ENDMEMBER MIXING ANALYSIS PRINCIPLES AND EXAMPLES Mark Williams

END-MEMBER MIXING ANALYSIS: PRINCIPLES AND EXAMPLES Mark Williams and Fengjing Liu Department of Geography and Institute of Arctic and Alpine Research, University of Colorado, Boulder, CO 80309

EMMA ADVANTAGES • Use more tracers than components • Quantitatively evaluate potential end-members • Quantitatively evaluate results of the mixing model

PART 2: EMMA AND PCA ¡ EMMA Notation ¡ Over-Determined Situation ¡ Orthogonal Projection ¡ Notation of Mixing Spaces ¡ Steps to Perform EMMA

DEFINITION OF END-MEMBER ¡ For EMMA, we use end-members instead of components to describe water contributing to stream from various compartments and geographic areas ¡ End-members are components that have more extreme solute concentrations than streamflow [Christophersen and Hooper, 1992]

EMMA NOTATION (1) ¡ Hydrograph separations using multiple tracers simultaneously; ¡ Use more tracers than necessary to test consistency of tracers; ¡ Typically use solutes as tracers Modified from Hooper, 2001

EMMA NOTATION (2) ¡ Measure p solutes; define mixing space (S- Space) to be p-dimensional ¡ Assume that there are k linearly independent end-members (k < p) ¡ B, matrix of end-members, (k p); each row bj (1 p) ¡ X, matrix of streamflow samples, (n observations p solutes); each row xi (1 p)

PROBLEM STATEMENT ¡ Find a vector fi of mixing proportions such that ¡ Note that this equation is the same as generalized one for mixing model; the resymbolizing is for simplification and consistency with EMMA references ¡ Also note that this equation is overdetermined because k < p, e. g. , 6 solutes for 3 end-members

SOLUTION FOR OVERDETERMINED EQUATIONS ¡ Must choose objective function: minimize sum of squared error ¡ Solution is normal equation [Christophersen et al. , 1990; Hooper et al. , 1990]: ¡ Constraint: all proportions must sum to 1 ¡ Solutions may be > 1 or < 0; this issue will be elaborated later

ORTHOGONAL PROJECTIONS ¡ Following the normal equation, the predicted streamflow chemistry is [Christophersen and Hooper, 1992]: ¡ Geometrically, this is the orthogonal projection of xi into the subspace defined by B, the endmembers

This slide is from Hooper, 2001

OUR GOALS ACHIEVED SO FAR? • We measure chemistry of streamflow and end-members. • Then, we can derive fractions of end-members contributing to streamflow using equations above. • So, our goals achieved? • Not quite, because we also want to test end-members as well as mixing model. • We need to define the geometry of the solute “cloud” (S -space) and project end-members into S-space! • How? Use PCA to determine number and orientation of axes in S-space. Modified from Hooper, 2001

