Energy and Metabolism I Energy Basics I Energy

Energy and Metabolism

I. Energy Basics

I. Energy Basics A. Forms of Energy - energy is the capacity to cause change

I. Energy Basics A. Forms of Energy - energy is the capacity to cause change 1. kinetic = energy of a moving body - thermal = energy of moving atoms - light = energy of moving photons - electricity = energy of moving charge 2. potential = energy in matter due to location/structure - potential kinetic (position) - potential electric (like in a battery or across a membrane) - chemical (energy that can be release by the breaking of chemical bonds)

I. Energy Basics A. Forms of Energy B. Laws of Thermodynamics

I. Energy Basics A. Forms of Energy B. Laws of Thermodynamics 1. Conservation of Energy: Energy/matter can not be created or destroyed, but it can be transferred and transformed.

I. Energy Basics A. Forms of Energy B. Laws of Thermodynamics 1. Conservation of Energy: Energy/matter can not be created or destroyed, but it can be transferred and transformed. 2. Law of Entropy: Every energy transformation increases the entropy of the universe.

Transformations 4 H 2 He + E = light E Light E Thermal E of skin, water Thermal E of skin Thermal E of water Potential on board Kinetic of diver Chemical E thermal body heat Chemical E kinetic E of muscles Kinetic E of muscles Potential E on board
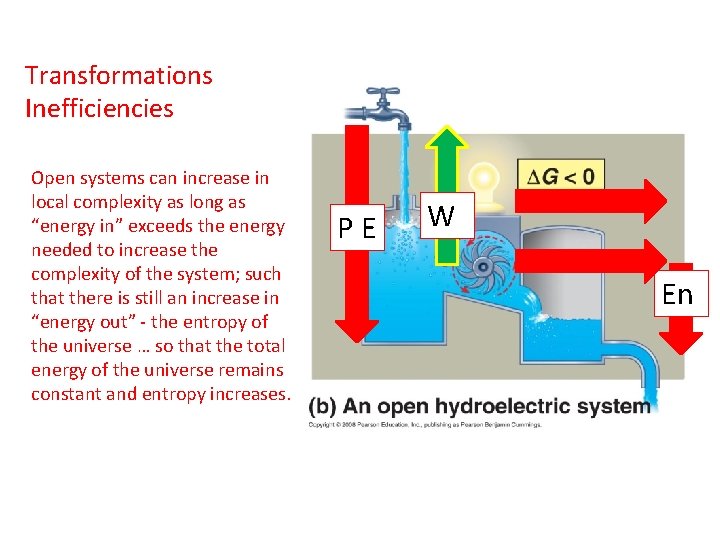
Transformations Inefficiencies Open systems can increase in local complexity as long as “energy in” exceeds the energy needed to increase the complexity of the system; such that there is still an increase in “energy out” - the entropy of the universe … so that the total energy of the universe remains constant and entropy increases. PE W En
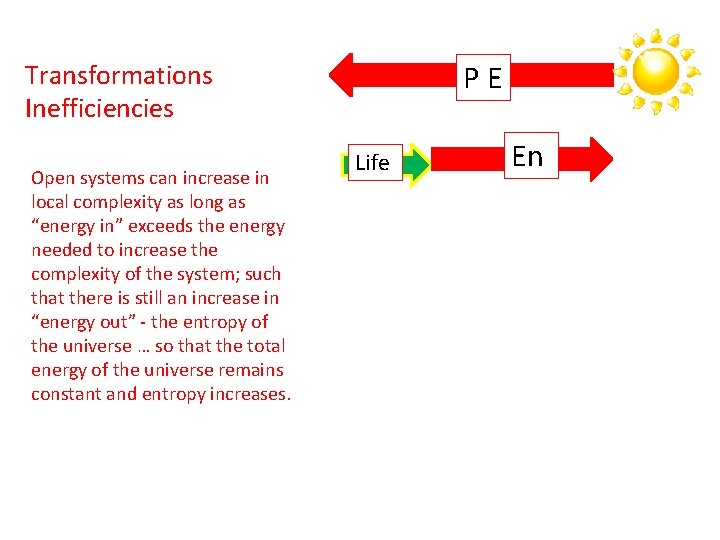
PE Transformations Inefficiencies Open systems can increase in local complexity as long as “energy in” exceeds the energy needed to increase the complexity of the system; such that there is still an increase in “energy out” - the entropy of the universe … so that the total energy of the universe remains constant and entropy increases. Life En
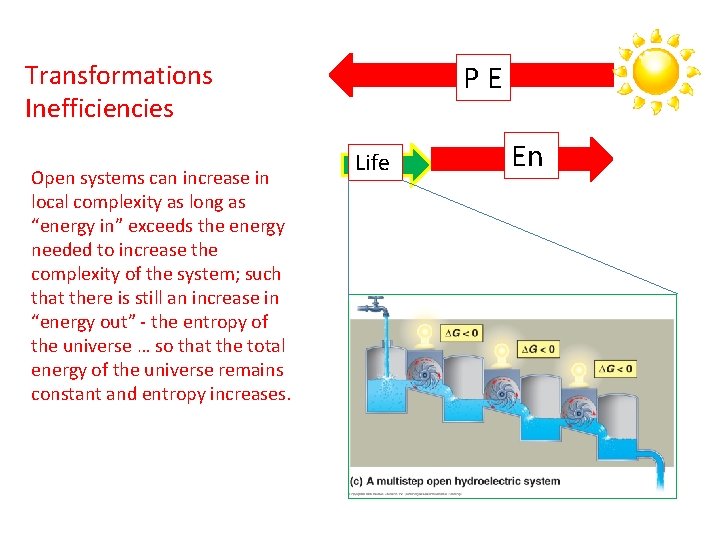
PE Transformations Inefficiencies Open systems can increase in local complexity as long as “energy in” exceeds the energy needed to increase the complexity of the system; such that there is still an increase in “energy out” - the entropy of the universe … so that the total energy of the universe remains constant and entropy increases. Life En

