Energy Change During Chemical Reactions Endothermic and Exothermic


















- Slides: 36

Energy Change During Chemical Reactions Endothermic and Exothermic Changes

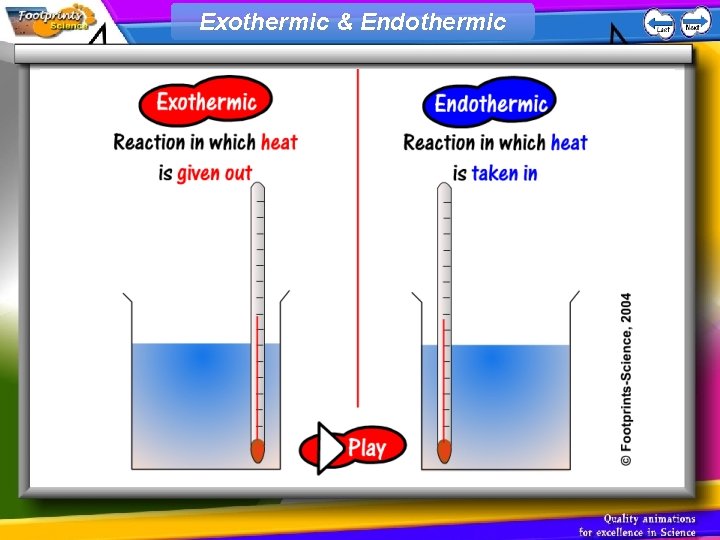
Exothermic & Endothermic

Exothermic reactions 1. An exothermic reaction is one which releases heat energy to the surroundings 2. The temperature of the surroundings increases

Exothermic Reactions Exothermic reactions increase in temperature. • Examples include: – – – Burning reactions including the combustion of fuels. Detonation of explosives. Reaction of acids with metals. Magnesium reacting with acid Thermit reaction © Boardworks Ltd 2003

Activity Say whether these processes are exothermic. 1. 2. 3. 4. 5. Charcoal burning A candle burning. A kettle boiling Ice melting A firework exploding yes no no yes You have to put heat in for boiling and melting. You get heat out from all the other processes © Boardworks Ltd 2003

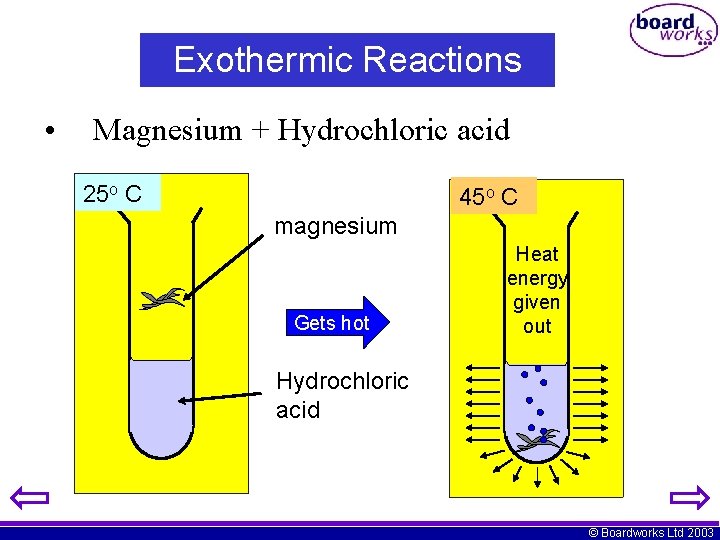
Exothermic Reactions • Magnesium + Hydrochloric acid 25 o C 45 o C magnesium Gets hot Heat energy given out Hydrochloric acid © Boardworks Ltd 2003

