Energy and Chemical Reactions Metabolism The totality of

Energy and Chemical Reactions Metabolism The totality of an organism’s chemical reactions Arises from interactions between molecules The transformation of matter and energy Subject to the laws of thermodynamics

Energy • The ability to move and change matter • Life requires energy. • Almost all energy for life is derived from the sun. • A productive way of viewing life is to see it as a set of coordinated chemical reactions. • This leads to an obvious question – What determines what chemical reactions are possible?

Thermodynamics l The study of energy Chemical energy transformation – The first law of thermodynamics • Energy can be transferred and transformed • Energy cannot be created or destroyed (a) First law of thermodynamics: Energy can be transferred or transformed but Neither created nor destroyed. For example, the chemical (potential) energy in food will be converted to the kinetic energy of the cheetah’s movement in (b).

Thermodynamics – The second law of thermodynamics • Spontaneous changes that do not require outside energy increase the entropy of the universe • Entropy= disorder in the universe entropy is always increasing. Heat co 2 + H 2 O (b) Second law of thermodynamics: Every energy transfer or transformation increases the disorder (entropy) of the universe. For example, disorder is added to the cheetah’s surroundings in the form of heat and the small molecules that are the by-products of metabolism.

The Direction of Spontaneous Reactions (and what it takes to go the other way)

Energy can be converted from one form to another On the platform, a diver has more potential energy. Climbing up converts kinetic energy of muscle movement to potential energy. Diving converts potential energy to kinetic energy. In the water, a diver has less potential energy.

