Energy and Matter Exchange in the Biosphere Biology

Energy and Matter Exchange in the Biosphere Biology 20 Unit A Pullishy - 2017

Section 1. 1 – How Energy Enters the Biosphere The Need for energy: Photosynthesis and Cellular Respiration • Lets look at the equations • Producers – ‘Self Feeders’ Autotrophs • Typically use sunlight to make energy but there are other forms of producers that don't photosynthesize (Chemosynthesis) • Consumers – ‘Other Feeders’ Heterotrophs • Herbivore, omnivore, carnivore.
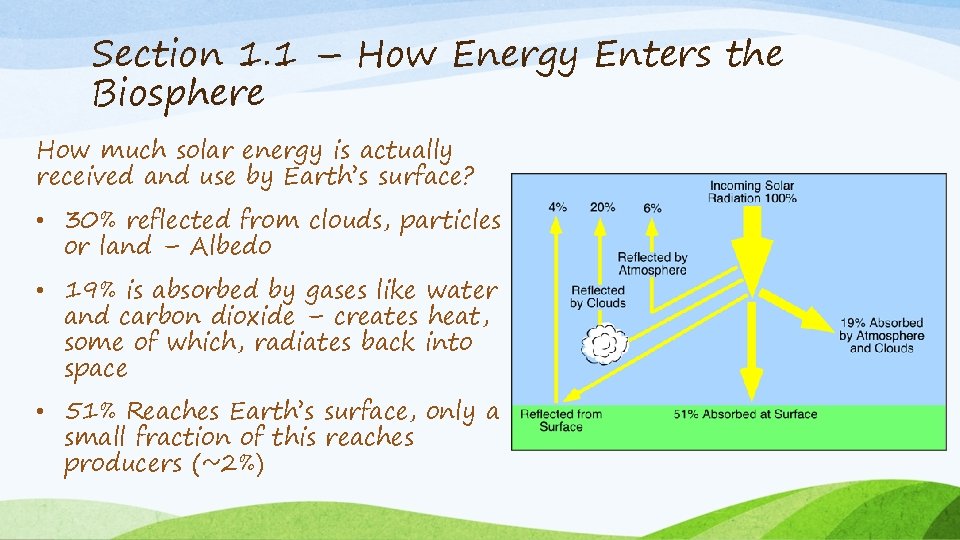
Section 1. 1 – How Energy Enters the Biosphere How much solar energy is actually received and use by Earth’s surface? • 30% reflected from clouds, particles or land – Albedo • 19% is absorbed by gases like water and carbon dioxide – creates heat, some of which, radiates back into space • 51% Reaches Earth’s surface, only a small fraction of this reaches producers (~2%)
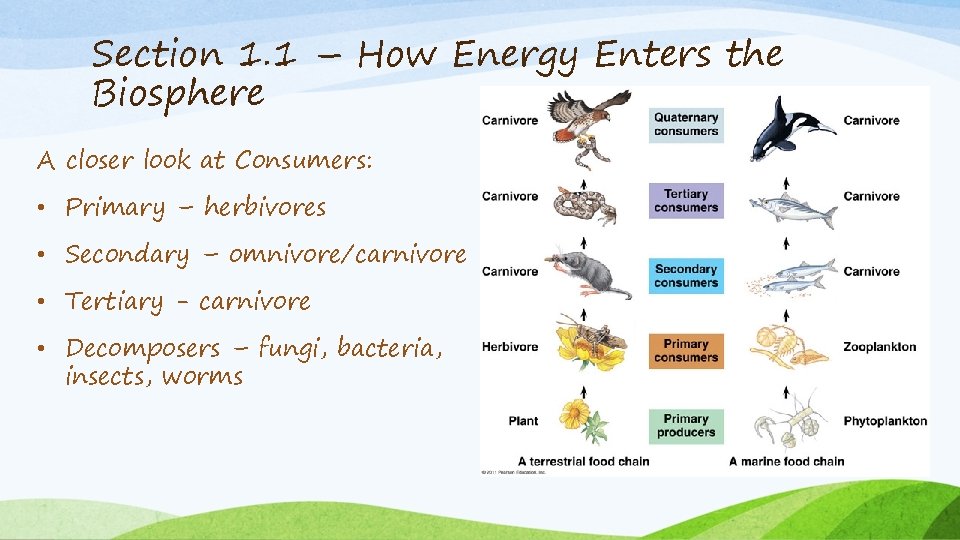
Section 1. 1 – How Energy Enters the Biosphere A closer look at Consumers: • Primary – herbivores • Secondary – omnivore/carnivore • Tertiary - carnivore • Decomposers – fungi, bacteria, insects, worms