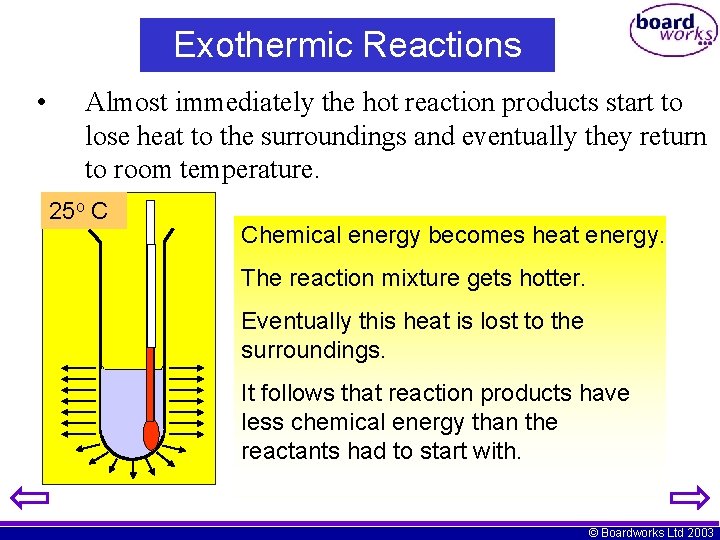
Exothermic Reactions • Almost immediately the hot reaction products start to lose heat to the surroundings and eventually they return to room temperature. 25 o C 45 Chemical energy becomes heat energy. The reaction mixture gets hotter. Eventually this heat is lost to the surroundings. It follows that reaction products have less chemical energy than the reactants had to start with. © Boardworks Ltd 2003

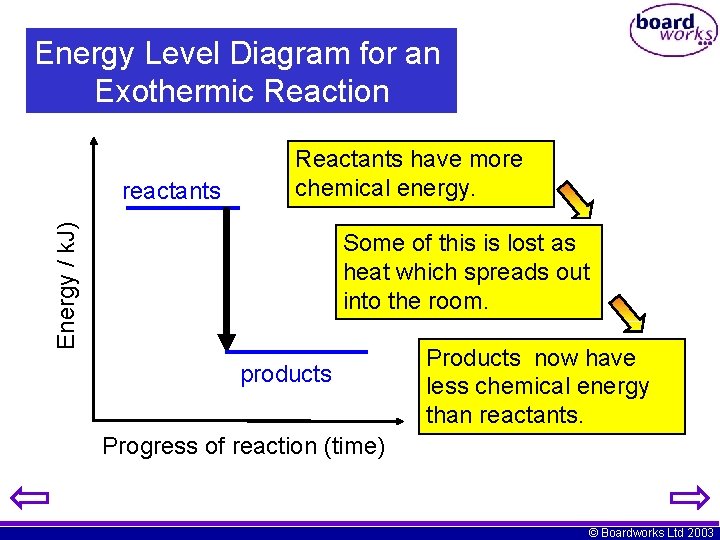
Energy Level Diagram for an Exothermic Reaction Energy / k. J) reactants Reactants have more chemical energy. Some of this is lost as heat which spreads out into the room. products Products now have less chemical energy than reactants. Progress of reaction (time) © Boardworks Ltd 2003

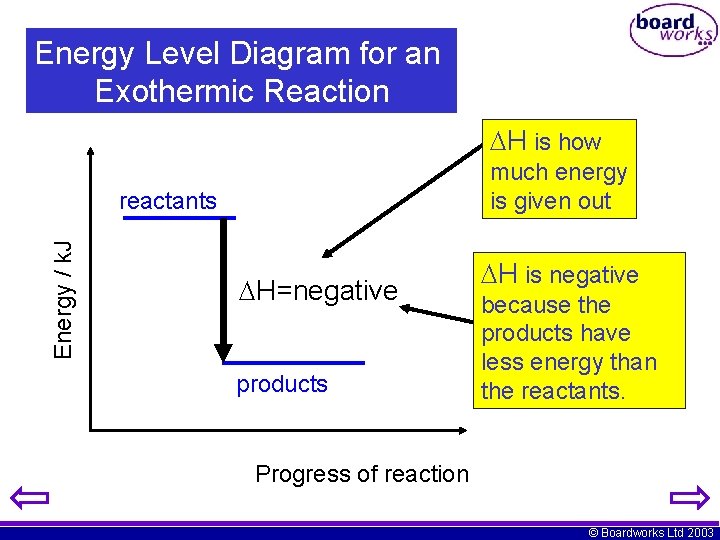
Energy Level Diagram for an Exothermic Reaction 2. H is how much energy is given out Energy / k. J reactants H=negative products H is negative because the products have less energy than the reactants. Progress of reaction © Boardworks Ltd 2003

