Energy and Earth Energy Its all about energy

Energy and Earth

Energy It’s all about energy. Energy and its transfer controls the Universe and Earth’s systems. What is energy? The ability to do work or make a change.

Sources of Energy Electromagnetic – Energy from the electromagnetic spectrum • Chemical – Energy released during a chemical reaction. • Nuclear – Energy released during fission or fusion •

Electromagnetic Energy • Energy that radiates from the sun. • Electromagnetic spectrum (EMS) arranged by wavelength. • All EM waves travel at the speed of light. • Only difference between EM waves is wavelength.

Chemical Energy • Energy released during a chemical reaction. • Burning

Nuclear Fusion • Atoms fuse together to form a larger atom. • Results in a release of energy. • Sun • Hydrogen atoms fuse together to form a helium atom.

Nuclear Fission • The splitting of an atom’s nucleus into two smaller nuclei. • Results in a release of energy. • Earth’s core • Atoms decay creating heat.

It’s Fairly Simple • Earth has two sources of energy: – Radioactive decay in the core (fission) - Internal – Fusion in the sun radiates to the Earth in the form of electromagnetic energy - External • Both sources of energy are nuclear.

States of Energy • Potential – Stored energy – gasoline, snow pack, reservoir. Kinetic – The energy of motion – flowing water, waves, landslides, thunderstorm. •

Thermal Energy • Heat is thermal energy transferring from one substance to another. • Differences in temperature cause the transfer. • Matter does NOT contain heat. • Matter contains thermal energy.

Thermal Energy (cont. ) • Heat is thermal energy in transit. • After heat is transferred, it ceases to be heat and becomes thermal energy again.
Transfer of Energy • Three ways to transfer energy 1 • Conduction – the movement of heat (energy) in solids by its contact with a hotter object. – slow process (core-mantle)

Transfer of Energy 2 • Convection – the movement of heat by currents in liquids & gases because of temperature and density differences. - Fast process (air, upper mantle)

Transfer of Energy 3 • Radiation – the transfer of heat in gases or a vacuum in the form of waves. • Hot objects radiate heat. – Very fast process (infrared, ultraviolet waves)
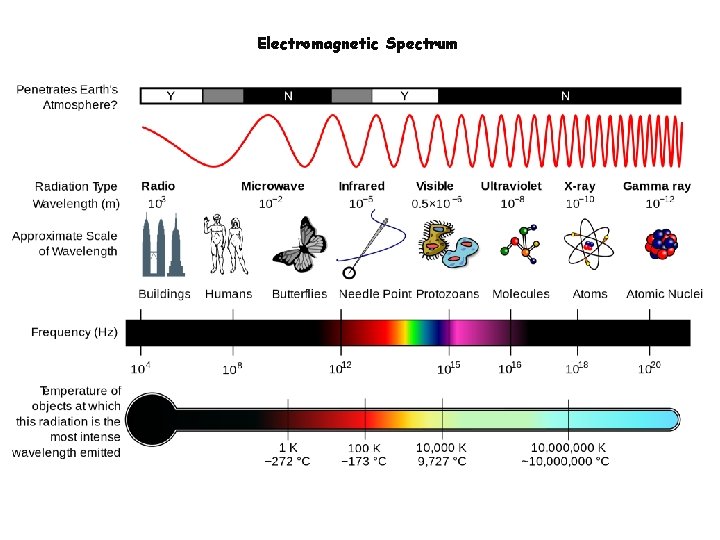
Electromagnetic Spectrum
The Temperature Scales
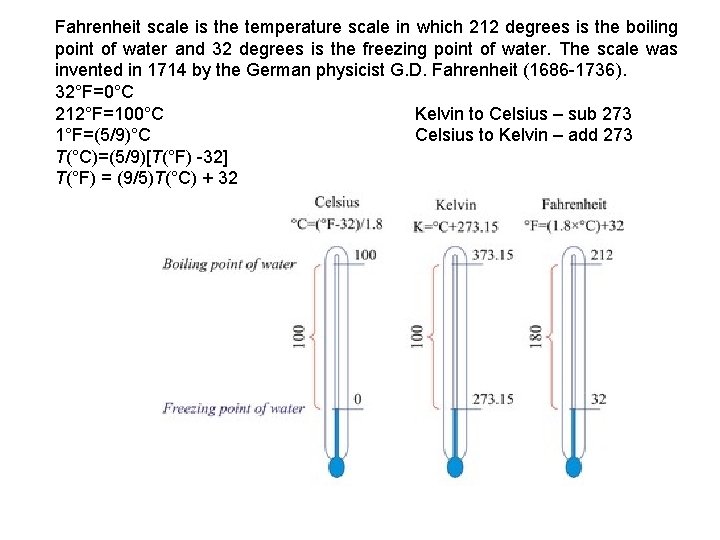
Fahrenheit scale is the temperature scale in which 212 degrees is the boiling point of water and 32 degrees is the freezing point of water. The scale was invented in 1714 by the German physicist G. D. Fahrenheit (1686 -1736). 32°F=0°C 212°F=100°C Kelvin to Celsius – sub 273 1°F=(5/9)°C Celsius to Kelvin – add 273 T(°C)=(5/9)[T(°F) -32] T(°F) = (9/5)T(°C) + 32

Atoms Become excited as the added energy causes electrons to jump up an energy level They move faster and farther apart.

