Energy Capacity to do work or transfer heat






















- Slides: 27

Energy: Capacity to do work or transfer heat Kinetic Energy: Moving energy with mass and velocity: Ex: Wind, heat, electricity, flowing water Potential Energy: Stored energy

The first law of thermodynamics: Energy cannot be created or destroyed. ● ● It can be changed from one form to another.

The second law of thermodynamics: energy quality always decreases. (Increase of entropy!) ● ● ● When energy changes from one form to another, it is always degraded to a more dispersed form. Energy efficiency is a measure of how much useful work is accomplished before it changes to its next form.

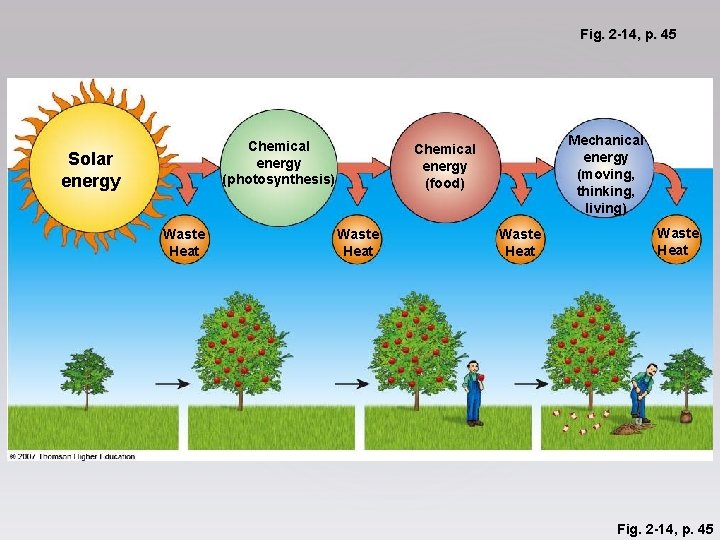
Fig. 2 -14, p. 45 Chemical energy (photosynthesis) Solar energy Waste Heat Mechanical energy (moving, thinking, living) Chemical energy (food) Waste Heat Fig. 2 -14, p. 45

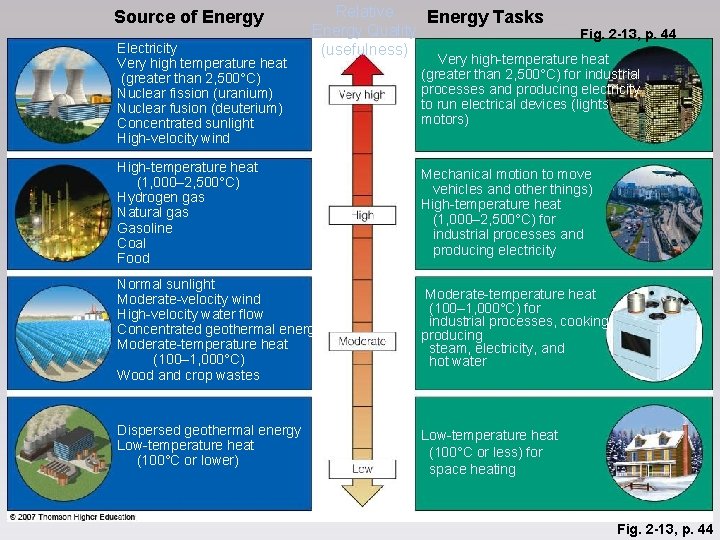
Source of Energy Electricity Very high temperature heat (greater than 2, 500°C) Nuclear fission (uranium) Nuclear fusion (deuterium) Concentrated sunlight High-velocity wind Relative Energy Tasks Energy Quality (usefulness) Fig. 2 -13, p. 44 Very high-temperature heat (greater than 2, 500°C) for industrial processes and producing electricity to run electrical devices (lights, motors) High-temperature heat (1, 000– 2, 500°C) Hydrogen gas Natural gas Gasoline Coal Food Mechanical motion to move vehicles and other things) High-temperature heat (1, 000– 2, 500°C) for industrial processes and producing electricity Normal sunlight Moderate-velocity wind High-velocity water flow Concentrated geothermal energy Moderate-temperature heat (100– 1, 000°C) Wood and crop wastes Moderate-temperature heat (100– 1, 000°C) for industrial processes, cooking, producing steam, electricity, and hot water Dispersed geothermal energy Low-temperature heat (100°C or lower) Low-temperature heat (100°C or less) for space heating Fig. 2 -13, p. 44



Law of Conservation of Mass • For any closed system, the mass must remain constant over time. • How is this important in the environment? We’re not “consuming” resources, we’re just borrowing them and transforming them. POLLUTION! We will always be faced with pollution of some sort. What kind? And how do we reduce it? These are the questions we ask in environmental science.