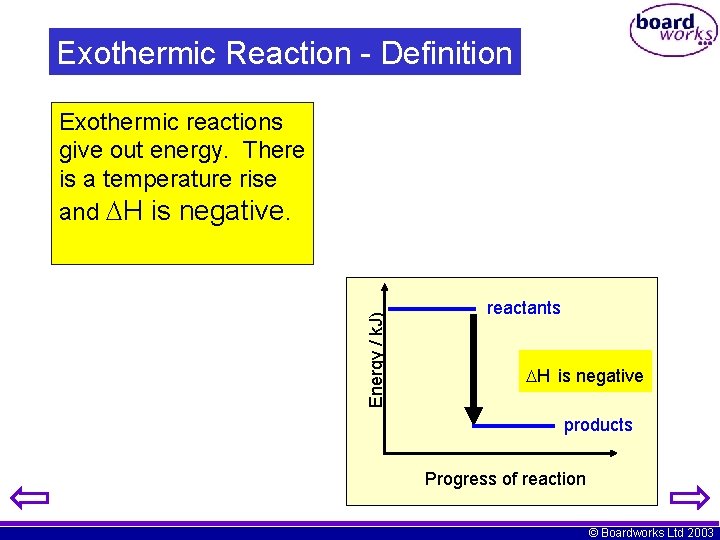
Exothermic Reaction - Definition Energy / k. J) Exothermic reactions give out energy. There is a temperature rise and H is negative. reactants H is negative products Progress of reaction © Boardworks Ltd 2003

Activity © Boardworks Ltd 2003

Endothermic reactions 1. An endothermic reaction is on which takes in heat energy from the surroundings 2. The temperature of the surroundings decreases

Endothermic Reactions Endothermic reactions cause a decrease in temperature. • • • Endothermic chemical reactions are relatively rare. A few reactions that give off gases are highly endothermic - get very cold. Dissolving salts in water is another process that is often endothermic. © Boardworks Ltd 2003

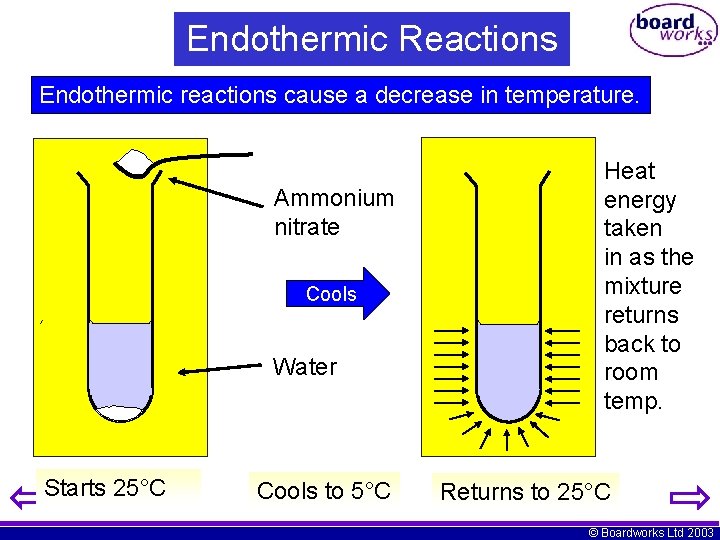
Endothermic Reactions Endothermic reactions cause a decrease in temperature. Ammonium nitrate Cools Water Starts 25°C Cools to 5°C Heat energy taken in as the mixture returns back to room temp. Returns to 25°C © Boardworks Ltd 2003