II. Metabolism Overview A. Catabolism and Anabolism: TO build a useful biomolecule (anabolism) or to do mechanical work (kinetic energy), the matter and energy must come from somewhere…. Except for photosynthesis, the source of energy used in living systems is chemical potential energy, harvested by catabolic processes called CELLULAR RESPIRATION.
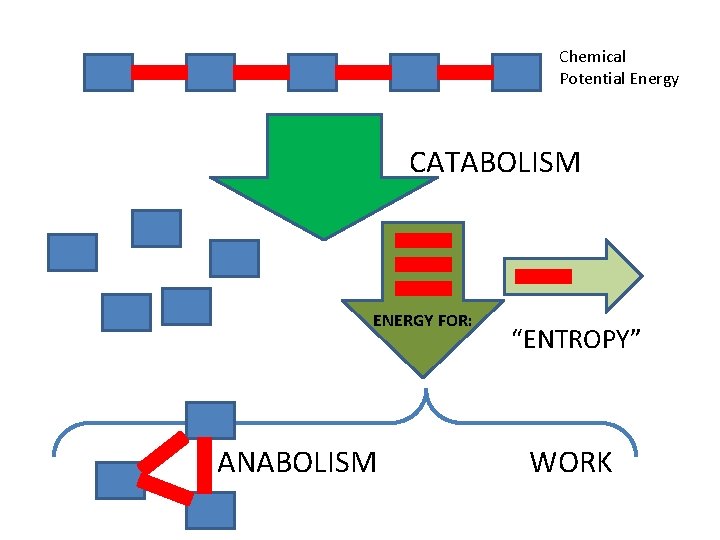
Chemical Potential Energy CATABOLISM ENERGY FOR: ANABOLISM “ENTROPY” WORK
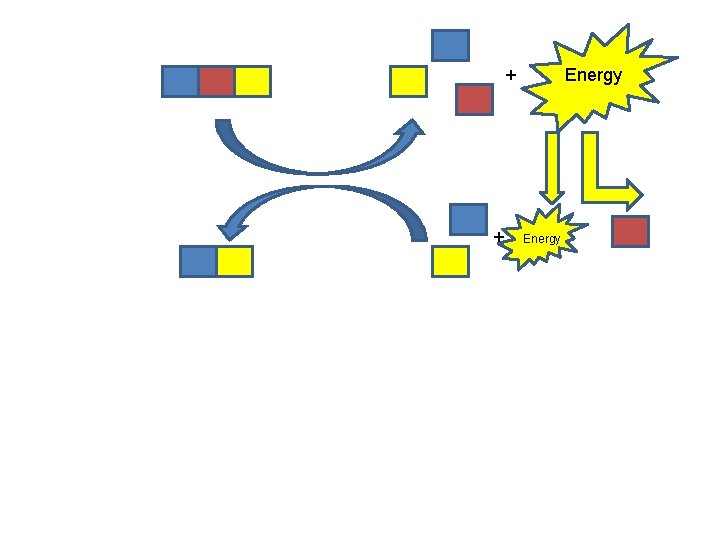
+ + Energy
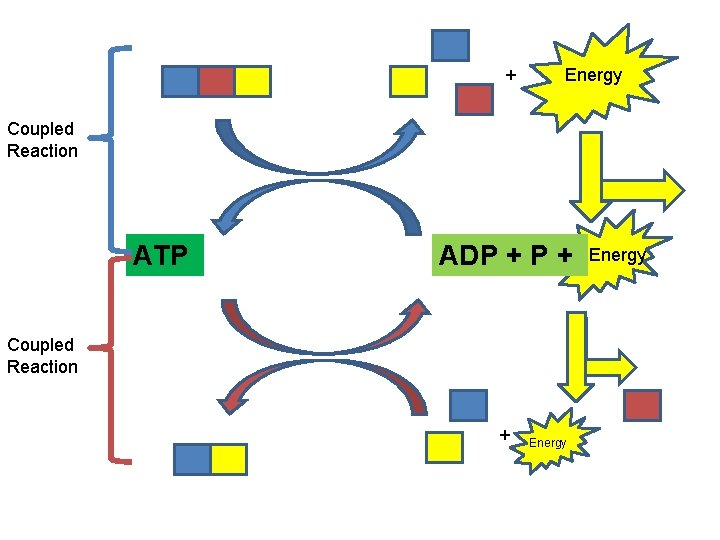
+ Energy Coupled Reaction ATP ADP + Coupled Reaction + Energy
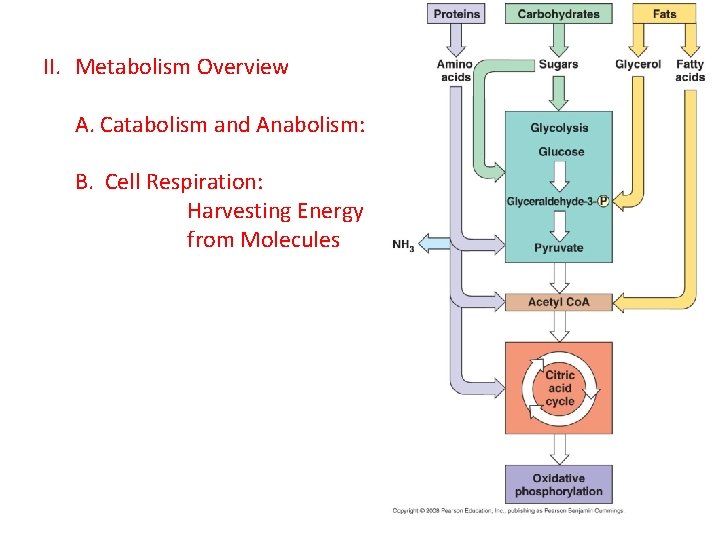
II. Metabolism Overview A. Catabolism and Anabolism: B. Cell Respiration: Harvesting Energy from Molecules
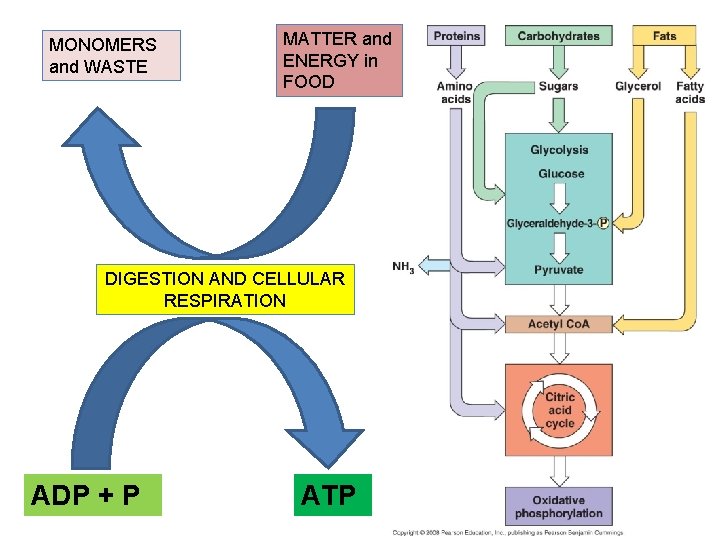
MONOMERS and WASTE MATTER and ENERGY in FOOD DIGESTION AND CELLULAR RESPIRATION ADP + P ATP
B. Cell Respiration: Focus on core process… Glucose metabolism
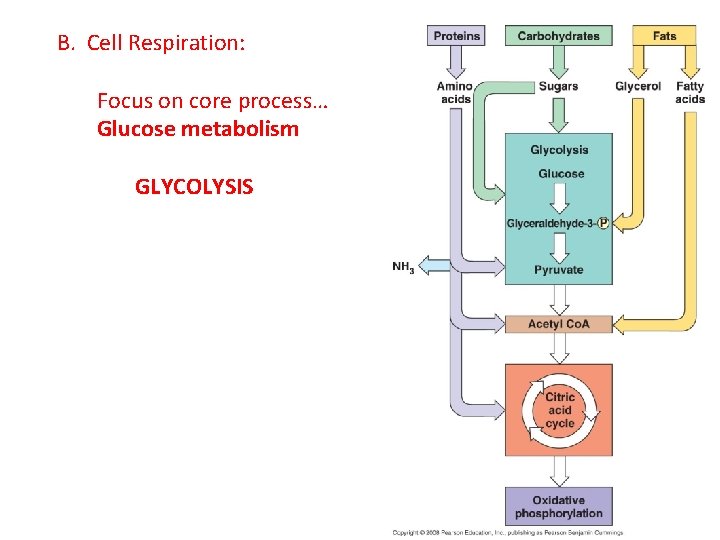
B. Cell Respiration: Focus on core process… Glucose metabolism GLYCOLYSIS
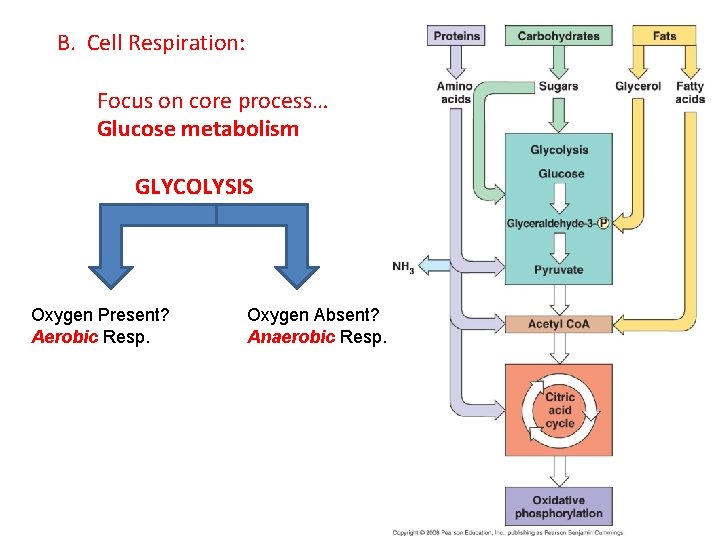
B. Cell Respiration: Focus on core process… Glucose metabolism GLYCOLYSIS Oxygen Present? Aerobic Resp. Oxygen Absent? Anaerobic Resp.
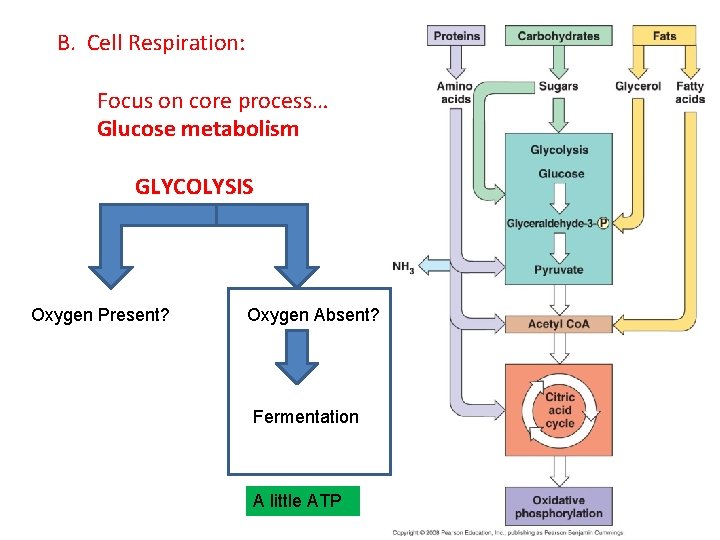
B. Cell Respiration: Focus on core process… Glucose metabolism GLYCOLYSIS Oxygen Present? Oxygen Absent? Fermentation A little ATP
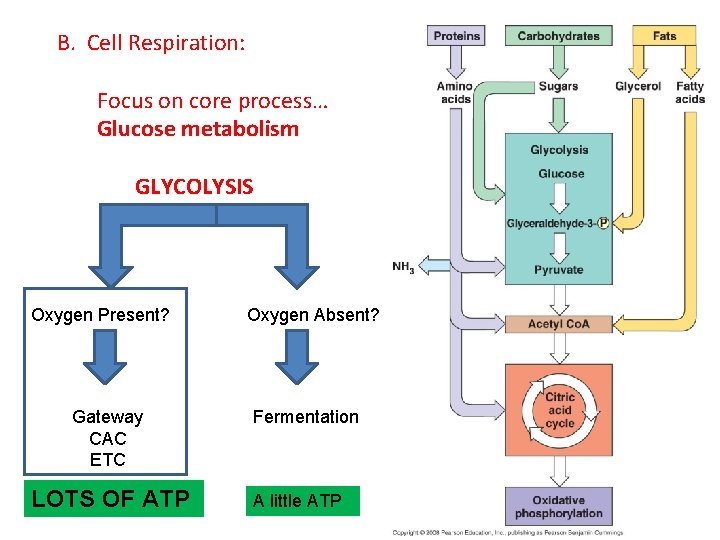
B. Cell Respiration: Focus on core process… Glucose metabolism GLYCOLYSIS Oxygen Present? Gateway CAC ETC LOTS OF ATP Oxygen Absent? Fermentation A little ATP