Homework Read Section 1. 1 Summary – Page 14 Section 1. 1 Questions 1 -8 due tomorrow Worksheet
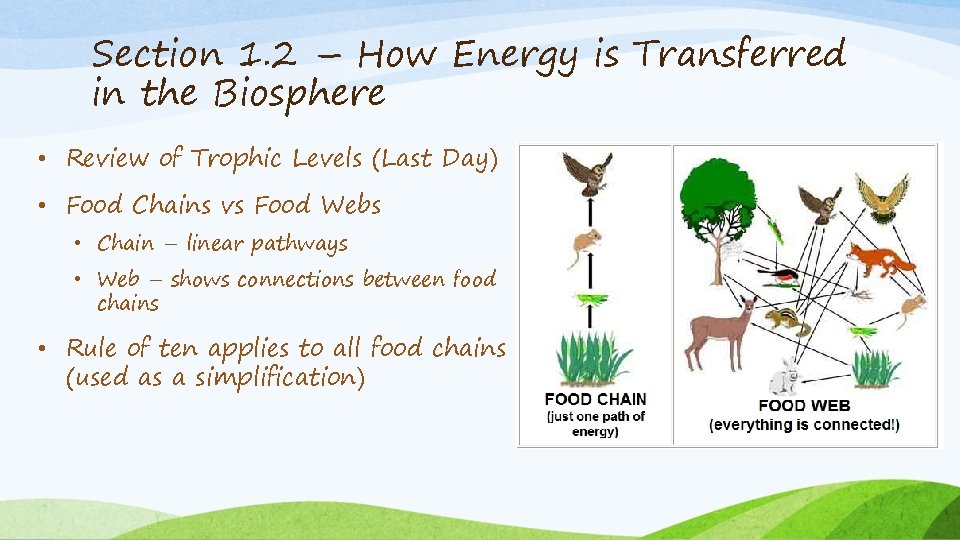
Section 1. 2 – How Energy is Transferred in the Biosphere • Review of Trophic Levels (Last Day) • Food Chains vs Food Webs • Chain – linear pathways • Web – shows connections between food chains • Rule of ten applies to all food chains (used as a simplification)
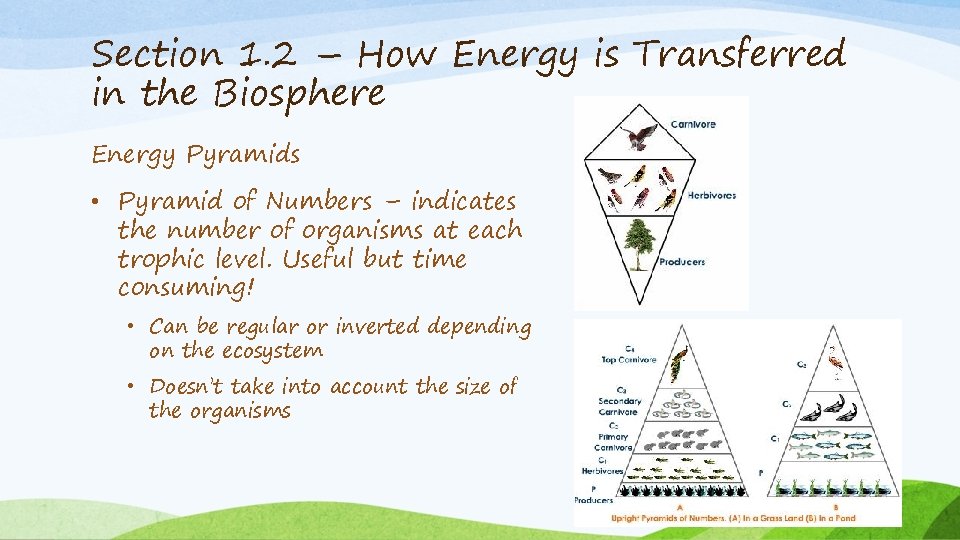
Section 1. 2 – How Energy is Transferred in the Biosphere Energy Pyramids • Pyramid of Numbers – indicates the number of organisms at each trophic level. Useful but time consuming! • Can be regular or inverted depending on the ecosystem • Doesn’t take into account the size of the organisms