EMMA PROCEDURES • Identification of Conservative Tracers - Bivariate solute-solute plots to screen data; • PCA Performance - Derive eigenvalues and eigenvectors; • Orthogonal Projection - Use eigenvectors to project chemistry of streamflow and end-members; • Screen End-Members - Calculate Euclidean distance of endmembers between their original values and S-space projections; • Hydrograph Separation - Use orthogonal projections and generalized equations for mixing model to get solutions! • Validation of Mixing Model - Predict streamflow chemistry using results of hydrograph separation and original end-member concentrations.
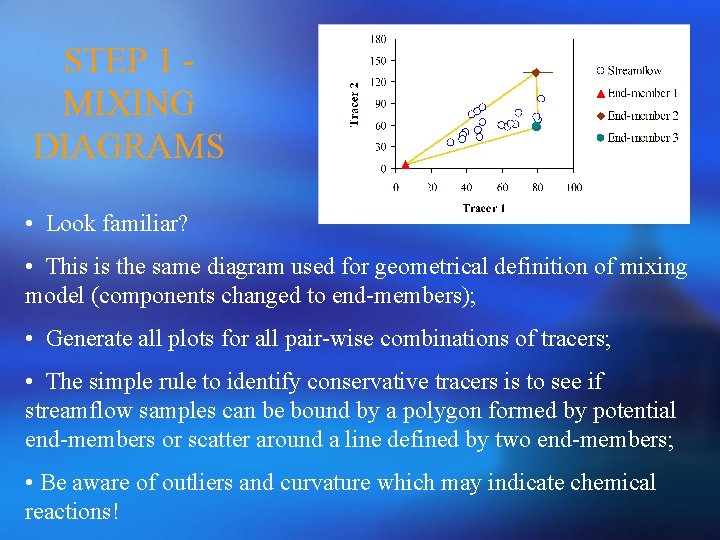
STEP 1 MIXING DIAGRAMS • Look familiar? • This is the same diagram used for geometrical definition of mixing model (components changed to end-members); • Generate all plots for all pair-wise combinations of tracers; • The simple rule to identify conservative tracers is to see if streamflow samples can be bound by a polygon formed by potential end-members or scatter around a line defined by two end-members; • Be aware of outliers and curvature which may indicate chemical reactions!

STEP 2 - PCA PERFORMANCE • For most cases, if not all, we should use correlation matrix rather than covariance matrix of conservative solutes in streamflow to derive eigenvalues and eigenvectors; • Why? This treats each variable equally important and unitless; • How? Standardize the original data set using a routine software or minus mean and then divided by standard deviation; • To make sure if you are doing right, the mean should be zero and variance should be 1 after standardized!

APPLICATION OF EIGENVALUES • Eigenvalues can be used to infer the number of end-members that should be used in EMMA. How? • Sum up all eigenvalues; • Calculate percentage of each eigenvalue in the total eigenvalue; • The percentage should decrease from PCA component 1 to p (remember p is the number of solutes used in PCA); • How many eigenvalues can be added up to 90% (somewhat subjective! No objective criteria for this!)? Let this number be m, which means the number of PCA components should be retained (sometimes called # of mixing spaces); • (m +1) is equal to # of end-members we use in EMMA.

STEP 3 - ORTHOGONAL PROJECTION • X - Standardized data set of streamflow, (n p); • V - Eigenvectors from PCA, (m p); Remember only the first m eigenvectors to be used here! Project End-Members • Use the same equation above; • Now X represents a vector (1 p) for each end-member; • Remember X here should be standardized by subtracting streamflow mean and dividing by streamflow standard deviation!

STEP 4 - SCREEN END-MEMEBRS Geometrically • Plot a scatter plot for streamflow samples and end-members using the first and second PCA projections; • Eligible end-members should be vertices of a polygon (a line if m = 1, a triangle if m = 2, and a quadrilateral if m = 3) and should bind streamflow samples in a convex sense; Algebraically • Calculate the Euclidean distance between original chemistry and projections for each solute using the equations below: • j represent each solute and bj is the original solute value Those steps should lead to identification of eligible end-members!

STEP 5 - HYDROGRAPH SEPARATION • Use the retained PCA projections from streamflow and endmembers to derive flowpath solutions! • So, mathematically, this is the same as a general mixing model rather than the over-determined situation.

STEP 6 - PREDICTION OF STREAMFLOW CHEMISTRY • Multiply results of hydrograph separation (usually fractions) by original solute concentrations of end-members to reproduce streamflow chemistry for conservative solutes; • Comparison of the prediction with the observation can lead to a test of mixing model.

