Energies Forces and Bonds Dept Phys Tunghai Univ

Energies, Forces, and Bonds Dept. Phys. , Tunghai Univ. 93學年度第二學期‧生物物理 施奇廷

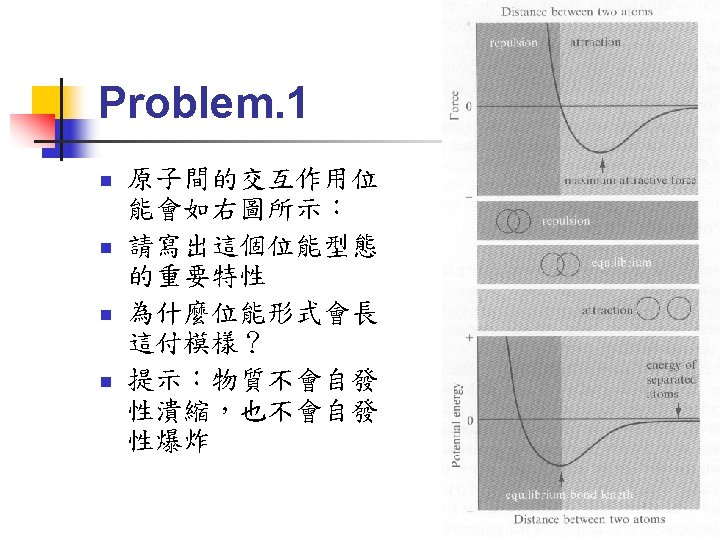
Interatomic Potentials for Strong Bonds n n n Relation between potential energy and force: For central force (only depend on the distance): The interatomic force is attractive for long distance and repulsive for short distance (see Fig. 3. 1)
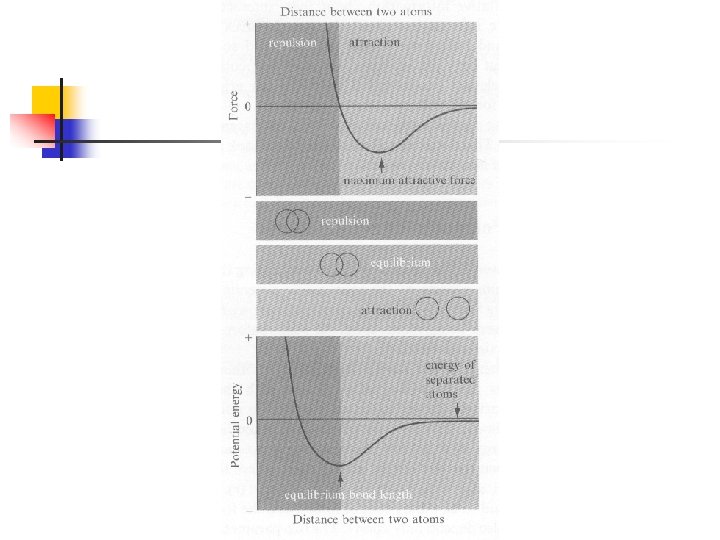

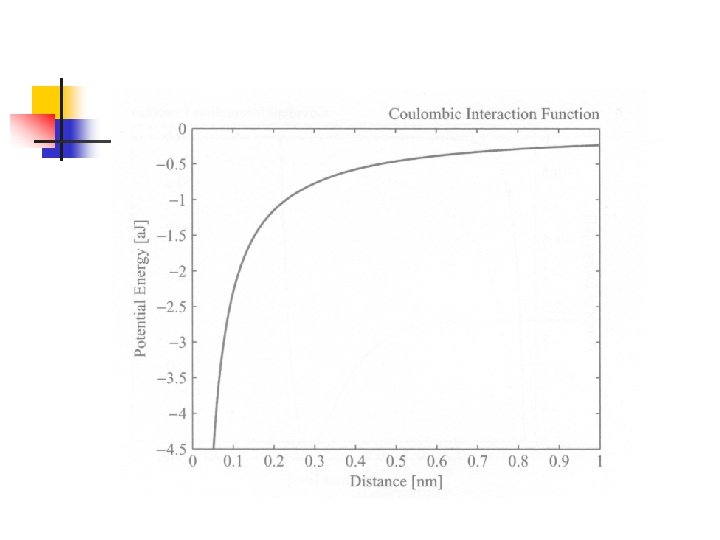
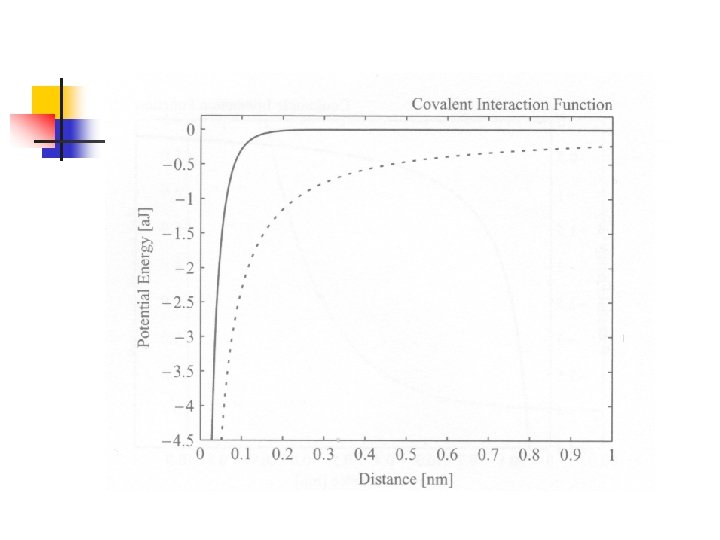
Interaction Functional Forms of Strong Bonds n 由前章所介紹的各種鍵結形式暗示我們, 分子中原子間的交互作用可能也有好幾 種形式(雖然都來自電磁交互作用): n n Closed-shell repulsion (Born-Mayer function) Coulombic interaction potential Covalent interaction potential Morse potential function