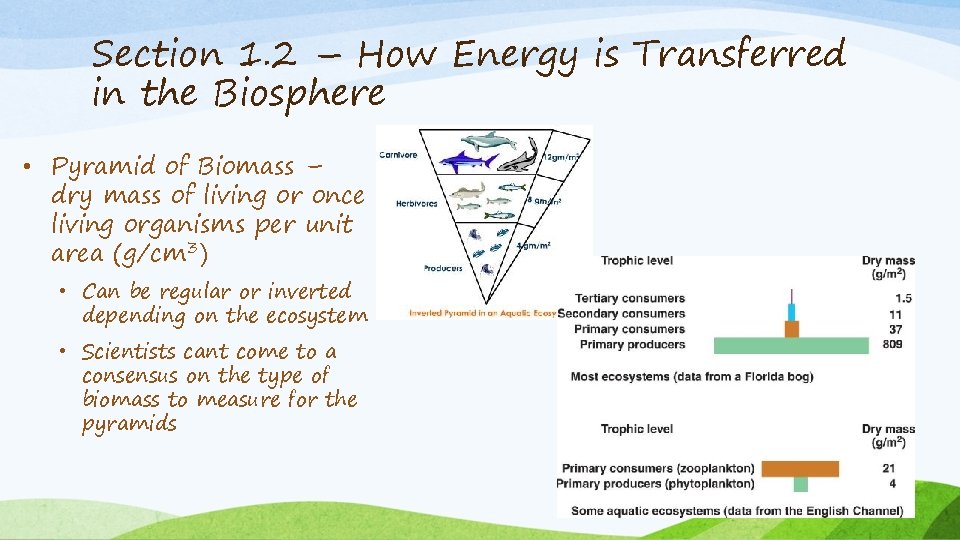
Section 1. 2 – How Energy is Transferred in the Biosphere • Pyramid of Biomass – dry mass of living or once living organisms per unit area (g/cm 3) • Can be regular or inverted depending on the ecosystem • Scientists cant come to a consensus on the type of biomass to measure for the pyramids
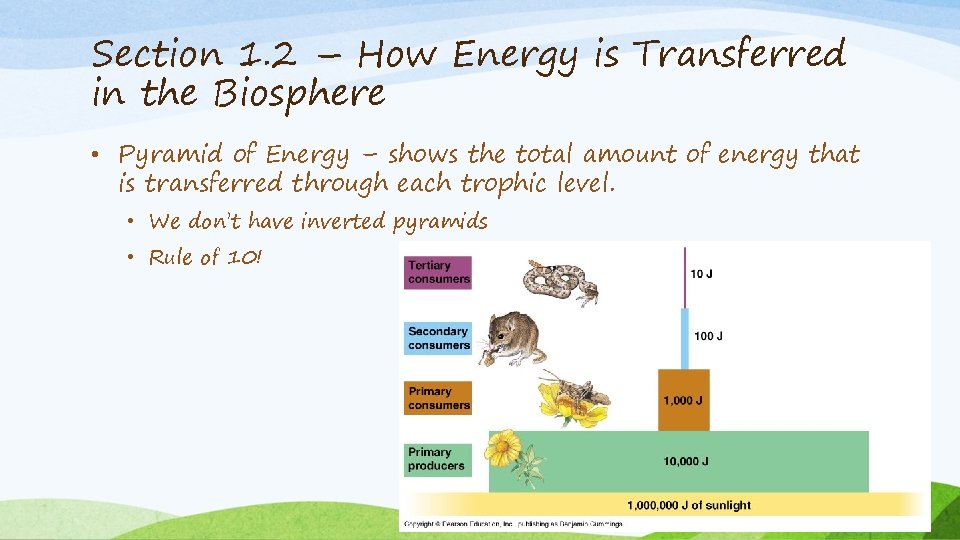
Section 1. 2 – How Energy is Transferred in the Biosphere • Pyramid of Energy – shows the total amount of energy that is transferred through each trophic level. • We don’t have inverted pyramids • Rule of 10!