Endothermic Reactions • The cold reaction products start to gain heat from the surroundings and eventually return to room temperature. o o. CC 25 5 The reactants gain energy. This comes from the substances used in the reaction and the reaction gets cold. Eventually heat is absorbed from the surroundings and the mixture returns to room temperature. Overall the chemicals have gained energy. © Boardworks Ltd 2003

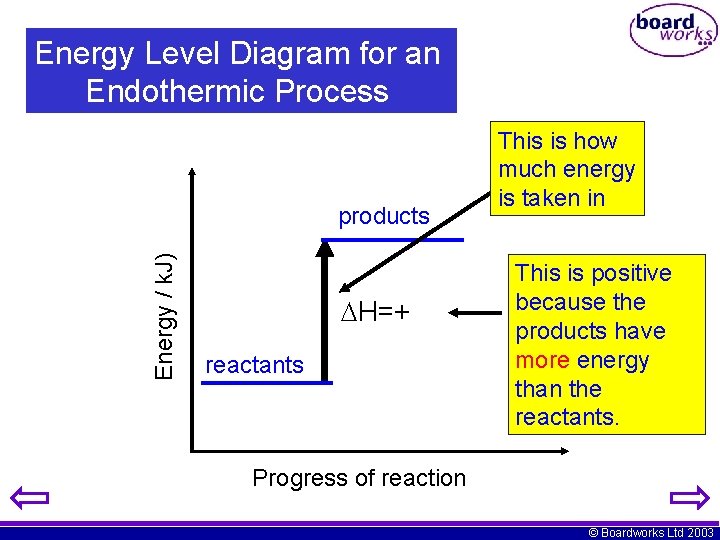
Energy Level Diagram for an Endothermic Process Energy / k. J) products H=+ reactants This is how much energy is taken in This is positive because the products have more energy than the reactants. Progress of reaction © Boardworks Ltd 2003

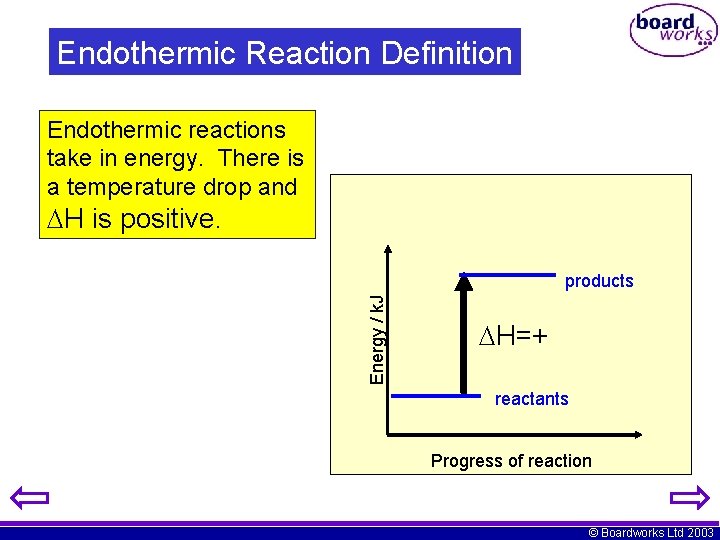
Endothermic Reaction Definition Endothermic reactions take in energy. There is a temperature drop and H is positive. Energy / k. J products H=+ reactants Progress of reaction © Boardworks Ltd 2003

© Boardworks Ltd 2003

Activity Are these endothermic or exothermic? 1. A red glow spread throughout the mixture and the temperature rose. 2. The mixture bubbled vigorously but the temperature dropped 150 C. 3. Hydrazine and hydrogen peroxide react so explosively and powerfully that they are used to power rockets into space. 4. The decaying grass in the compost maker was considerably above the outside temperature. exo endo exo © Boardworks Ltd 2003