B. Respiration: 1. Glycolysis: - Occurs in presence OR absence of oxygen gas. - All cells do this! (very primitive pathway) - Occurs in the cytoplasm of all cells
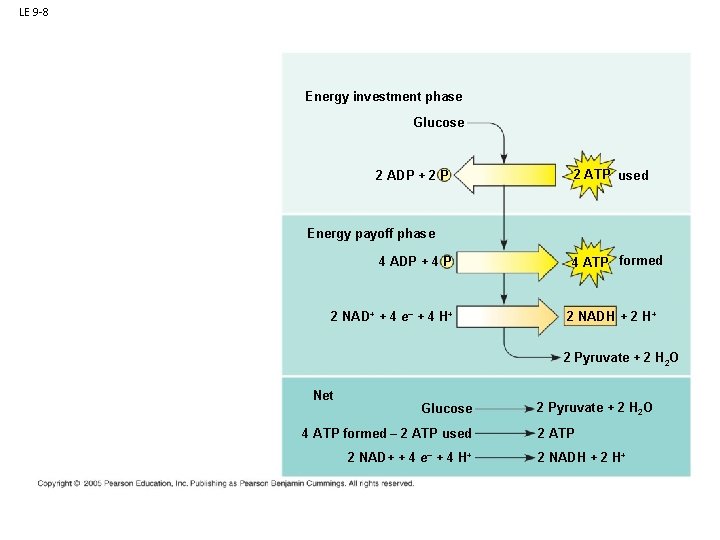
LE 9 -8 Energy investment phase Glucose 2 ADP + 2 P 2 ATP used Glycolysis Energy payoff phase ATP ATP 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
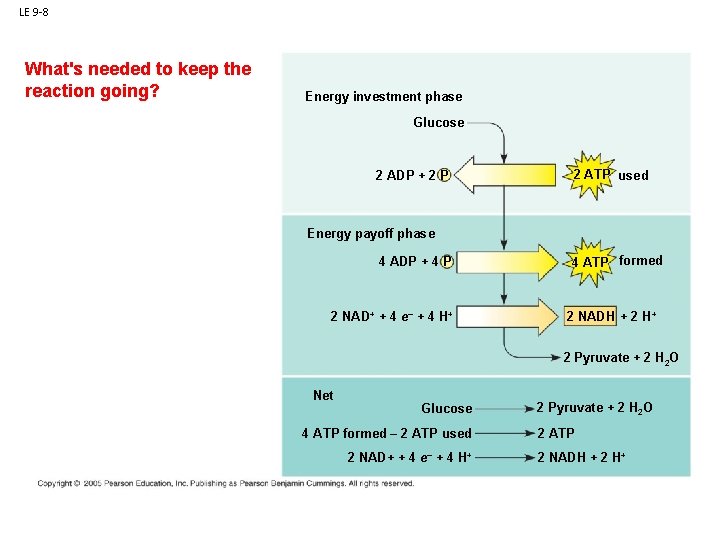
LE 9 -8 What's needed to keep the reaction going? Energy investment phase Glucose 2 ADP + 2 P 2 ATP used Glycolysis Energy payoff phase ATP ATP 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
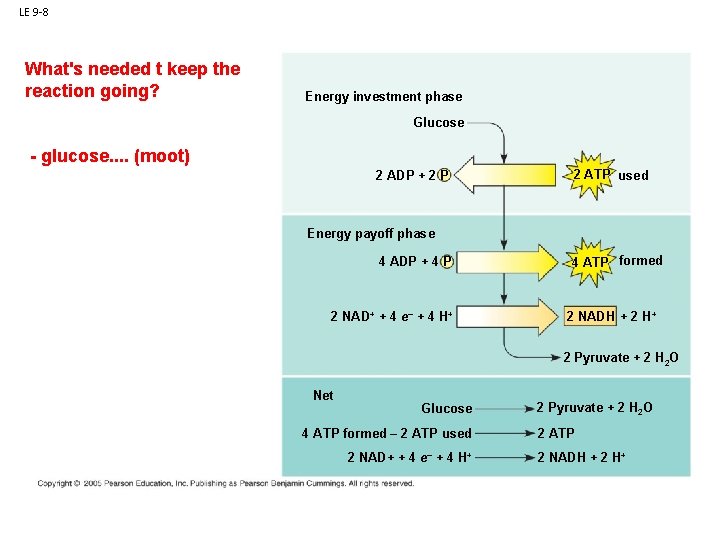
LE 9 -8 What's needed t keep the reaction going? Energy investment phase Glucose - glucose. . (moot) 2 ADP + 2 P 2 ATP used Glycolysis Energy payoff phase ATP ATP 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
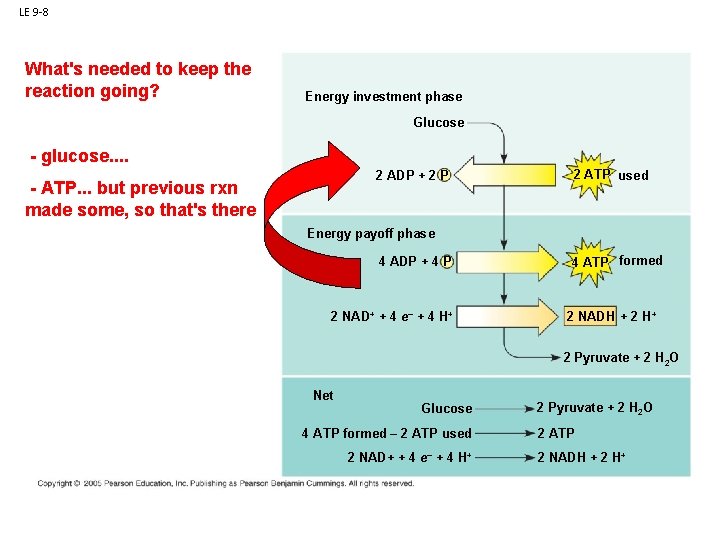
LE 9 -8 What's needed to keep the reaction going? Energy investment phase Glucose - glucose. . 2 ADP + 2 P Glycolysisbut previous rxn - ATP. . . made some, so that's there 2 ATP used Energy payoff phase ATP ATP 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
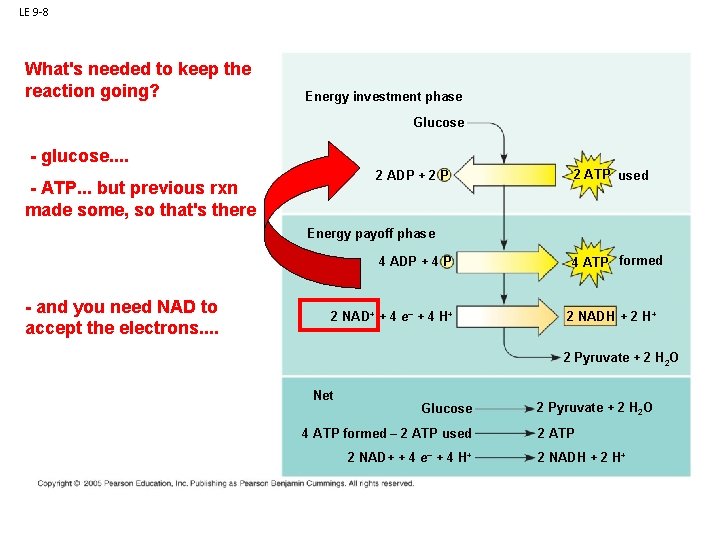
LE 9 -8 What's needed to keep the reaction going? Energy investment phase Glucose - glucose. . 2 ADP + 2 P Glycolysisbut previous rxn - ATP. . . made some, so that's there 2 ATP used Energy payoff phase ATP ATP - and you need NAD to accept the electrons. . 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
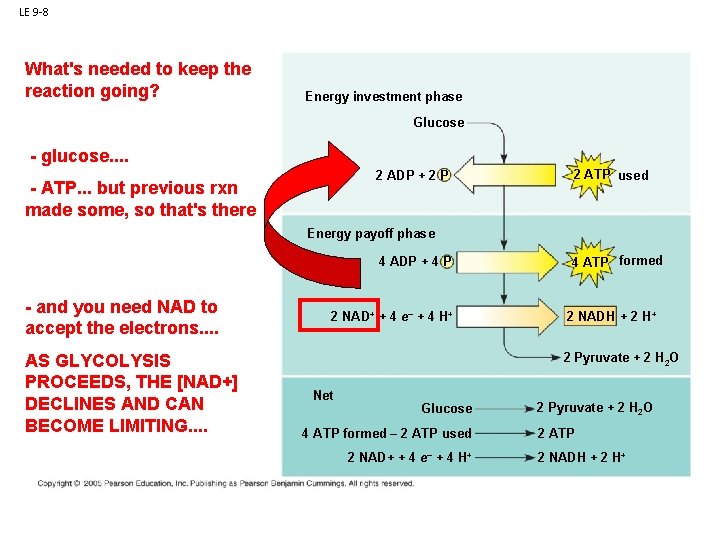
LE 9 -8 What's needed to keep the reaction going? Energy investment phase Glucose - glucose. . 2 ADP + 2 P Glycolysisbut previous rxn - ATP. . . made some, so that's there 2 ATP used Energy payoff phase ATP ATP - and you need NAD to accept the electrons. . AS GLYCOLYSIS PROCEEDS, THE [NAD+] DECLINES AND CAN BECOME LIMITING. . 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
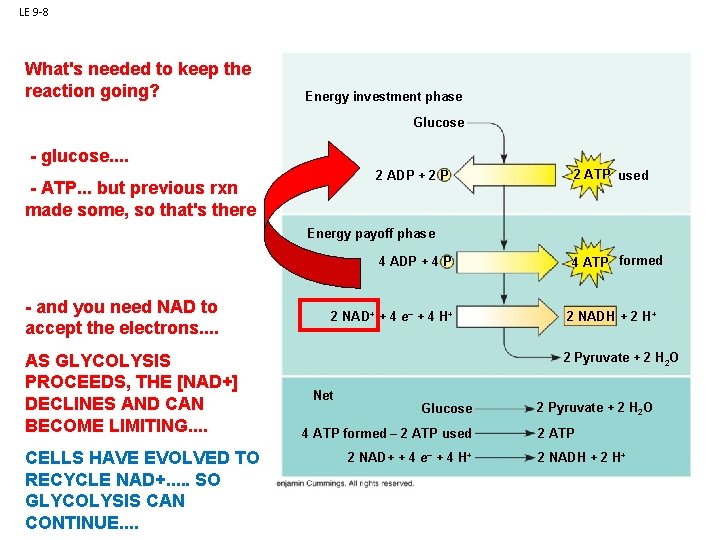
LE 9 -8 What's needed to keep the reaction going? Energy investment phase Glucose - glucose. . 2 ADP + 2 P Glycolysisbut previous rxn - ATP. . . made some, so that's there 2 ATP used Energy payoff phase ATP ATP - and you need NAD to accept the electrons. . AS GLYCOLYSIS PROCEEDS, THE [NAD+] DECLINES AND CAN BECOME LIMITING. . CELLS HAVE EVOLVED TO RECYCLE NAD+. . . SO GLYCOLYSIS CAN CONTINUE. . 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 4 ATP formed 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
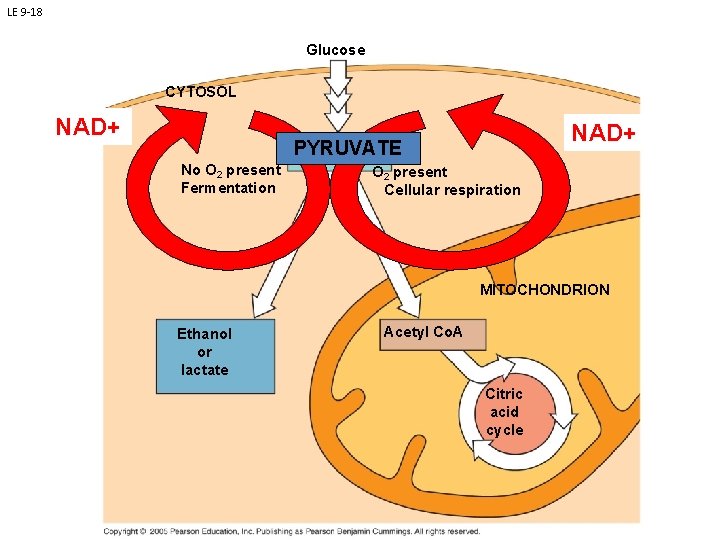
LE 9 -18 Glucose CYTOSOL NAD+ PYRUVATE Pyruvate No O 2 present Fermentation O 2 present Cellular respiration MITOCHONDRION Ethanol or lactate Acetyl Co. A Citric acid cycle