PROBLEM ON OUTLIERS • PCA is very sensitive to outliers; • If any outliers are found in the mixing diagrams of PCA projections, check if there are physical reasons; • Outliers have negative or > 1 fractions; • See next slide how to resolve outliers using a geometrical approach for an end-member model.
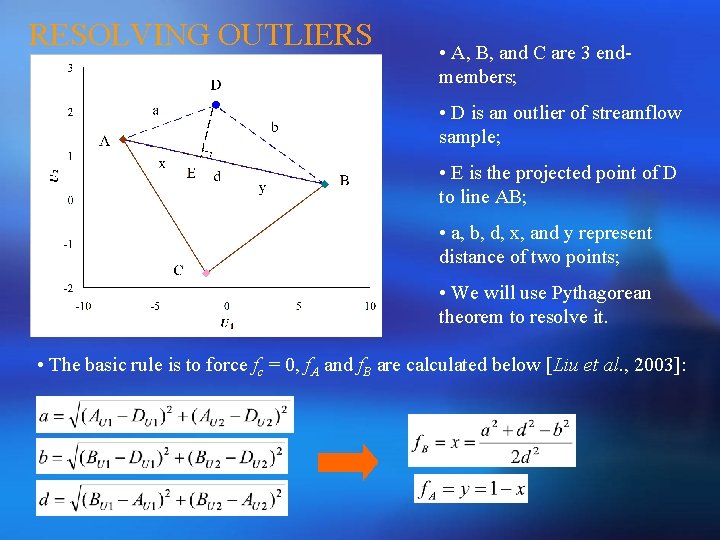
RESOLVING OUTLIERS • A, B, and C are 3 endmembers; • D is an outlier of streamflow sample; • E is the projected point of D to line AB; • a, b, d, x, and y represent distance of two points; • We will use Pythagorean theorem to resolve it. • The basic rule is to force fc = 0, f. A and f. B are calculated below [Liu et al. , 2003]:

APPLICATION IN GREEN LAKES VALLEY: RESEARCH SITE Green Lake 4 Sample Collection • Stream water weekly grab samples • Snowmelt - snow lysimeter • Soil water - zero tension lysimeter • Talus water – biweekly to monthly Sample Analysis • Delta 18 O and major solutes
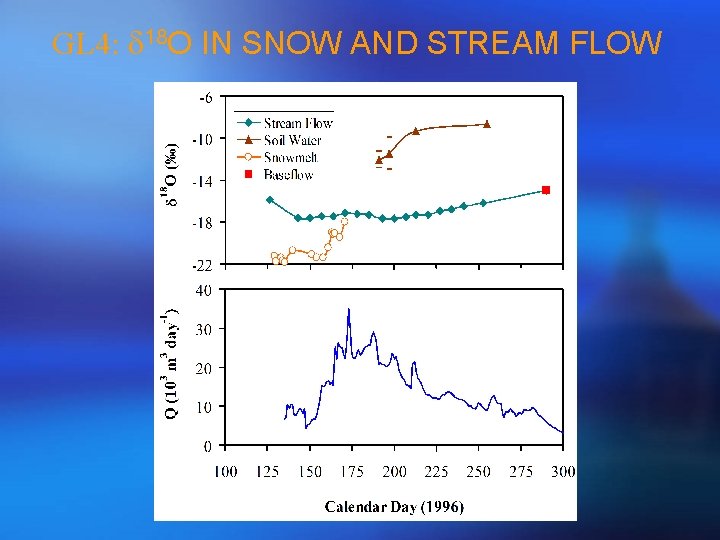
GL 4: d 18 O IN SNOW AND STREAM FLOW
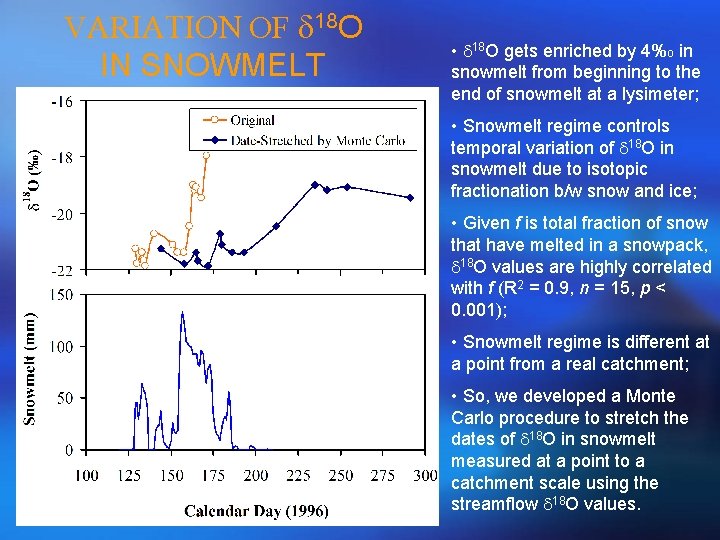
VARIATION OF d 18 O IN SNOWMELT • d 18 O gets enriched by 4%o in snowmelt from beginning to the end of snowmelt at a lysimeter; • Snowmelt regime controls temporal variation of d 18 O in