Homework Assignments: Biomagnification Activity Section 1. 2 Questions 1 -9 due tomorrow Investigation 1 C Ecology of an endangered prairie ecosystem. (Today and tomorrow).

Section 2. 1 – The Role of Water in Cycles of Matter • Hydrological Cycle – involves all phases of water (97% liquid) • Water vapour is a greenhouse gas • Water has several properties that make it a great carrier of other materials and effective at transferring energy • Universal solvent – polar • High boiling and melting point - Hydrogen bonding! • Adhesive and cohesive properties • High specific heat capacity

Video: Water

Section 2. 1 – The Role of Water in Cycles of Matter Activity – Societal uses of water Page 41 • About 97. 5 % of the water that is consumed in Alberta comes from surface water, such as the water in lakes and rivers. The rest of the water that is consumed comes from ground water, which collects when rain or melted snow filters down through the ground accumulates underground in large gaps in the rocks.
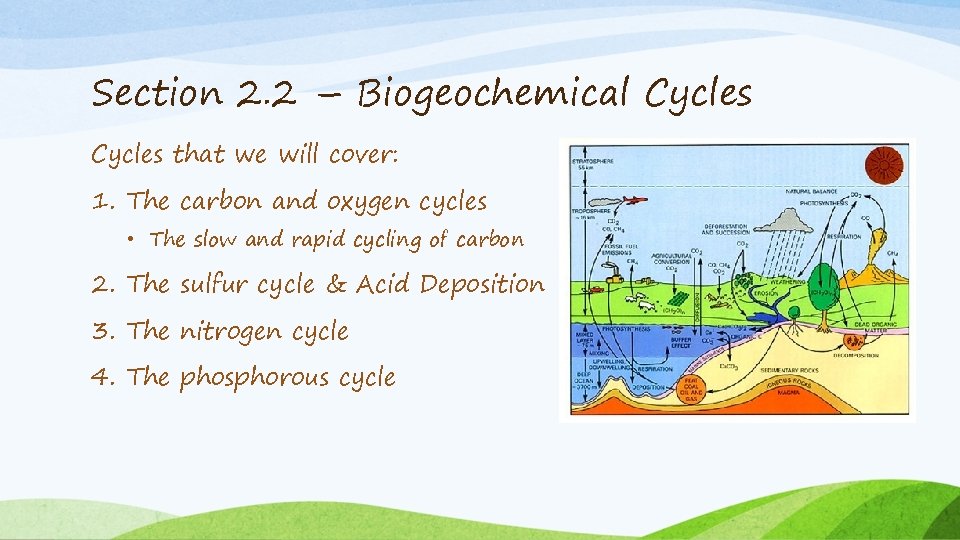
Section 2. 2 – Biogeochemical Cycles that we will cover: 1. The carbon and oxygen cycles • The slow and rapid cycling of carbon 2. The sulfur cycle & Acid Deposition 3. The nitrogen cycle 4. The phosphorous cycle
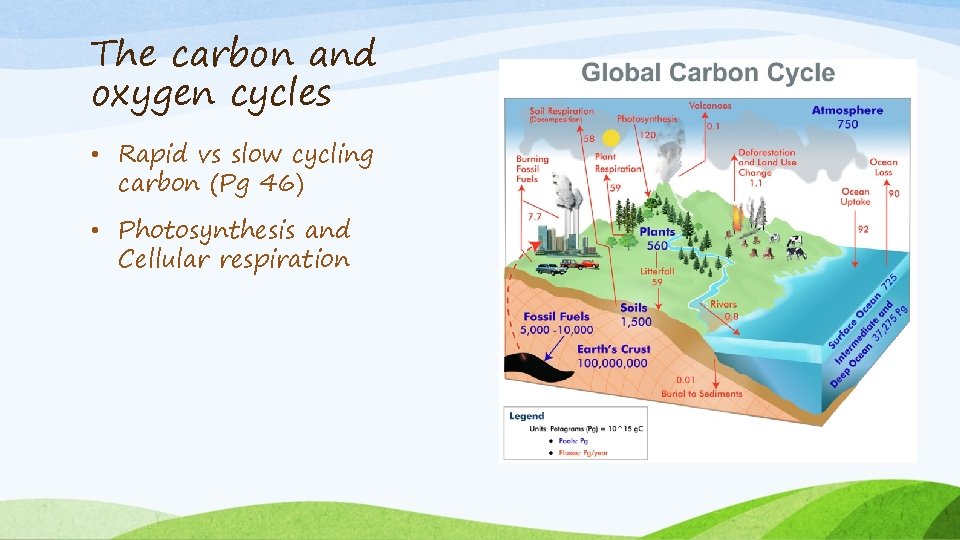
The carbon and oxygen cycles • Rapid vs slow cycling carbon (Pg 46) • Photosynthesis and Cellular respiration

