ENERGY AND MATTER Specific Heat 1 SPECIFIC HEAT













- Slides: 14

ENERGY AND MATTER Specific Heat 1

SPECIFIC HEAT Specific heat : • the amount of heat that raises the temperature of 1 g of a substance by 1 °C • different for different substances • the SI system has units of J/g C • the metric system has units of cal/g C 2

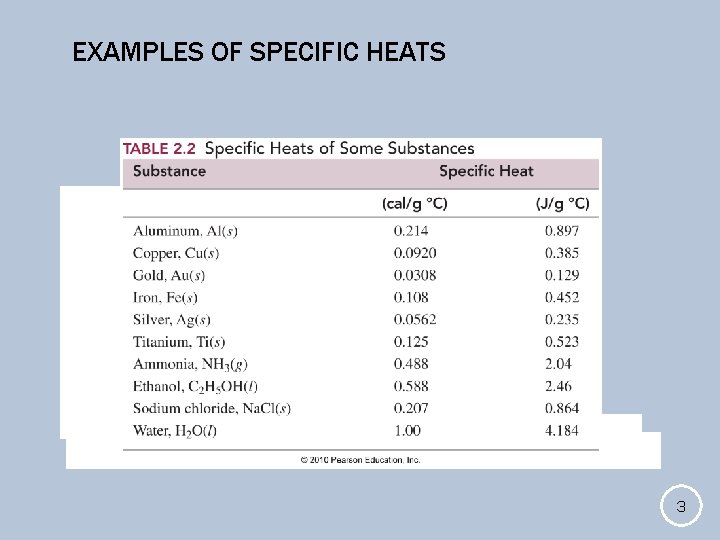
EXAMPLES OF SPECIFIC HEATS 3

LEARNING CHECK A. For the same amount of heat added, a substance with a large specific heat 1. has a smaller increase in temperature 2. has a greater increase in temperature B. When ocean water cools, the surrounding air 1) cools 2) warms 3) stays the same C. Sand in the desert is hot in the day and cool at night. Sand must have a 1) high specific heat 2) low specific heat 4

SOLUTION A. For the same amount of heat added, a substance with a large specific heat 1) has a smaller increase in temperature B. When ocean water cools, the surrounding air 2) warms C. Sand in the desert is hot in the day and cool at night. Sand must have a 2) low specific heat 5

EXAMPLE OF CALCULATING SPECIFIC HEAT What is the specific heat of a metal if 24. 8 g absorbs 65. 7 cal of energy and the temperature rises from 20. 2 C to 24. 5 C? STEP 1 Given: 24. 8 g, 65. 7 cal, ΔT = 20. 2 C to 24. 5 C Need: SHmetal = cal/g C STEP 2 Plan: ΔT = 24. 5 C – 20. 2 C = 4. 3 C SH = Heat(cal) g C 6

EXAMPLE OF CALCULATING SPECIFIC HEAT (CONTINUED) STEP 3 Set up to calculate SH: 65. 7 cal = 0. 62 cal/g C (24. 8 g)(4. 3 C) 7

HEAT EQUATION Rearranging the specific heat expression gives the heat equation. Heat = g x T x cal (or J) = cal (or J) g °C The amount of heat lost or gained by a substance is calculated from the Mass of substance (g) Temperature change ( T) Specific heat of the substance (cal/g °C) or (J/g °C) 8

GUIDE TO CALCULATIONS USING SPECIFIC HEAT 9

SAMPLE CALCULATION FOR HEAT A hot-water bottle contains 750 g of water at 65 °C. If the water cools to body temperature (37 °C), how many calories of heat could be transferred to sore muscles? STEP 1 Given: 750 g of water cools from 65 °C to 37 °C SHwater = 1. 00 cal/g °C Need: calories of heat transferred STEP 2 Calculate the temperature change T: 65 °C – 37 °C = 28 °C 10

SAMPLE CALCULATION FOR HEAT STEP 3 Write the heat equation: Heat (cal) = mass(g) x T x SH STEP 4 Substitute given values and solve for heat: 750 g x 28 °C x 1. 00 cal g °C = 21 000 cal 11

LEARNING CHECK How many kilojoules are needed to raise the temperature of 325 g of water from 15. 0 °C to 77. 0 °C? 1) 20. 2 k. J 2) 84. 3 k. J 3) 105 k. J 12

SOLUTION 2) 84. 3 k. J STEP 1 Given: 325 g of water warms from 15. 0 °C to 77. 0 °C SHwater = 4. 184 J/g °C 1 k. J = 1000 J Need: kilojoules of heat needed STEP 2 Calculate the temperature change T: 77. 0 °C – 15. 0 °C = 62. 0 °C STEP 3 Write the heat equation: Heat (joules) = mass (g) x T x SH 13

SOLUTION (CONTINUED) STEP 4 Substitute given values and solve for heat: 325 g x 62. 0 °C x 4. 184 J x 1 k. J /g °C x 1 k. J/1000 J = 84. 3 k. J (2) 14