • https: //study. com/cimages/videopreview-full/nonrenewable-resources-definitionexamples_133782. jpg • https: //www. thetalkingdemocrat. com/wp-content/uploads/2018/03/Greenhouse-Effect-diagram-1024 x 700850 x 581. jpg • http: //www. powerworldanalysis. com/wp-content/uploads/2018/03/Alternative-energy. png • http: //www. theunrealtimes. com/wp-content/uploads/2013/05/entropy. jpg • http: //www. schoolvideos. in/wp-content/uploads/2016/02/Untitled-20. png
- Slides: 24