Flow of energy through life l Life is built on chemical reactions – transforming energy from one form to another sun solar energy ATP & organic molecules ATP & organic molecules
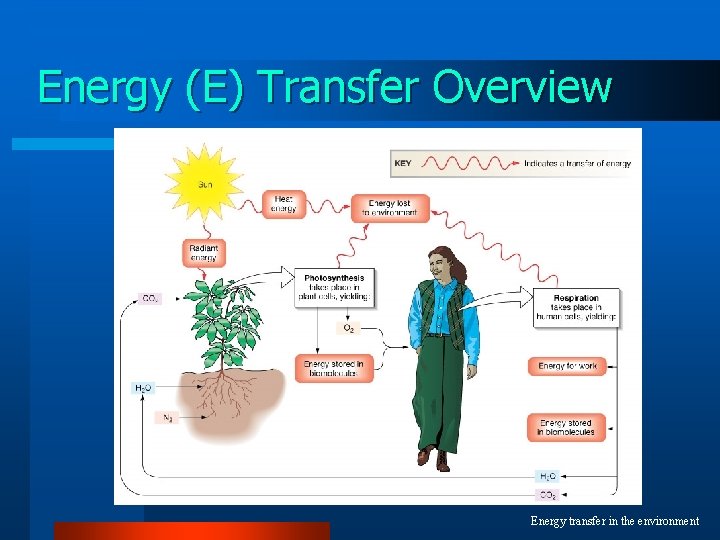
Energy (E) Transfer Overview Energy transfer in the environment
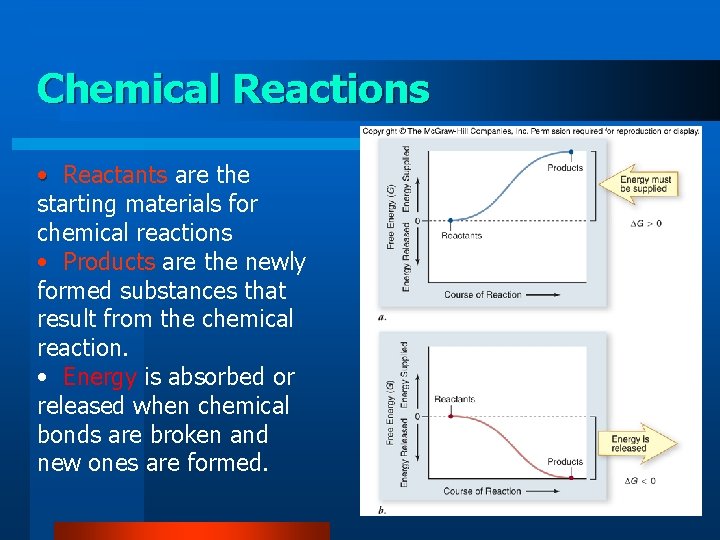
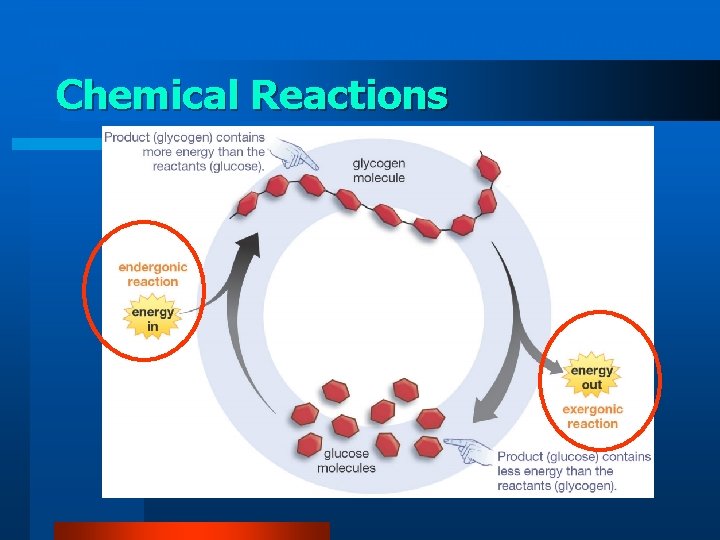
Chemical Reactions • Reactants are the starting materials for chemical reactions • Products are the newly formed substances that result from the chemical reaction. • Energy is absorbed or released when chemical bonds are broken and new ones are formed.