B. Respiration 1. Glycolysis: 2. Anaerobic Respiration a. in plants, fungi, and bacteria: Ethyl Alcohol Fermentation
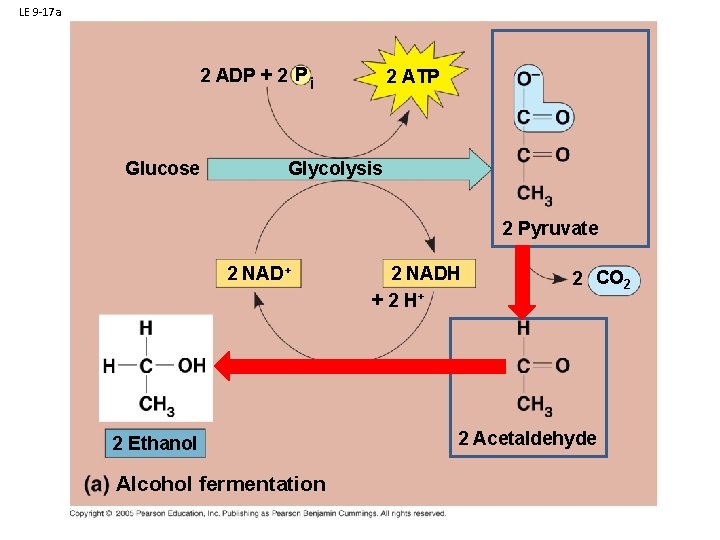
LE 9 -17 a 2 ADP + 2 P i Glucose 2 ATP Glycolysis 2 Pyruvate 2 NAD+ 2 Ethanol Alcohol fermentation 2 NADH + 2 H+ 2 CO 2 2 Acetaldehyde