Endothermic and exothermic reactions Step 1: Energy must be SUPPLIED to break bonds: Step 2: Energy is RELEASED when new bonds are made: A reaction is EXOTHERMIC if more energy is RELEASED then SUPPLIED. If more energy is SUPPLIED then is RELEASED then the reaction is ENDOTHERMIC

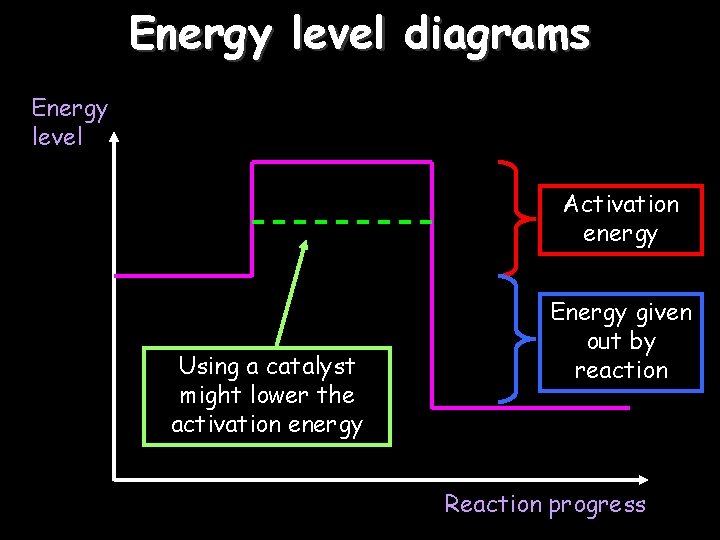
Energy level diagrams Energy level Activation energy Using a catalyst might lower the activation energy Energy given out by reaction Reaction progress

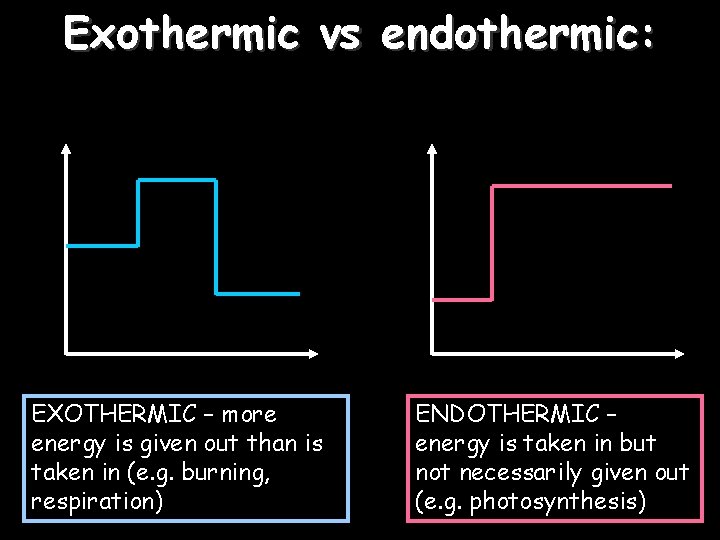
Exothermic vs endothermic: EXOTHERMIC – more energy is given out than is taken in (e. g. burning, respiration) ENDOTHERMIC – energy is taken in but not necessarily given out (e. g. photosynthesis)