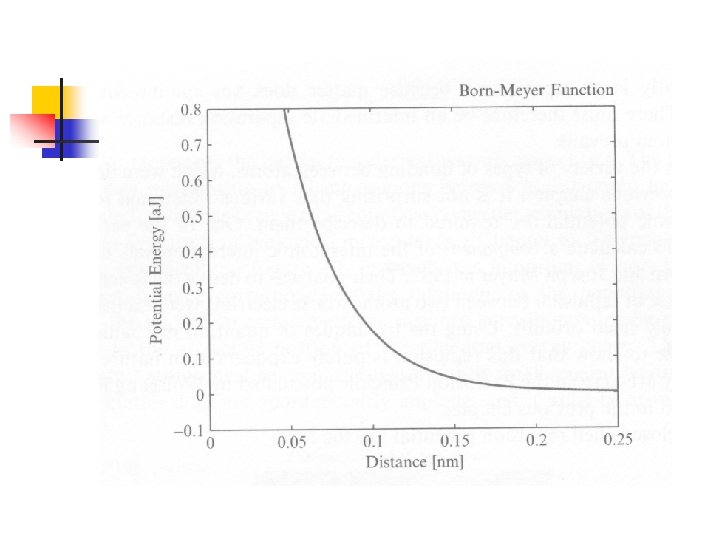
Closed-Shell Repulsion n 1932, derived by Max Born and Joseph Mayer 計算兩個原子,其外層軌域為全填滿 由於Pauli’s exclusion principle,兩者靠近 時會發生很強的排斥力:




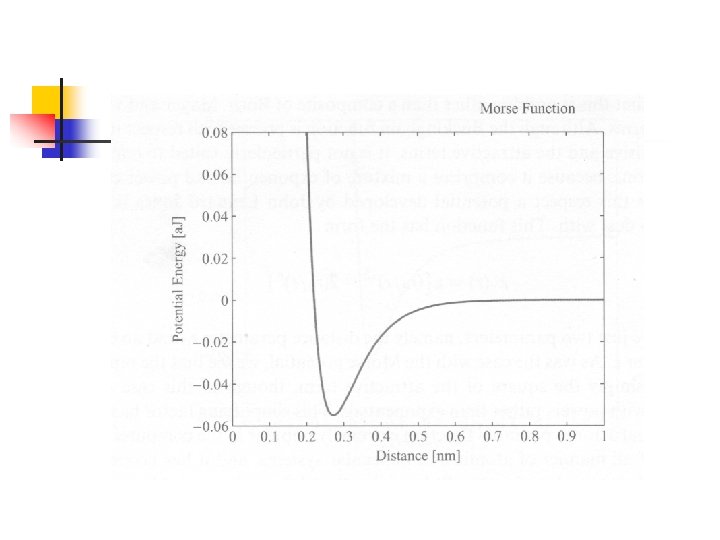
Morse Function n 金屬鍵的交互作用形式 包含Born-Mayer interaction, Coulomb interaction (repulsion of the positive ions), and the electron-electron interaction n r 0為一長度單位,滿足: n 當r=r 0時為位能之最低點,極為平衡位置 n