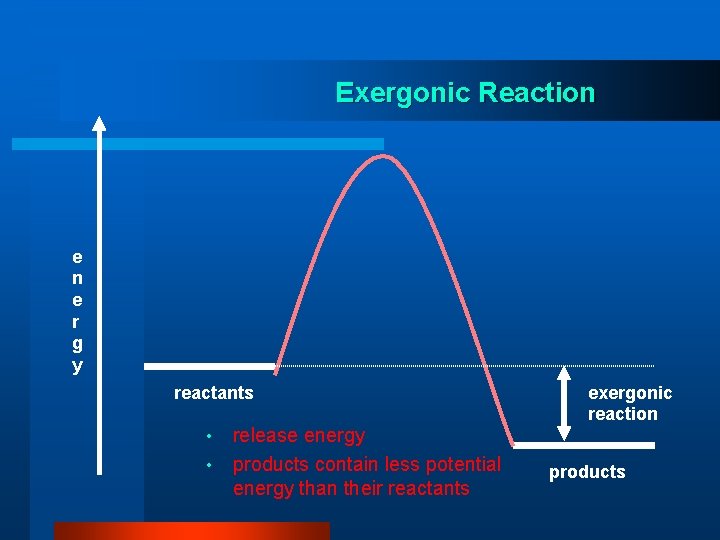
Exergonic Reaction e r g y reactants • • release energy products contain less potential energy than their reactants exergonic reaction products
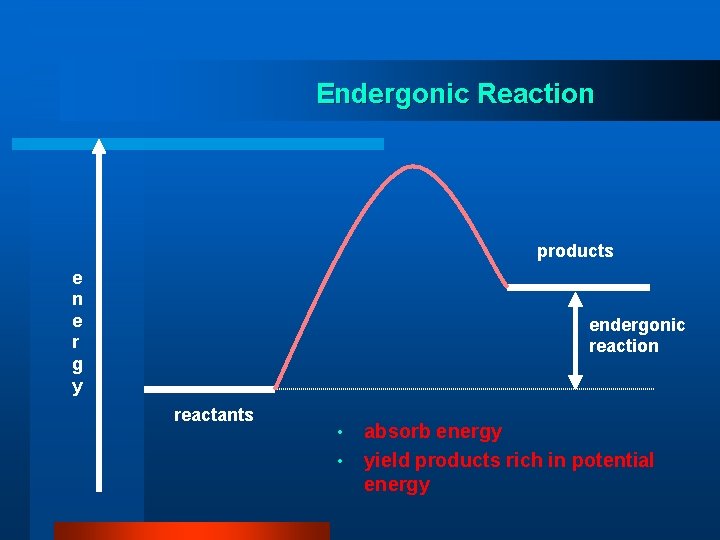
Endergonic Reaction products e n e r g y endergonic reaction reactants • • absorb energy yield products rich in potential energy
The “Nature of Life” – Coupling Favorable to Unfavorable Reactions Chemical Reactions
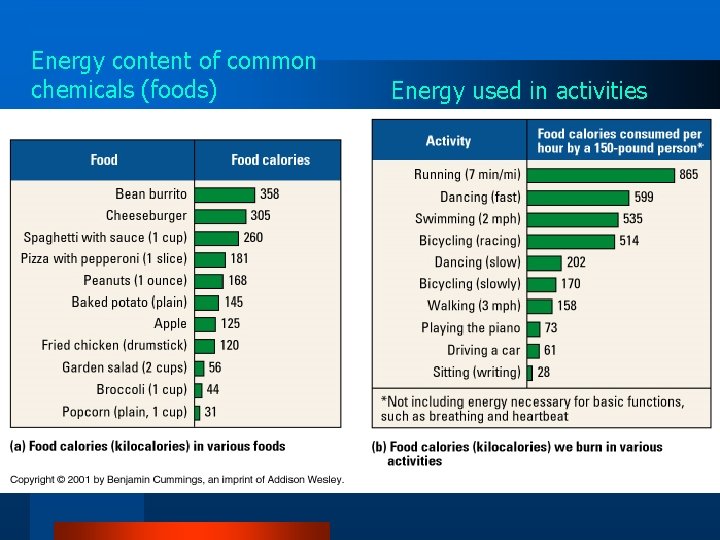
Energy content of common chemicals (foods) Energy used in activities
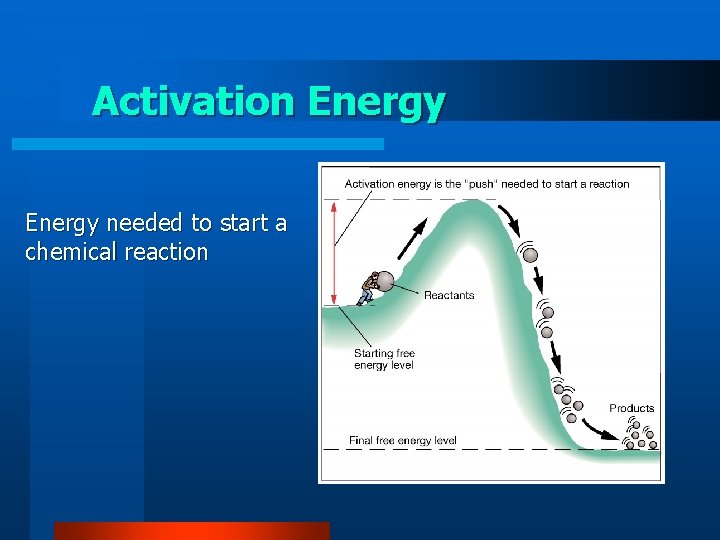
Activation Energy needed to start a chemical reaction