ENERGY FLOW, TROPHIC PYRAMIDS, PRIMARY PRODUCTIVITY 300 trout are needed to support one man for a year. The trout, in turn, must consume 90, 000 frogs, that must consume 27 million grasshoppers that live off of 1, 000 tons of grass. -- G. Tyler Miller, Jr. , American Chemist (1971)

Predator

Omnivore

Consumer/Heterotroph

Decomposer

Producers/Autotrophs

Primary Consumer

Detritivore

Carnivore

Secondary Consumer

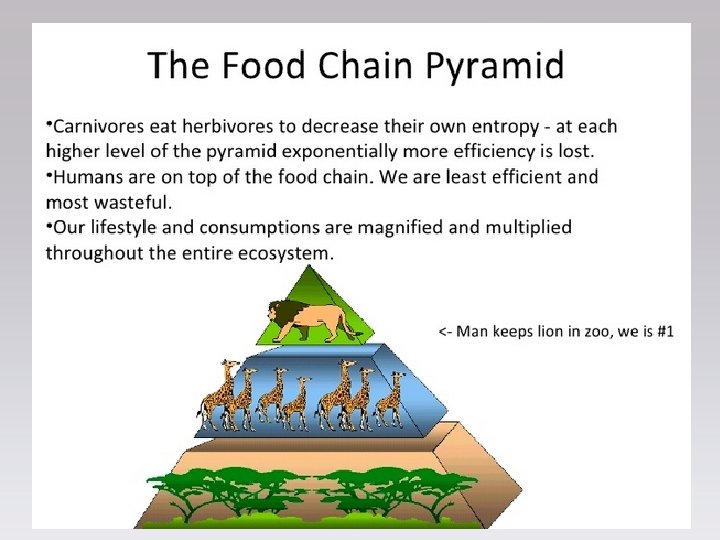
Ecological Pyramids Graphical models of the quantitative differences between trophic levels of a single ecosystem Usually Pyramids become narrower toward the top 1. Energy Pyramid (also sometimes called Trophic Pyramid) 2. Biomass Pyramid 3. Pyramid of Numbers

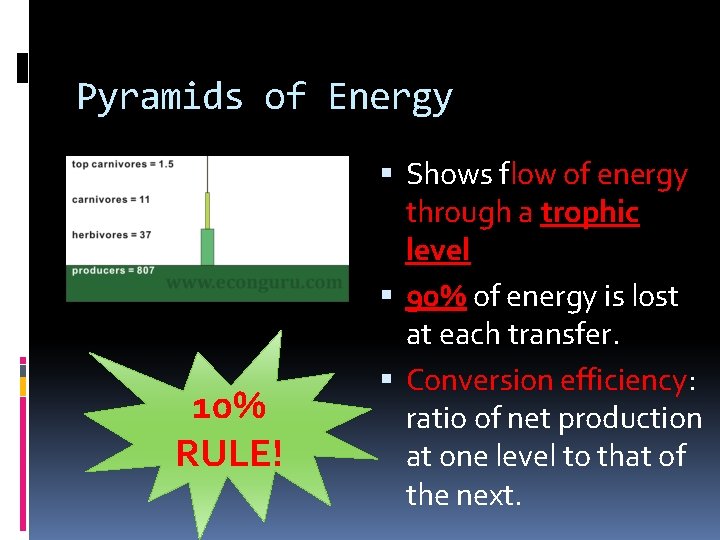
Pyramids of Energy 10% RULE! Shows flow of energy through a trophic level 90% of energy is lost at each transfer. Conversion efficiency: ratio of net production at one level to that of the next.

Pyramids of Energy: The remaining 90% is lost as: 1) feces 2) urine 3) used in respiration and lost as heat Loss?

Pyramids of Biomass = total mass of organisms The "missing" mass, that not eaten, becomes detritus and is decomposed. http: //cr. middlebury. edu/es/altenergylife/biomass%20 logo. gif

Pyramids of Biomass Measured in grams of biomass per square meter (g/m 2)

Pyramid of Numbers Number of individuals at each trophic level. If the size of an organism is small, more can be present. I. e. : ten mice in place of one rabbit. http: //www. biology. com/kimball. html? maincontent=F/Food. Chains. html

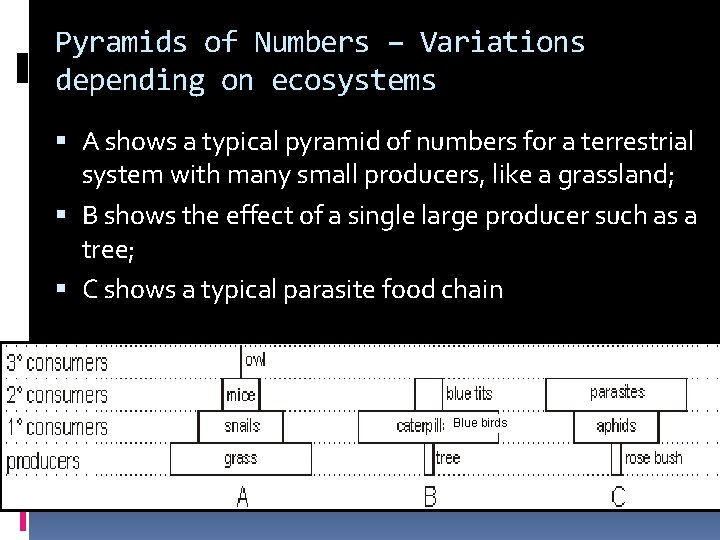
Pyramids of Numbers – Variations depending on ecosystems A shows a typical pyramid of numbers for a terrestrial system with many small producers, like a grassland; B shows the effect of a single large producer such as a tree; C shows a typical parasite food chain Blue birds

Inverted Pyramids Occurs in BOTH pyramids of numbers and biomass Occurs more often with numbers Can also result from seasonal fluctuations, weather and natural disasters

How does the Pyramid Structure Affect Ecosystems? In a typical pyramid, higher trophic levels have less total biomass than those below less energy is available lower level sets the limit at the next level Pyramid of energy can never be inverted (no additional energy is entering the system).