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration - Glycolosis a. in plants, fungi, and bacteria: Ethyl Alcohol Fermentation b. in animals: Lactic Acid Fermentation
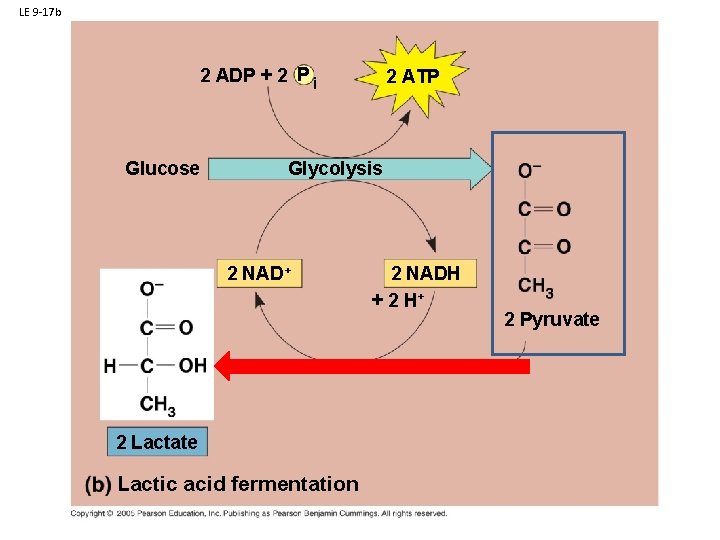
LE 9 -17 b 2 ADP + 2 P i Glucose 2 ATP Glycolysis 2 NAD+ 2 Lactate Lactic acid fermentation 2 NADH + 2 H+ 2 Pyruvate

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration - Glycolosis a. in plants, fungi, and bacteria: Ethyl Alcohol Fermentation b. in animals: Lactic Acid Fermentation In both processes, NAD is recycled so glycolysis can continue… that is the primary goal Energy harvest by glycolysis can continue at a low rate.

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration 3. Aerobic Respiration

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration 3. Aerobic Respiration (in mitochondria of eukaryotic cells) - Had Glycolysis: C 6 (glucose) a - Gateway step: 2 C 3 (pyruvate) + ATP, NADH 2 C 2 (acetyl) + 2 C (CO 2) + NADH b - Citric Acid Cycle: 2 C 2 (acetyl) 4 C (CO 2) + NADH, FADH, ATP c - Electron Transport Chain: convert energy in NADH, FADH to ATP
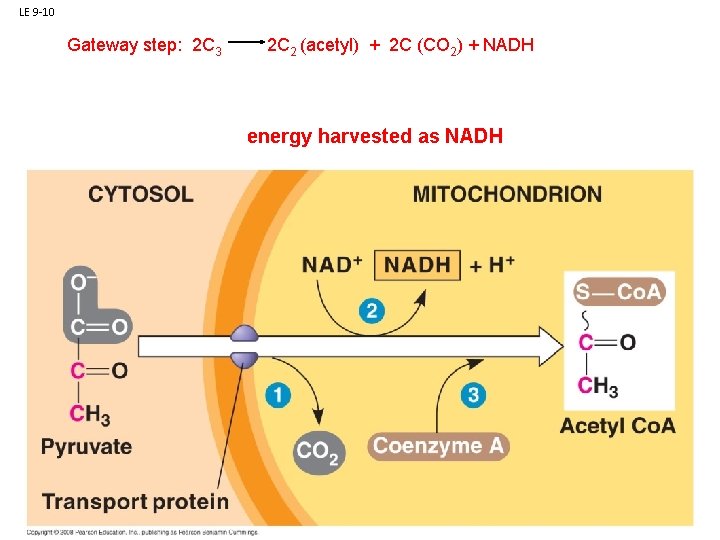
LE 9 -10 Gateway step: 2 C 3 2 C 2 (acetyl) + 2 C (CO 2) + NADH energy harvested as NADH NAD+ NADH + H+ Acetyl Co A Pyruvate Transport protein CO 2 Coenzyme A