snowmelt due to isotopic fractionation b/w snow and ice; • Given f is total fraction of snow that have melted in a snowpack, d 18 O values are highly correlated with f (R 2 = 0. 9, n = 15, p < 0. 001); • Snowmelt regime is different at a point from a real catchment; • So, we developed a Monte Carlo procedure to stretch the dates of d 18 O in snowmelt measured at a point to a catchment scale using the streamflow d 18 O values.
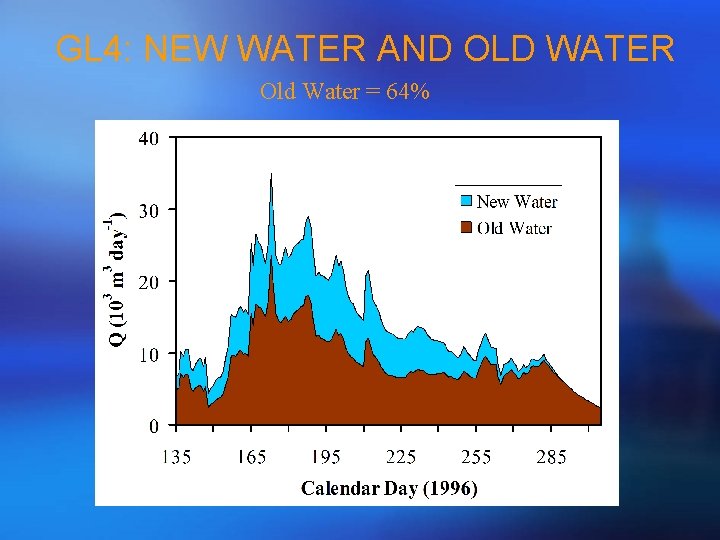
GL 4: NEW WATER AND OLD WATER Old Water = 64%
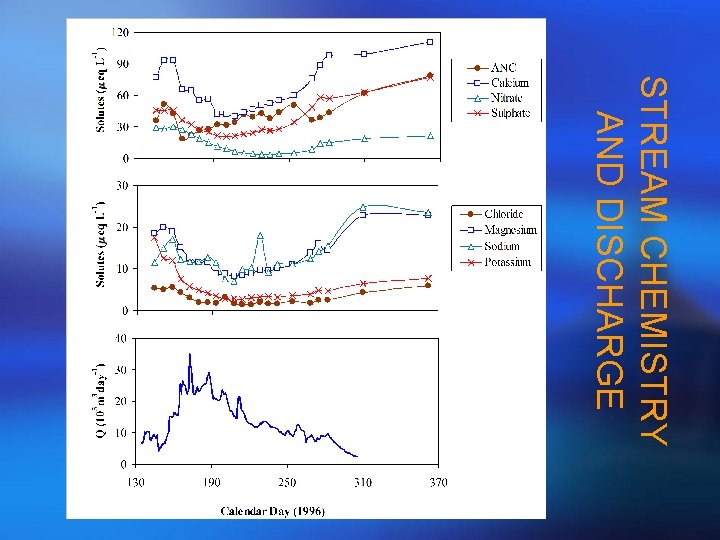