Enzymes • Substances that increase the speed of chemical. reactions • Are not changed in the reaction • Essential for life • Most are proteins, some are RNA.
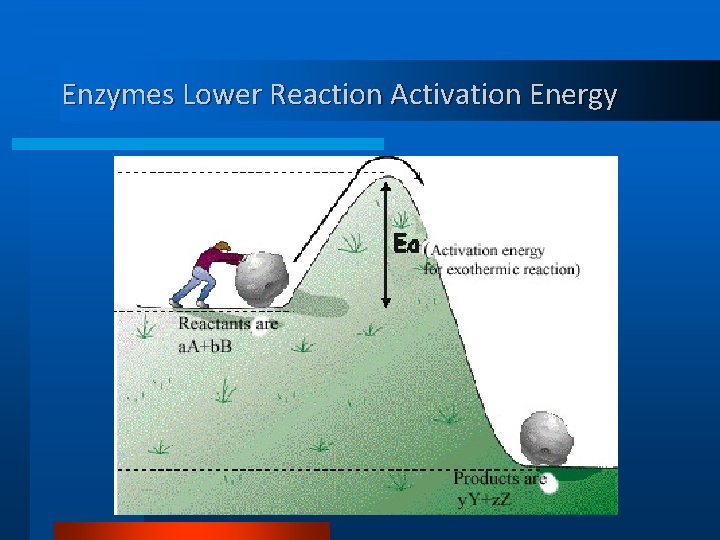
Enzymes Lower Reaction Activation Energy
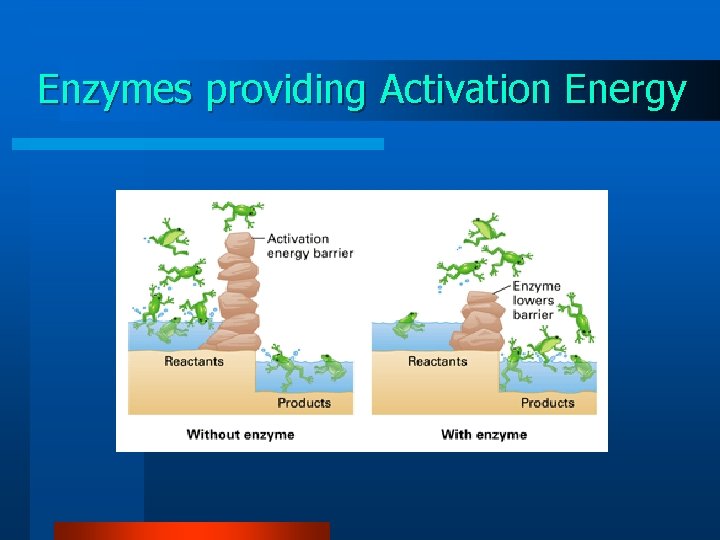
Enzymes providing Activation Energy
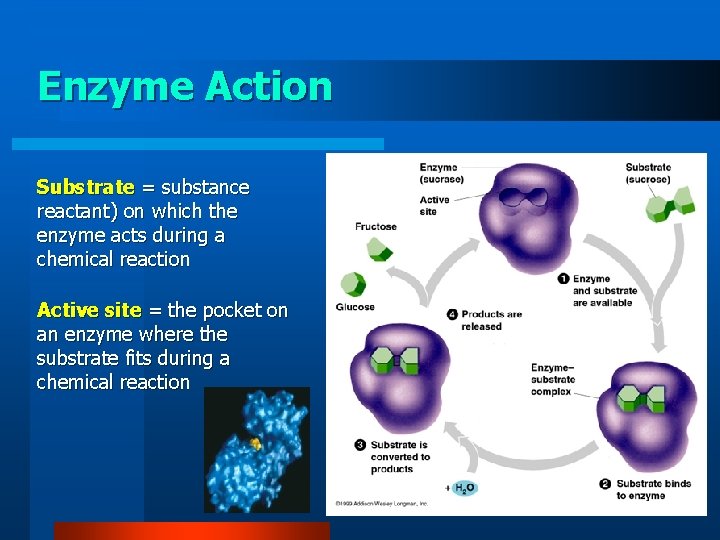
Enzyme Action Substrate = substance reactant) on which the enzyme acts during a chemical reaction Active site = the pocket on an enzyme where the substrate fits during a chemical reaction

Cellular environment affects enzyme activity l Enzyme activity is influenced by – temperature – salt concentration – p. H l Reaction rate is affected by amount of substrate

ASK QUESTIONS!!!!!
- Slides: 20