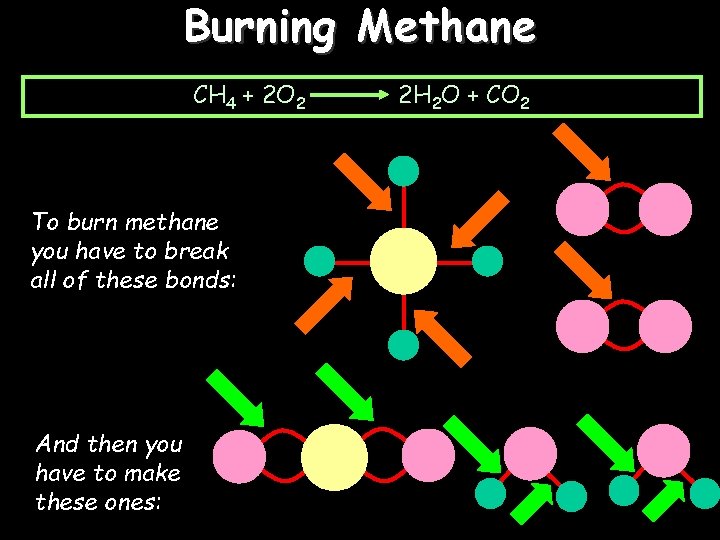
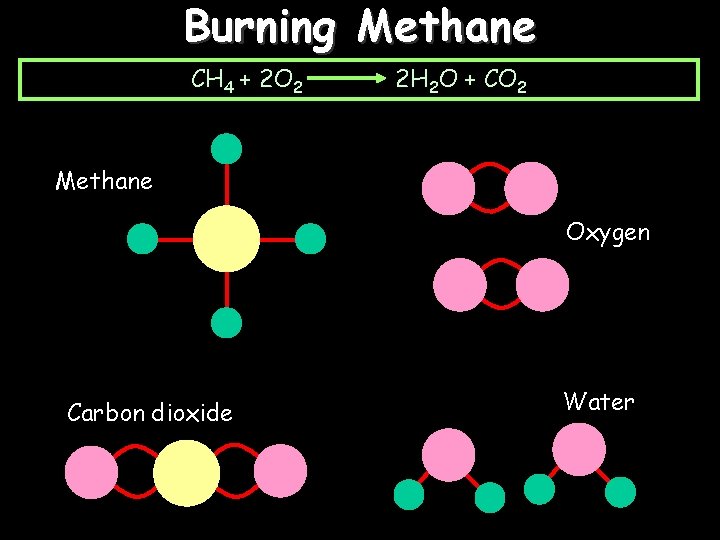
Burning Methane CH 4 + 2 O 2 To burn methane you have to break all of these bonds: And then you have to make these ones: 2 H 2 O + CO 2

Burning Methane CH 4 + 2 O 2 2 H 2 O + CO 2 Methane Oxygen Carbon dioxide Water

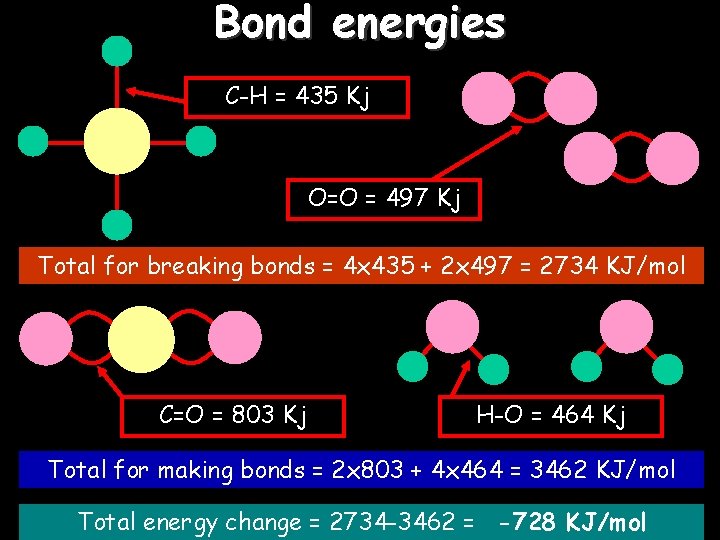
Bond energies C-H = 435 Kj O=O = 497 Kj Total for breaking bonds = 4 x 435 + 2 x 497 = 2734 KJ/mol C=O = 803 Kj H-O = 464 Kj Total for making bonds = 2 x 803 + 4 x 464 = 3462 KJ/mol Total energy change = 2734 -3462 = -728 KJ/mol