The carbon and oxygen cycles

The sulfur cycle & Acid Deposition • Sulphur is one of the components that make up proteins and vitamins. Proteins consist of amino acids that contain sulphur atoms. • Plants and algae use sulphate (SO 42 -) which dissolves readily in water and can be absorbed. • It enters the atmosphere through both natural and human sources. Natural recourses can be for instance volcanic eruptions, bacterial processes, evaporation from water, or decaying organisms.

The sulfur cycle & Acid Deposition • When sulphur enters the atmosphere through human activity, this is mainly a consequence of industrial processes where sulphur dioxide (SO 2) and hydrogen sulphide (H 2 S) gases are emitted on a wide scale. • When sulphur dioxide enters the atmosphere it will react with oxygen to produce sulphur trioxide gas (SO 3), or with other chemicals in the atmosphere, to produce sulphur salts. Sulphur dioxide may also react with water to produce sulphuric acid (H 2 SO 4), both of these can lead to acid precipitation.
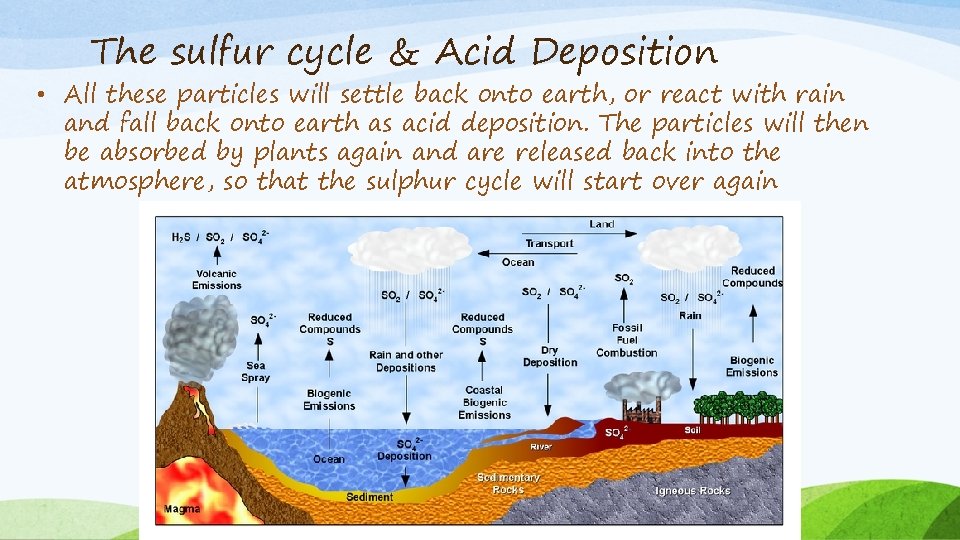
The sulfur cycle & Acid Deposition • All these particles will settle back onto earth, or react with rain and fall back onto earth as acid deposition. The particles will then be absorbed by plants again and are released back into the atmosphere, so that the sulphur cycle will start over again