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration 3. Aerobic Respiration (in mitochondria of eukaryotic cells) - Had Glycolysis: C 6 (glucose) a - Gateway step: 2 C 3 (pyruvate) + ATP, NADH 2 C 2 (acetyl) + 2 C (CO 2) + NADH b - Citric Acid Cycle: 2 C 2 (acetyl) 4 C (CO 2) + NADH, FADH, ATP c - Electron Transport Chain: convert energy in NADH, FADH to ATP
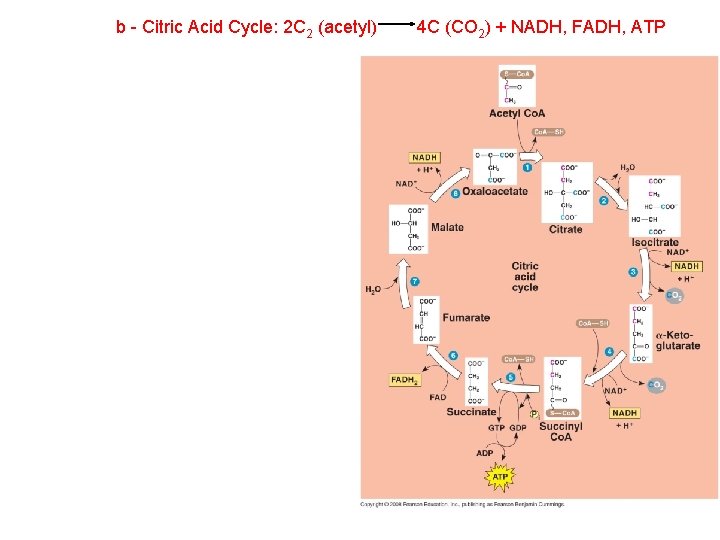
b - Citric Acid Cycle: 2 C 2 (acetyl) 4 C (CO 2) + NADH, FADH, ATP
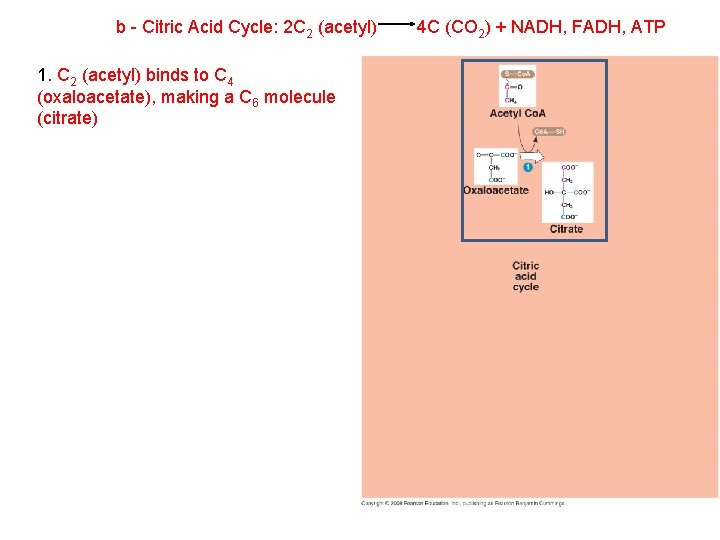
b - Citric Acid Cycle: 2 C 2 (acetyl) 1. C 2 (acetyl) binds to C 4 (oxaloacetate), making a C 6 molecule (citrate) 4 C (CO 2) + NADH, FADH, ATP
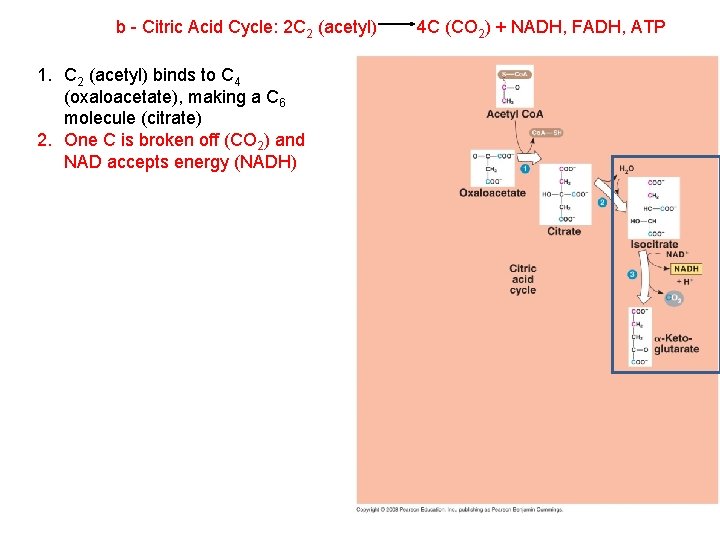
b - Citric Acid Cycle: 2 C 2 (acetyl) 1. C 2 (acetyl) binds to C 4 (oxaloacetate), making a C 6 molecule (citrate) 2. One C is broken off (CO 2) and NAD accepts energy (NADH) 4 C (CO 2) + NADH, FADH, ATP
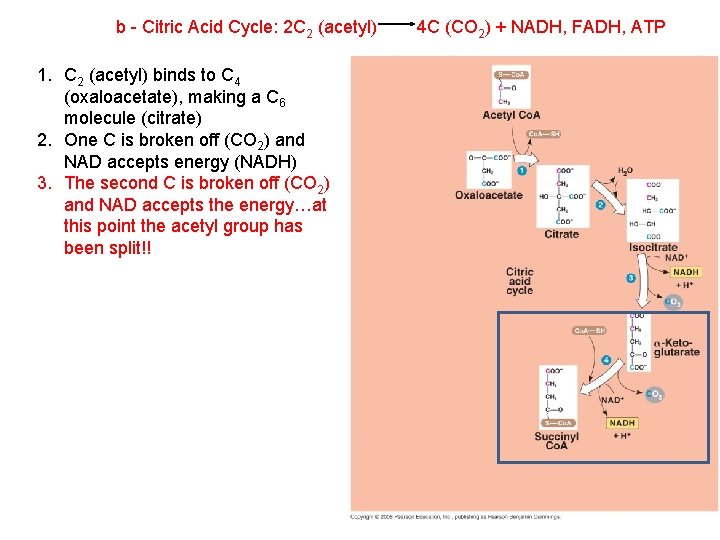
b - Citric Acid Cycle: 2 C 2 (acetyl) 1. C 2 (acetyl) binds to C 4 (oxaloacetate), making a C 6 molecule (citrate) 2. One C is broken off (CO 2) and NAD accepts energy (NADH) 3. The second C is broken off (CO 2) and NAD accepts the energy…at this point the acetyl group has been split!! 4 C (CO 2) + NADH, FADH, ATP
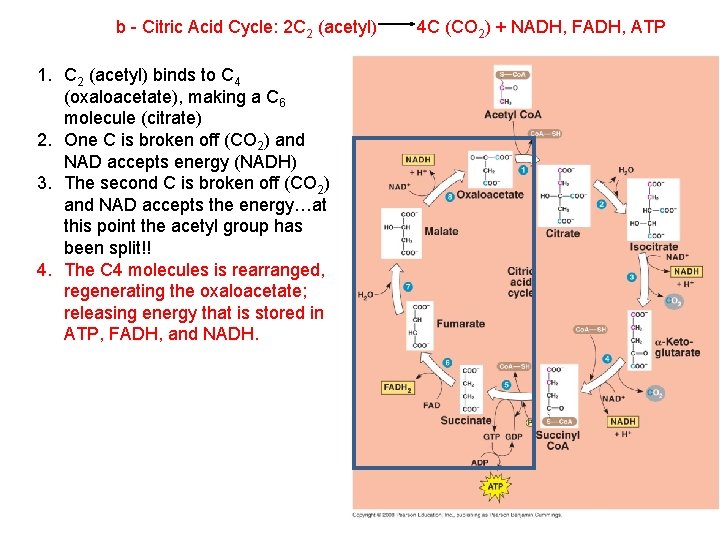
b - Citric Acid Cycle: 2 C 2 (acetyl) 1. C 2 (acetyl) binds to C 4 (oxaloacetate), making a C 6 molecule (citrate) 2. One C is broken off (CO 2) and NAD accepts energy (NADH) 3. The second C is broken off (CO 2) and NAD accepts the energy…at this point the acetyl group has been split!! 4. The C 4 molecules is rearranged, regenerating the oxaloacetate; releasing energy that is stored in ATP, FADH, and NADH. 4 C (CO 2) + NADH, FADH, ATP
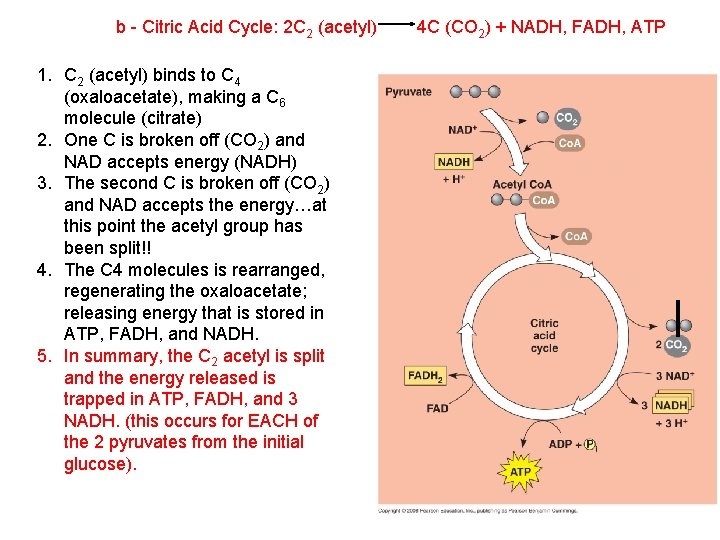
b - Citric Acid Cycle: 2 C 2 (acetyl) 1. C 2 (acetyl) binds to C 4 (oxaloacetate), making a C 6 molecule (citrate) 2. One C is broken off (CO 2) and NAD accepts energy (NADH) 3. The second C is broken off (CO 2) and NAD accepts the energy…at this point the acetyl group has been split!! 4. The C 4 molecules is rearranged, regenerating the oxaloacetate; releasing energy that is stored in ATP, FADH, and NADH. 5. In summary, the C 2 acetyl is split and the energy released is trapped in ATP, FADH, and 3 NADH. (this occurs for EACH of the 2 pyruvates from the initial glucose). 4 C (CO 2) + NADH, FADH, ATP