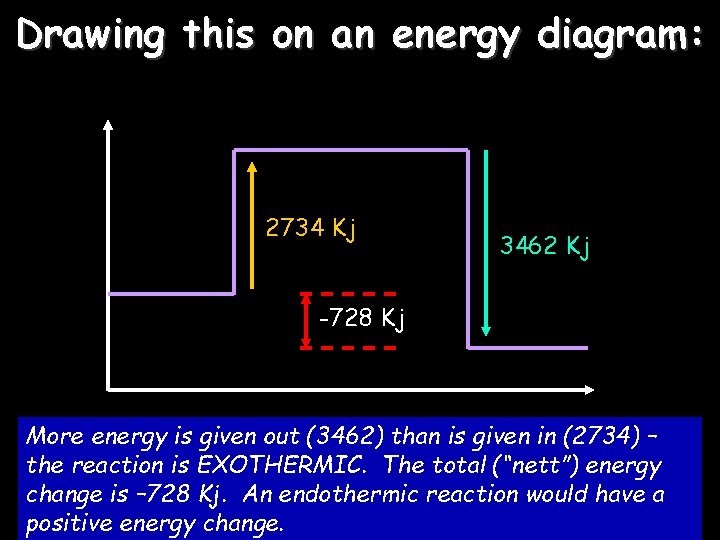
Drawing this on an energy diagram: 2734 Kj 3462 Kj -728 Kj More energy is given out (3462) than is given in (2734) – the reaction is EXOTHERMIC. The total (“nett”) energy change is – 728 Kj. An endothermic reaction would have a positive energy change.

Bond energy values C-H = 435 KJ/mol O-H = 464 KJ/mol O=O = 497 KJ/mol C=O = 803 KJ/mol C-O = 360 KJ/mol C-C = 346 KJ/mol

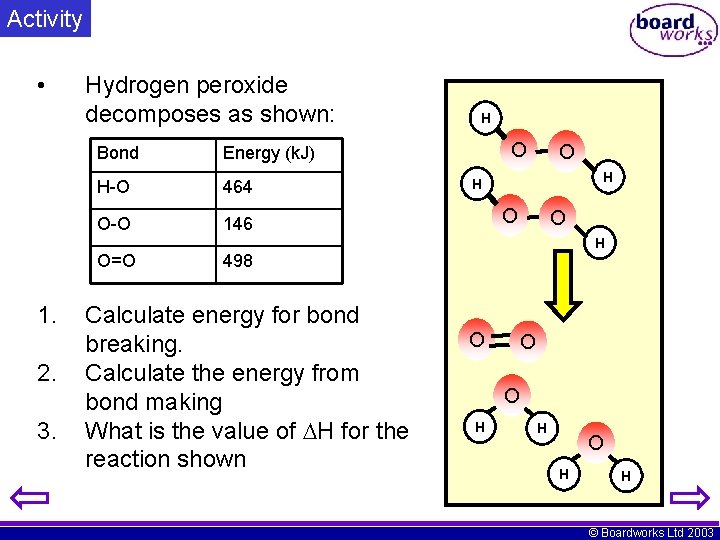
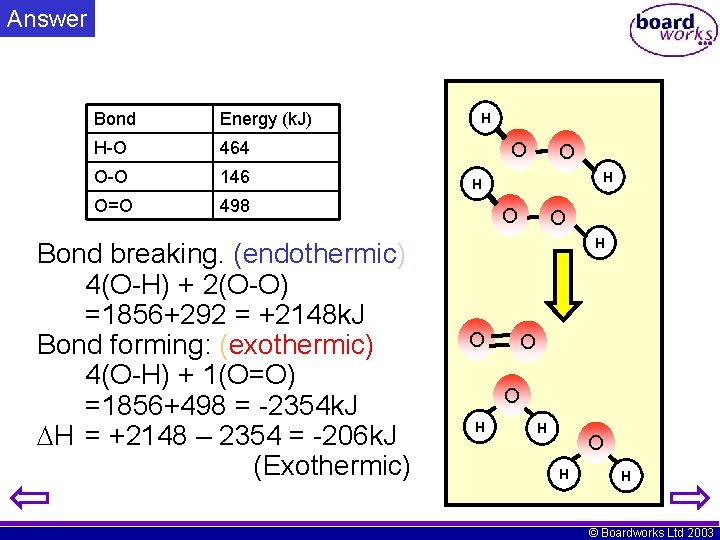
Activity • Hydrogen peroxide decomposes as shown: Bond Energy (k. J) H-O 464 O-O 146 H O O H O=O 1. 2. 3. 498 Calculate energy for bond breaking. Calculate the energy from bond making What is the value of H for the reaction shown O O O H H © Boardworks Ltd 2003