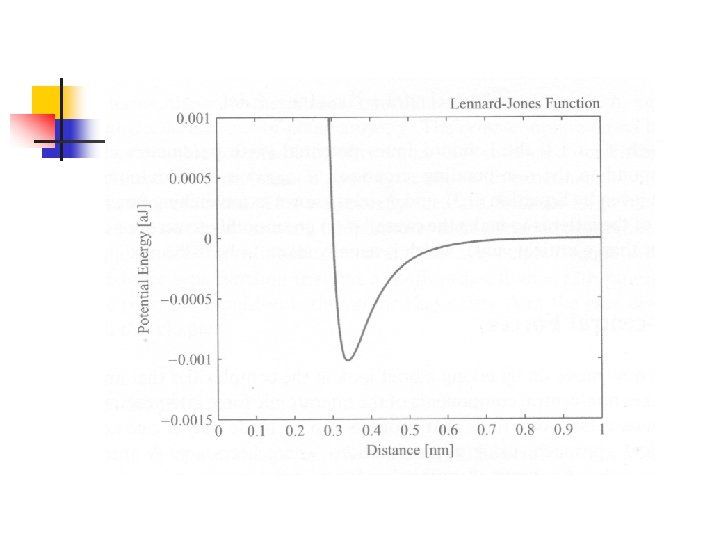
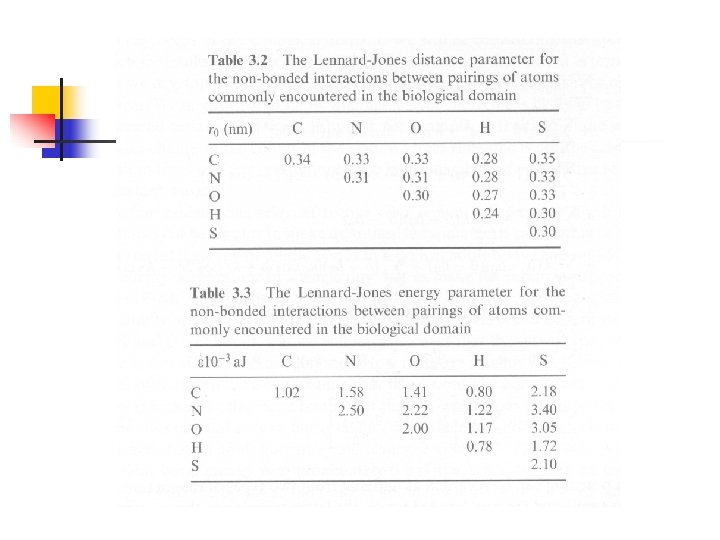
Interatomic Potentials for Weak Bonds n n Van der Waals interaction: 由於原子外圍的電 子分佈隨時改變,經常會產生微小的暫時性電 偶極(instantaneous dipole),這些電偶極會 調整到適當的方向產生吸引力,其形式為: 這項吸引力經常與Pauli exclusion principle所 引起的排斥力(Born-Mayer interaction)合併 成一個函數,稱為Buckingham potential function




Total Energy of a Molecule

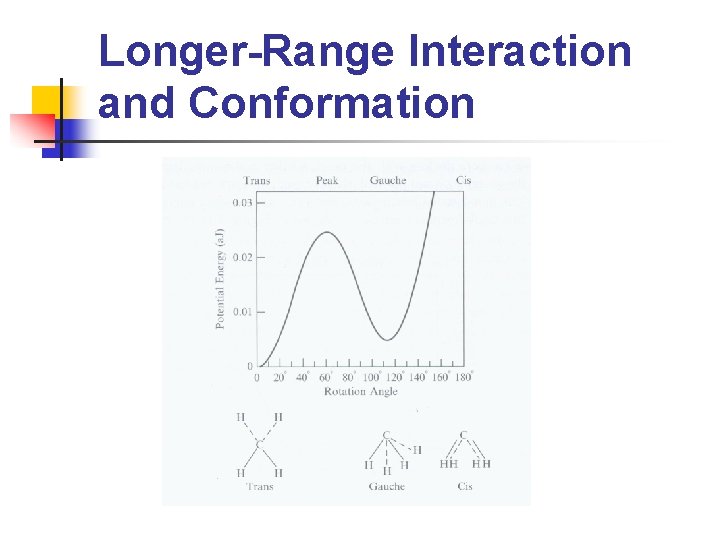
Longer-Range Interaction and Conformation


- Slides: 27