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration 3. Aerobic Respiration a - Glycolysis: C 6 (glucose) b - Gateway step: 2 C 3 (pyruvate) + ATP, NADH 2 C 2 (acetyl) + 2 C (CO 2) + NADH c - Citric Acid Cycle: 2 C 2 (acetyl) 4 C (CO 2) + NADH, FADH, ATP d - Electron Transport Chain: convert energy in NADH, FADH to ATP
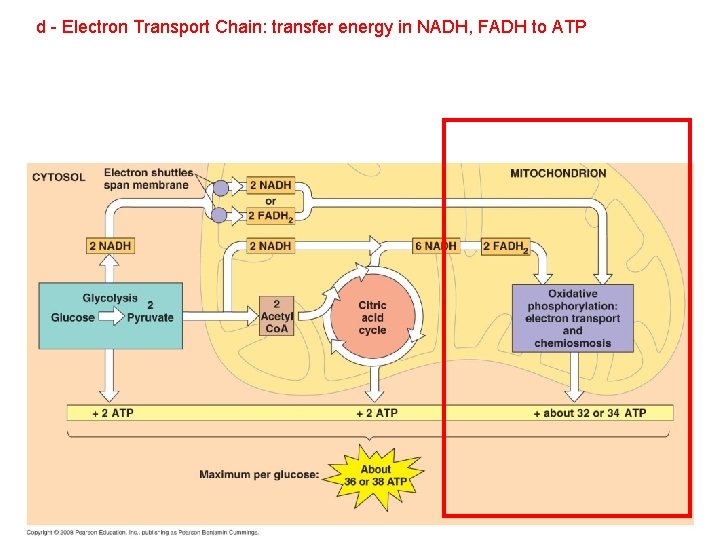
d - Electron Transport Chain: transfer energy in NADH, FADH to ATP
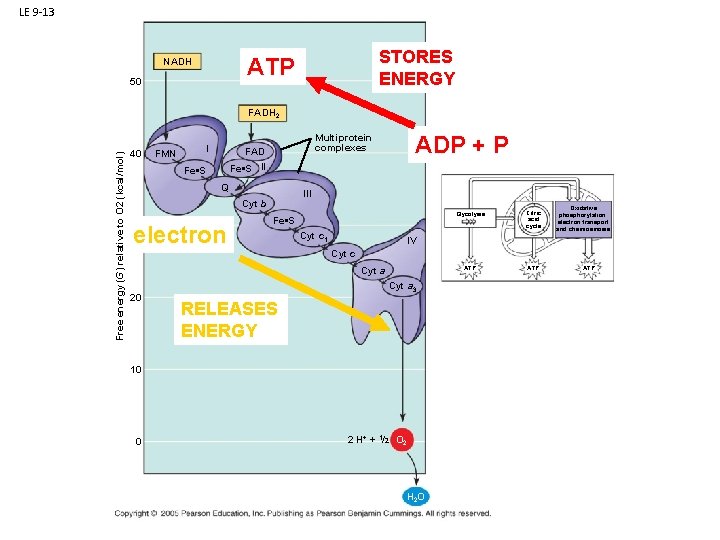
LE 9 -13 STORES ENERGY ATP NADH 50 Free energy (G) relative to O 2 (kcal/mol) FADH 2 40 FMN I Fe • S Q III Cyt b 30 electron ADP + P Multiprotein complexes FAD Fe • S II Fe • S Cyt c 1 Glycolysis Citric acid cycle ATP Oxidative phosphorylation: electron transport and chemiosmosis IV Cyt c Cyt a 20 Cyt a 3 RELEASES ENERGY 10 0 2 H+ + 1/2 O 2 H 2 O ATP
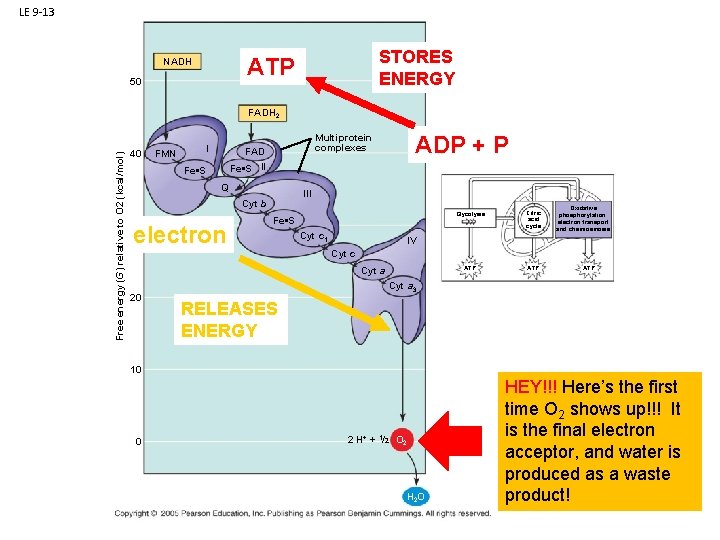
LE 9 -13 STORES ENERGY ATP NADH 50 Free energy (G) relative to O 2 (kcal/mol) FADH 2 40 FMN I Fe • S Q III Cyt b 30 electron ADP + P Multiprotein complexes FAD Fe • S II Fe • S Cyt c 1 Glycolysis Citric acid cycle ATP Oxidative phosphorylation: electron transport and chemiosmosis IV Cyt c Cyt a 20 ATP Cyt a 3 RELEASES ENERGY 10 0 2 H+ + 1/2 O 2 H 2 O HEY!!! Here’s the first time O 2 shows up!!! It is the final electron acceptor, and water is produced as a waste product!
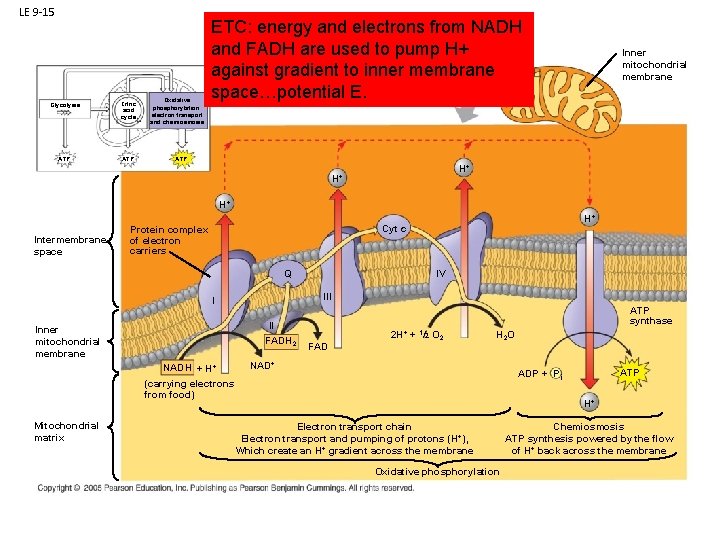
LE 9 -15 Glycolysis Citric acid cycle ATP Oxidative phosphorylation: electron transport and chemiosmosis ETC: energy and electrons from NADH and FADH are used to pump H+ against gradient to inner membrane space…potential E. Inner mitochondrial membrane ATP H+ H+ H+ Intermembrane space H+ Cyt c Protein complex of electron carriers Q III I II FADH 2 Inner mitochondrial membrane NADH + H+ IV ATP synthase FAD 2 H+ + 1/2 O 2 H 2 O NAD+ Mitochondrial matrix ATP ADP + P i (carrying electrons from food) H+ Electron transport chain Electron transport and pumping of protons (H+), Which create an H+ gradient across the membrane Oxidative phosphorylation Chemiosmosis ATP synthesis powered by the flow of H+ back across the membrane
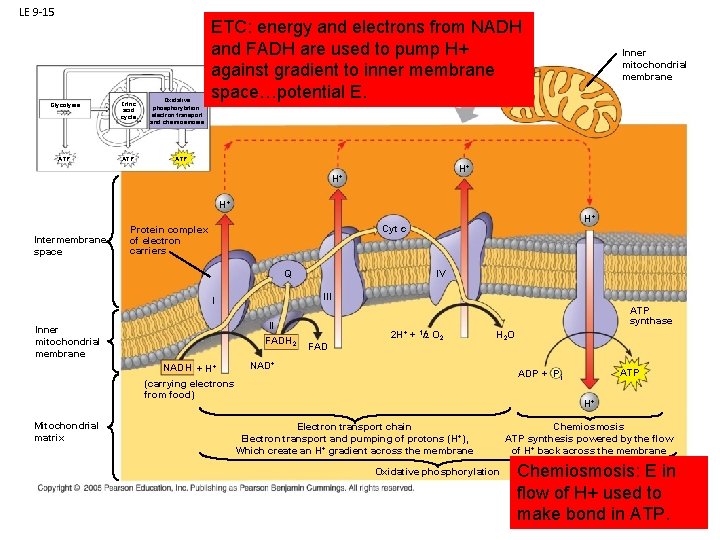
LE 9 -15 Glycolysis Citric acid cycle ATP Oxidative phosphorylation: electron transport and chemiosmosis ETC: energy and electrons from NADH and FADH are used to pump H+ against gradient to inner membrane space…potential E. Inner mitochondrial membrane ATP H+ H+ H+ Intermembrane space H+ Cyt c Protein complex of electron carriers Q III I II FADH 2 Inner mitochondrial membrane NADH + H+ IV ATP synthase FAD 2 H+ + 1/2 O 2 H 2 O NAD+ Mitochondrial matrix ATP ADP + P i (carrying electrons from food) H+ Electron transport chain Electron transport and pumping of protons (H+), Which create an H+ gradient across the membrane Oxidative phosphorylation Chemiosmosis ATP synthesis powered by the flow of H+ back across the membrane Chemiosmosis: E in flow of H+ used to make bond in ATP.