The nitrogen cycle is the set of biogeochemical processes by which nitrogen undergoes chemical reactions, changes form, and moves through difference reservoirs on earth, including living organisms. Nitrogen is required for all organisms to live and grow because it is the essential component of DNA, RNA, and protein. However, most organisms cannot use atmospheric nitrogen, the largest reservoir. The five processes in the nitrogen cycle • i. Nitrogen fixation • ii. Nitrogen uptake • iii. Nitrogen mineralization • iv. Nitrification • v. De-nitrification

The nitrogen cycle I. Nitrogen fixation: N 2 -> NH 4+ • Nitrogen fixation is the process wherein N 2 is converted to ammonium, essential because it is the only way that organisms can attain nitrogen directly from the atmosphere. • Nitrogen fixing bacteria often form symbiotic relationships with host plants. This symbiosis is well-known to occur in the legume family of plants (e. g. beans, peas, and clover). In this relationship, nitrogen fixing bacteria inhabit legume root nodules and receive carbohydrates and a favourable environment from their host plant in exchange for some of the nitrogen they fix. • There also nitrogen fixing bacteria that exist without plant hosts, known as free-living nitrogen fixers. In aquatic environments, blue-green algae (really a bacteria called cyanobacteria) is an important free-living nitrogen fixer.

The nitrogen cycle II. Nitrogen uptake: NH 4+ -> Organic N • The ammonia produced by nitrogen fixing bacteria is usually quickly incorporated into protein and other organic nitrogen compounds, either by a host plant, the bacteria itself, or another soil organism. III. Nitrogen mineralization: Organic N ->NH 4+ • After nitrogen is incorporated into organic matter, it is often converted back into inorganic nitrogen by a process called nitrogen decay. When organisms die, decomposers (such as bacteria and fungi) consume the organic matter and lead to the process of decomposition. • During this process, a significant amount of the nitrogen contained within the dead organism is converted to ammonium. Once in the form of ammonium, nitrogen is available for use by plants or further transformation into nitrate (NO 3–) through the process called nitrification.

The nitrogen cycle IV. Nitrification: NH 4+ -> NO 3– • Some of the ammonium produced by decomposition is converted to nitrate via a process called nitrification. Nitrification requires the presence of oxygen, so nitrification can happen only in oxygen-rich environments like circulating or flowing waters and the very surface layers of soils and sediments. • Ammonium ions are positively charged and therefore stick to negatively charged clay particles and soil organic matter. The positive charge prevents ammonium nitrogen from being washed out of the soil (or leached) by rainfall. • In contrast, the negatively charged nitrate ion is not held by soil particles and so can be washed down the soil profile, leading to decreased soil fertility and nitrate enrichment of downstream surface and groundwater’s.

The nitrogen cycle V. De-nitrification: NO 3– -> N 2+ N 2 O • Through de-nitrification, oxidized forms of nitrogen such as nitrate and nitrite (NO 2 ) are converted to di-nitrogen (N 2) and, to a lesser extent, nitrous oxide gas. De-nitrification is an anaerobic process that is carried out by denitrifying bacteria, which convert nitrate to nitrogen in the following sequence: NO 3– -> NO 2– -> NO ->N 2 • Nitric oxide and nitrous oxide are both environmentally important gases. Nitric oxide (NO) contributes to smog, and nitrous oxide (N 2 O) is an important greenhouse gas, thereby contributing to global climate change.

The phosphorous cycle • Phosphorus is a chemical element found on Earth in numerous compound forms, such as the phosphate ion (PO 43 -), located in water, soil and sediments. The quantities of phosphorus in soil are generally small, and this often limits plant growth. That is why people often apply phosphate fertilisers on farmland. Animals absorb phosphates by eating plants or plant-eating animals. • Phosphorus is an essential nutrient for animals and plants. It plays a critical role in cell development and is a key component of molecules that store energy, such as ATP (adenosine triphosphate), DNA and lipids (fats and oils).