Answer Bond Energy (k. J) H-O 464 O-O 146 O=O 498 Bond breaking. (endothermic) 4(O-H) + 2(O-O) =1856+292 = +2148 k. J Bond forming: (exothermic) 4(O-H) + 1(O=O) =1856+498 = -2354 k. J H = +2148 – 2354 = -206 k. J (Exothermic) H O O O H H © Boardworks Ltd 2003

Activity Copy the summary using the words from the box to fill in the gaps: endothermic exothermic lose common positive 1. Exothermic reactions are _____. common endothermic 2. Reactions that get cold are called _____. 3. Bond forming is an _______ exothermic process. positive H. 4. Endothermic reactions have a _____ 5. In exothermic reactions the chemicals ___ lose chemical energy. continued © Boardworks Ltd 2003

Activity Copy the summary using the words from the box to fill in the gaps: more endothermic activation 1. The energy needed to start off a reaction is called the activation ______ energy 2. In endothermic reactions bond breaking more energy than is produced by requires ___ bond forming. endothermic process. 3. Bond breaking is an _____ © Boardworks Ltd 2003

Which of the following is an endothermic process? A. B. C. D. Burning wood Reacting an acid and alkali Reacting magnesium with acid Dissolving ammonium nitrate in water © Boardworks Ltd 2003

Which of the following is true for an exothermic process? products Energy / k. J A. The reactants lose energy to the surroundings B. The reaction gets cold C. The energy diagram will be as shown D. The reaction will have a H that is positive (+). reactants Progress of reaction © Boardworks Ltd 2003

Which of the following is true when chemical bonds are broken? A. The process is exothermic B. Energy is given out C. A physical change is occurring D. The reaction will have a H that is positive(+). © Boardworks Ltd 2003

Which of the following is true for an exothermic reaction? A. Bond breaking involves a bigger energy change than bond making B. Bond making involves a bigger energy change than bond breaking C. Bond making involves the same energy change as bond breaking © Boardworks Ltd 2003

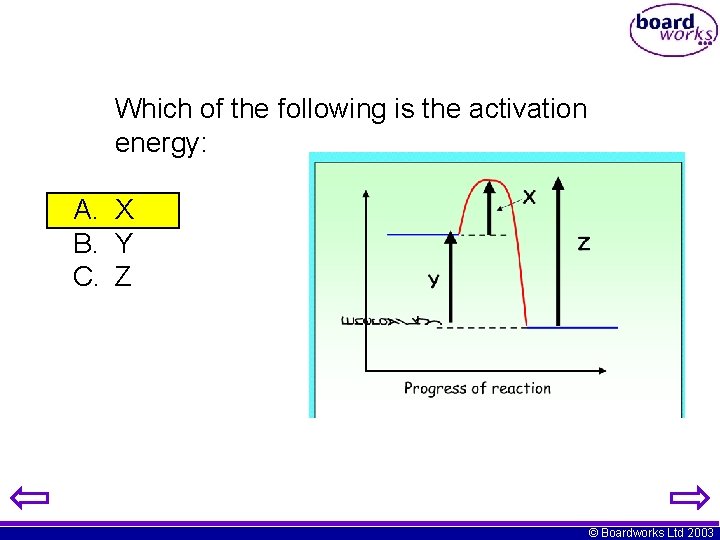
Which of the following is the activation energy: A. X B. Y C. Z © Boardworks Ltd 2003