B. Respiration: 1. Glycolysis: 2. Anaerobic Respiration 3. Aerobic Respiration a - Glycolysis: C 6 (glucose) b - Gateway step: 2 C 3 (pyruvate) + ATP, NADH 2 C 2 (acetyl) + 2 C (CO 2) + NADH c - Citric Acid Cycle: 2 C 2 (acetyl) 4 C (CO 2) + NADH, FADH, ATP d - Electron Transport Chain: convert energy in NADH, FADH to ATP - OXYGEN is just an electron ACCEPTOR - WATER is produced as a metabolic waste - All carbons in glucose have been separated - Energy has been harvested and stored in bonds in ATP
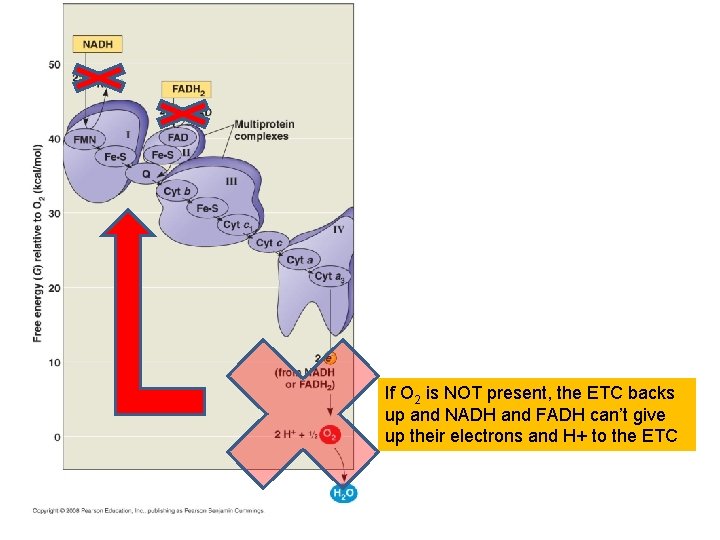
If O 2 is NOT present, the ETC backs up and NADH and FADH can’t give up their electrons and H+ to the ETC
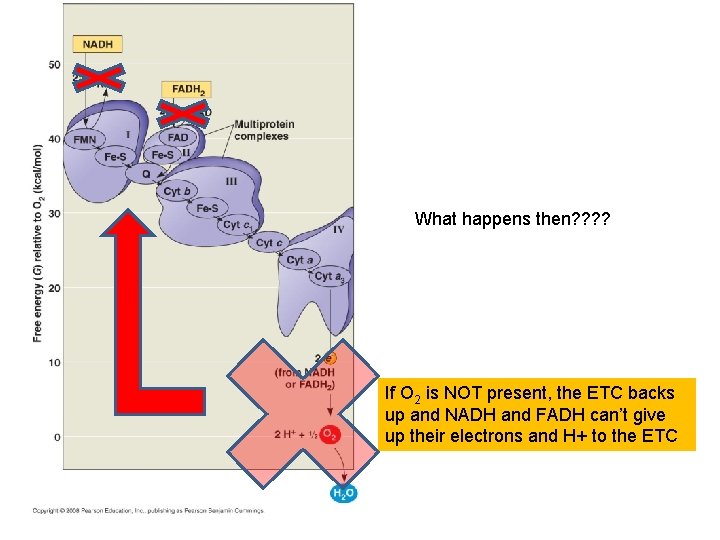
What happens then? ? If O 2 is NOT present, the ETC backs up and NADH and FADH can’t give up their electrons and H+ to the ETC
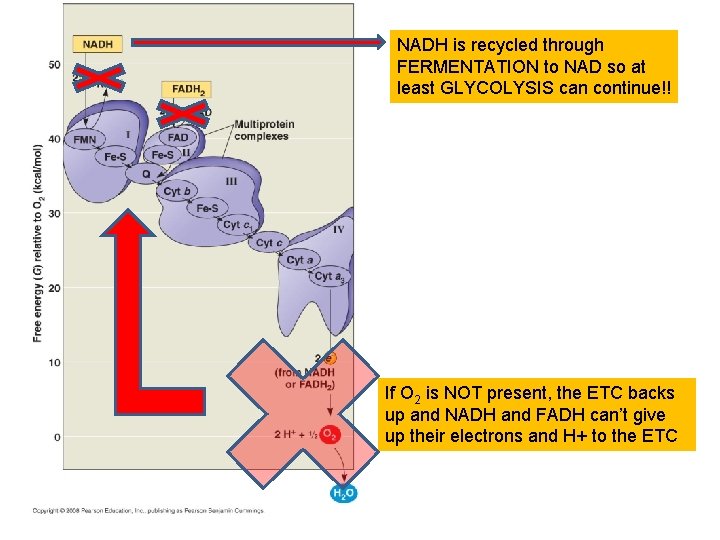
NADH is recycled through FERMENTATION to NAD so at least GLYCOLYSIS can continue!! If O 2 is NOT present, the ETC backs up and NADH and FADH can’t give up their electrons and H+ to the ETC
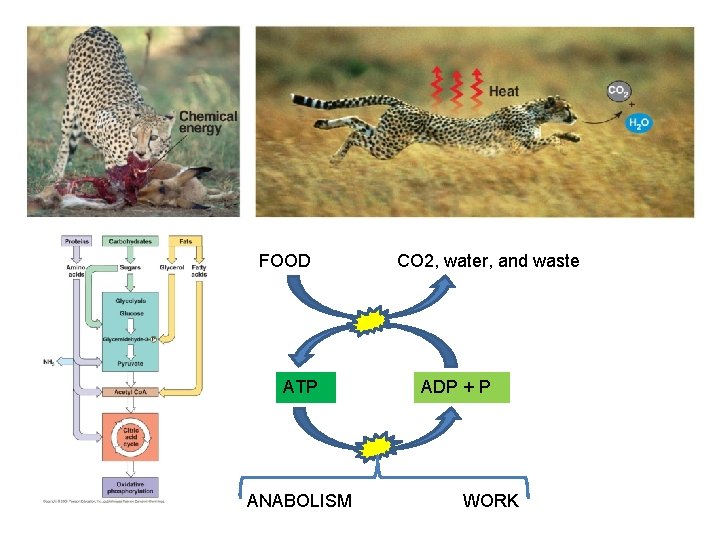
FOOD ATP ANABOLISM CO 2, water, and waste ADP + P WORK
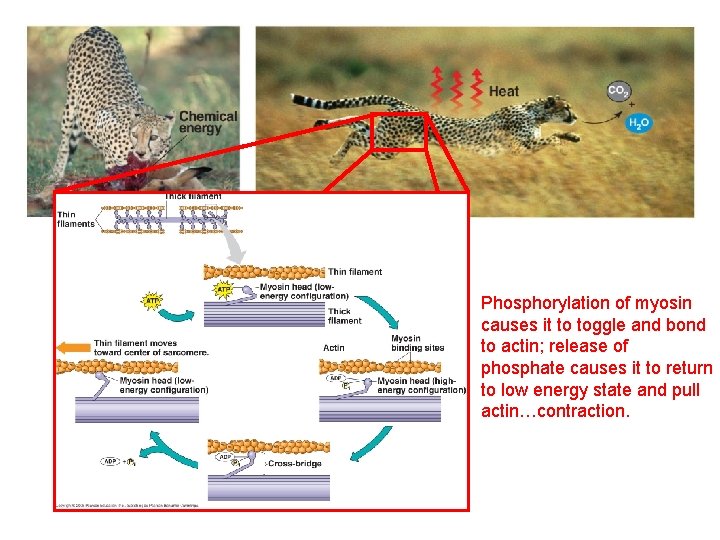
Phosphorylation of myosin causes it to toggle and bond to actin; release of phosphate causes it to return to low energy state and pull actin…contraction.
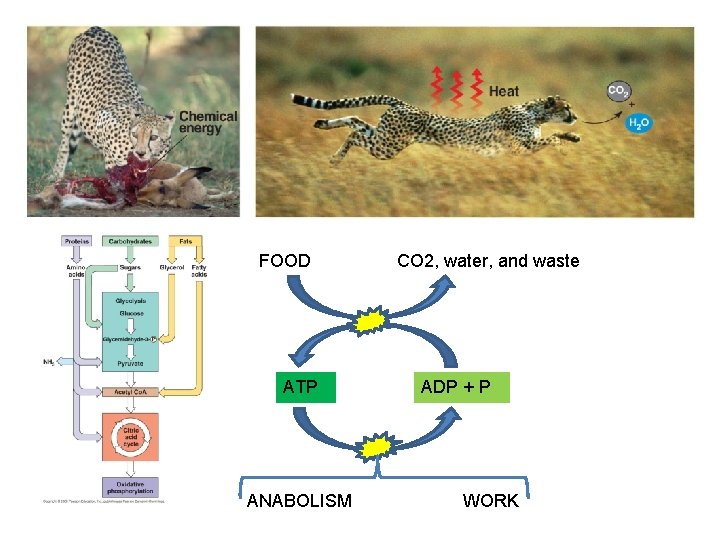
FOOD ATP ANABOLISM CO 2, water, and waste ADP + P WORK
- Slides: 59