STREAM CHEMISTRY AND DISCHARGE
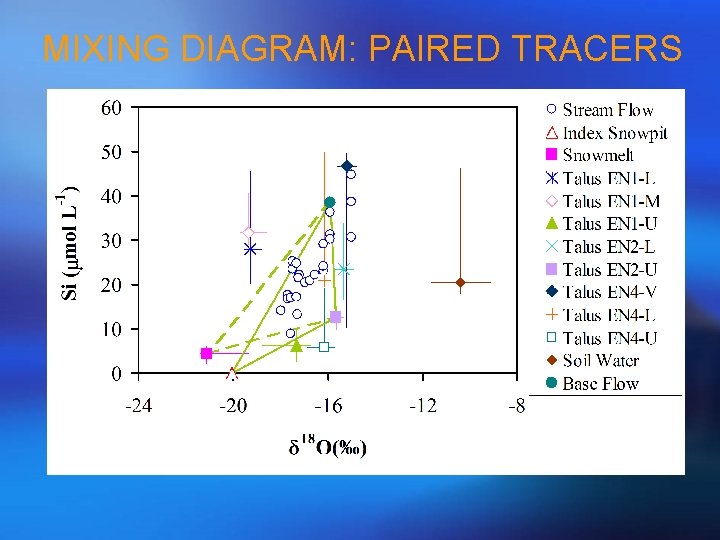
MIXING DIAGRAM: PAIRED TRACERS
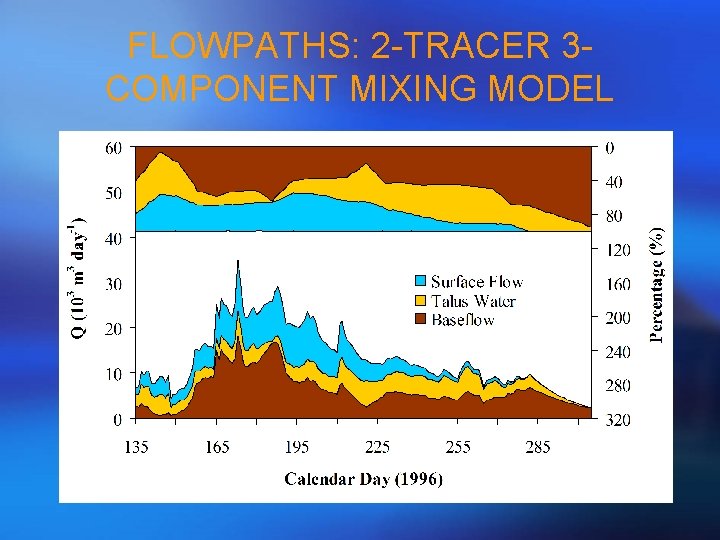
FLOWPATHS: 2 -TRACER 3 COMPONENT MIXING MODEL
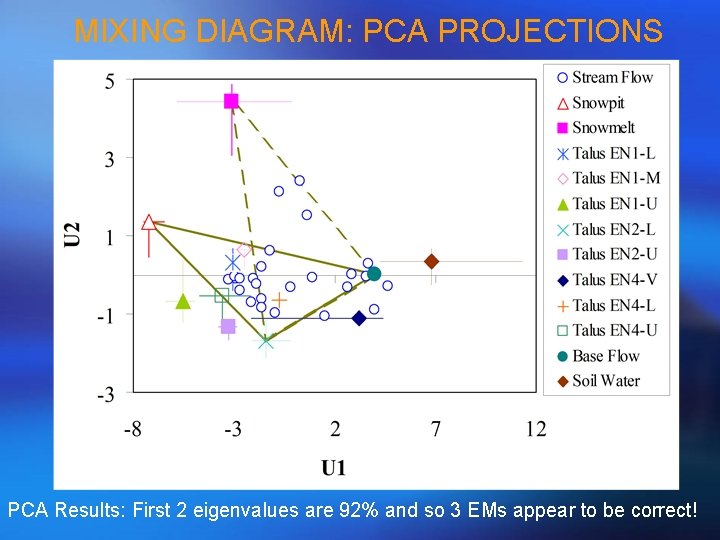
MIXING DIAGRAM: PCA PROJECTIONS PCA Results: First 2 eigenvalues are 92% and so 3 EMs appear to be correct!
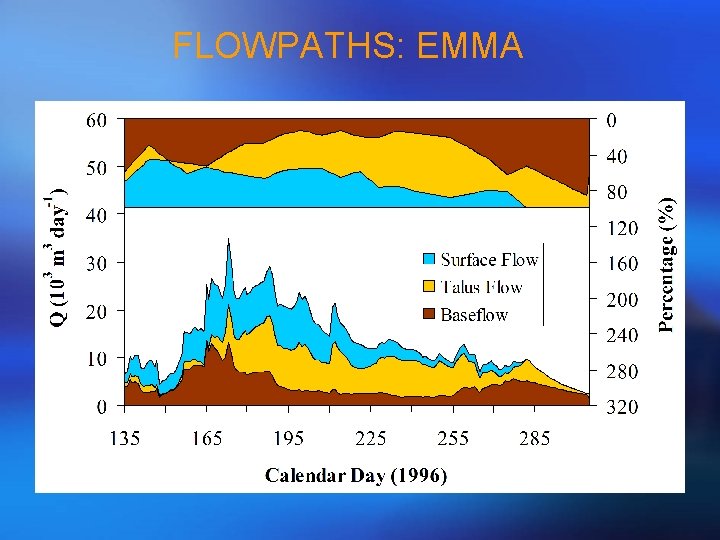
FLOWPATHS: EMMA
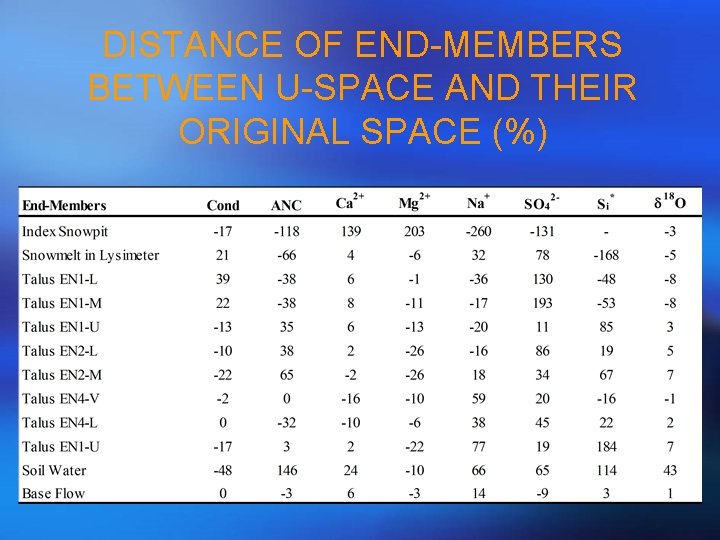
DISTANCE OF END-MEMBERS BETWEEN U-SPACE AND THEIR ORIGINAL SPACE (%)
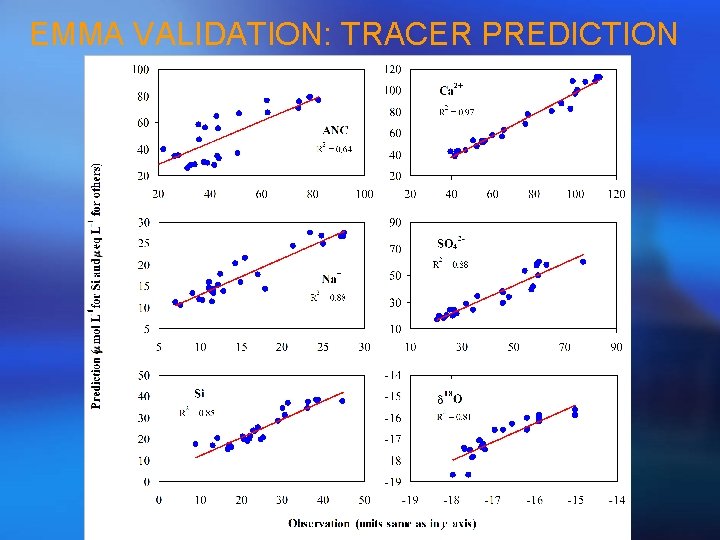
EMMA VALIDATION: TRACER PREDICTION