The phosphorous cycle Key steps of the phosphorus cycle • Over time, rain and weathering cause rocks to release phosphate ions and other minerals. This inorganic phosphate is then distributed in soils and water. • Plants take up inorganic phosphate from the soil. The plants may then be consumed by animals. Once in the plant or animal, the phosphate is incorporated into organic molecules such as DNA. When the plant or animal dies, it decays, and the organic phosphate is returned to the soil. • Within the soil, organic forms of phosphate can be made available to plants by bacteria that break down organic matter to inorganic forms of phosphorus. This process is known as mineralisation. • Phosphorus in soil can end up in waterways and eventually oceans. Once there, it can be incorporated into sediments over time.
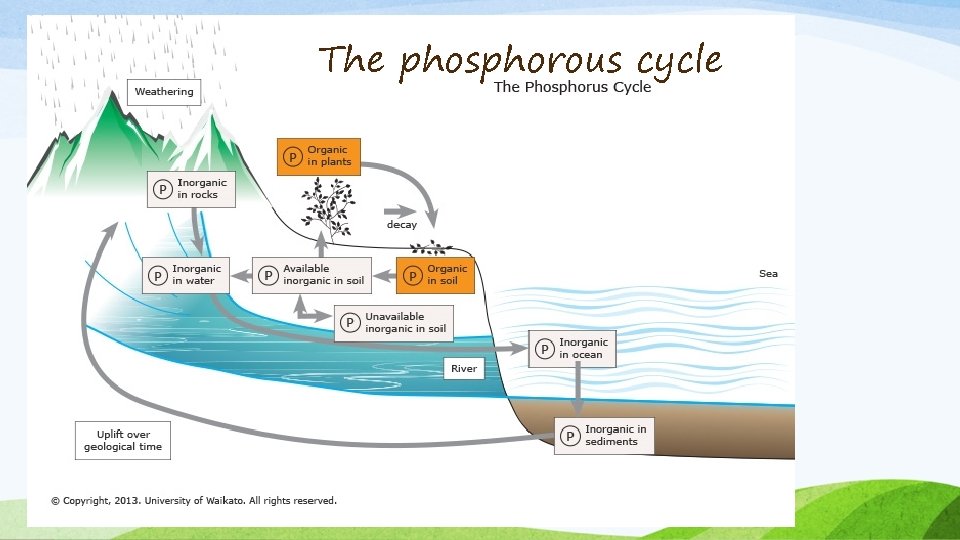
The phosphorous cycle

Homework Section 2. 1 Questions 1 -9 Section 2. 2 Questions 1 -8 Read Section 2. 3 (not covered in class)

Section 2. 2 – Biogeochemical Cycles Project • You will be assigned one of the biogeochemical cycles as the focus for your project. • Teach the class about your cycle – be sure to cover all of the curricular outcomes associated with your cycle(s) • Describe and illustrate your cycle(s) • Research the potential environmental problems associated with this cycle. • Create an information board/ lesson to inform the class about these issues. You need to provide newspaper articles or scientific articles. • You must include a reference section/bibliography.

Section 2. 2 – Biogeochemical Cycles Project • A description/explanation of the cycle you have chosen. • Something for students to do/work on/participate in to ensure their learning • Identify and discuss at least one environmental issue associated with the cycle you have chosen. (you can use your research here) For example: • What threat does it pose to the environment? • How much of a concern is this issue? Who does it affect? • For the issue(s) you identify, come up with two possible solutions to the problem. The solutions need to be realistic.

Section 2. 3 – The Balance of Matter and Energy Exchange • Productivity – the rate at which an ecosystems producers capture and store energy in a given unit of time. The rate at which organisms produce biomass • Depends on many variables: number of producers, amount of sunlight, nutrient availability, time of year, precipitation, climate ect…

Section 2. 3 – The Balance of Matter and Energy Exchange Activity: Biosphere in a bottle! Similar to the Investigation in your textbook on page 57.

Homework Unit A Review Questions 1 -23 Page 68 -69 Chapter 2 Test next Tuesday. Unit Exam coming up Thursday.
- Slides: 33