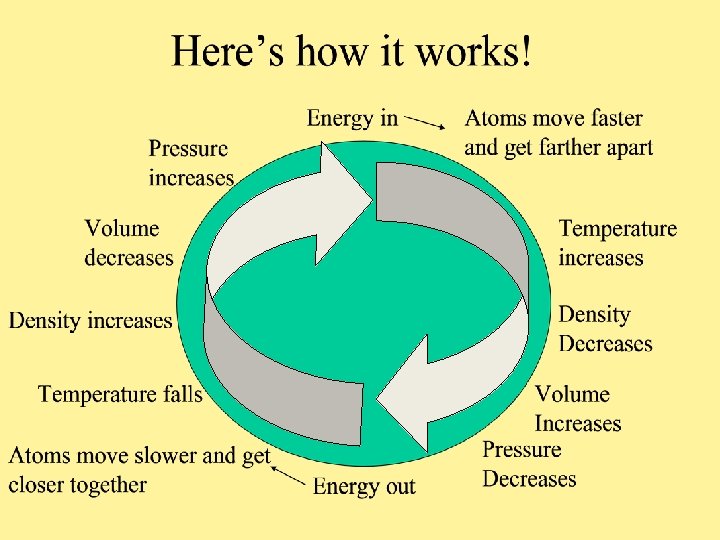
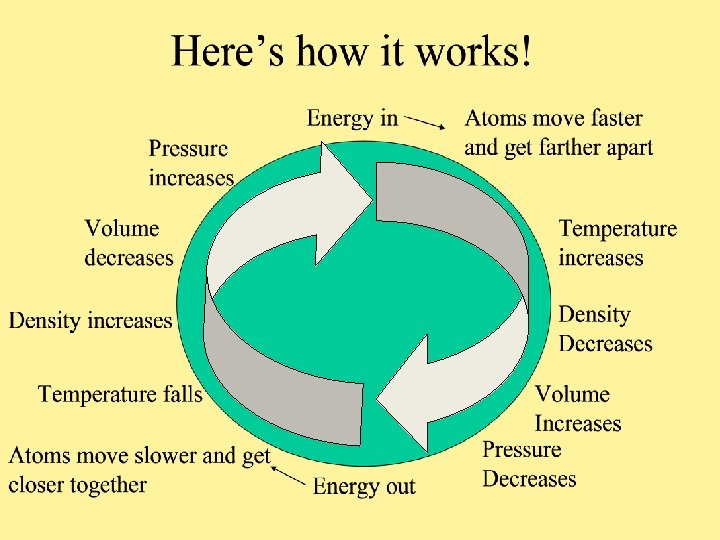
Temperature • Is the measure of how fast atoms are moving. The more energy into the system the faster the atoms will move and the higher the temperature. •

Density Is the measure of the number of atoms in a given area. • The faster the atoms move and the farther apart they get the fewer there will be in the same area. •

Volume • Is the space a set number of atoms occupy. The farther apart the atoms are the more space they take up. •

Pressure Is the measure of how many atoms are pushing on a given space. • The fewer the atoms on a given space the lower the pressure. •

A Note on Pressure will decrease as long as the atoms are able to move freely. • If the atoms are contained the pressure will increase as they try to spread out. • • Think of a tea kettle and a pot of boiling water. Increased pressure Decreased pressure

Laws of energy (or thermodynamics): 1 st: Law of conservation: You can’t create or destroy energy…you can only change its form. 2 nd: Law of entropy: When you increase energy, you always decrease order. When you decrease energy, you increase order (imagine a classroom full of energy…there is no order; but a classroom with no energy has lots of order) 3 rd: Absolute zero: The coldest possible temperature is -273 degrees Celsius. At that temperature the atoms stop moving completely. Even the atoms in solids stop vibrating. This is ‘absolute order’ Entropy is a measure of the unavailable energy in a closed thermodynamic system that is also usually considered to be a measure of the system's disorder.

Questions 1. The second law of thermodynamics says that ______. A) energy can be created but not destroyed B) energy can be created and destroyed C) energy can neither be created nor destroyed D) one usable form of energy can be completely converted into another usable form E) one usable form of energy cannot be completely converted into another usable form 2. _______ is a measure of randomness or disorder. A) Metabolism B) Synthesis C) Entropy D) A coupled reaction E) Thermodynamics

Questions 3. Which of the following processes produces a decrease in the entropy of the system? A. boiling water to form steam B. dissolving salt into water C. mixing two gases of different temperature into one container. D. freezing water to form ice E. melting ice to form water 4. The ______ law of thermodynamics states that energy can neither be created nor destroyed. A) first B) second C) third D) fourth E) zeroith

Focused Free Write: Connect these words/ideas together in a sensible manner (in context). thermometer molecules expands volume farther apart dense (density) temperature energy

Equilibrium / Homeostasis • Nature wants everything to be equal and balanced. • If hot and cold water are put together what happens? • The energy is spread equally until the temperature is the same throughout. • Open a bottle of perfume. Does the fragrance stay in the bottle or diffuse?

ENERGY & IT’S TRANSFER DRIVES THE EARTH SYSTEMS • OCEAN CURRENTS • PLATE TECTONICS • WEATHER SYSTEMS • RADIOACTIVE DECAY

- Slides: 32