LEADVILLE CASE STUDY ¡ Rich mining legacy ¡ Superfund site: over $100 M so far ¡ Complicated hydrology: Mine shafts Faults Drainage tunnels We know nothing about mountain groundwater! ¡ What are water sources to drainage tunnel? ¡ Complicated, rigorous test
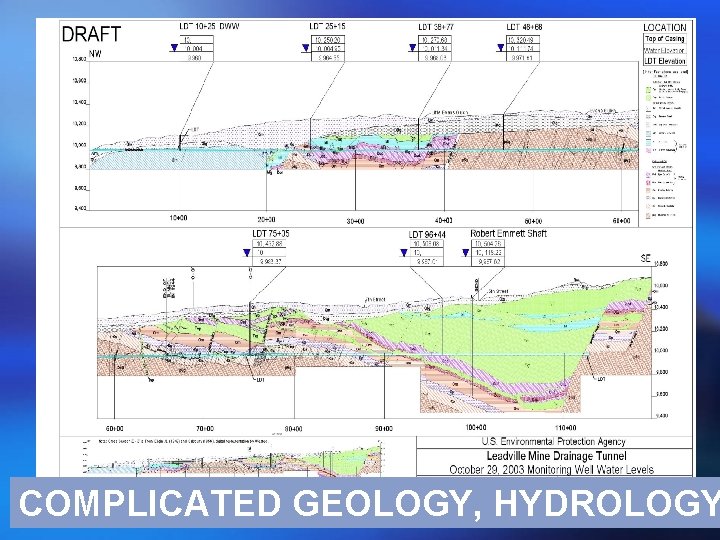
COMPLICATED GEOLOGY, HYDROLOGY
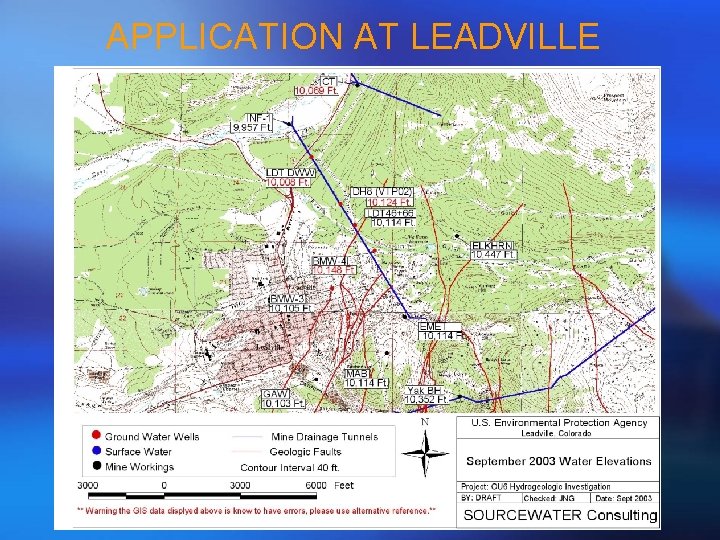
APPLICATION AT LEADVILLE
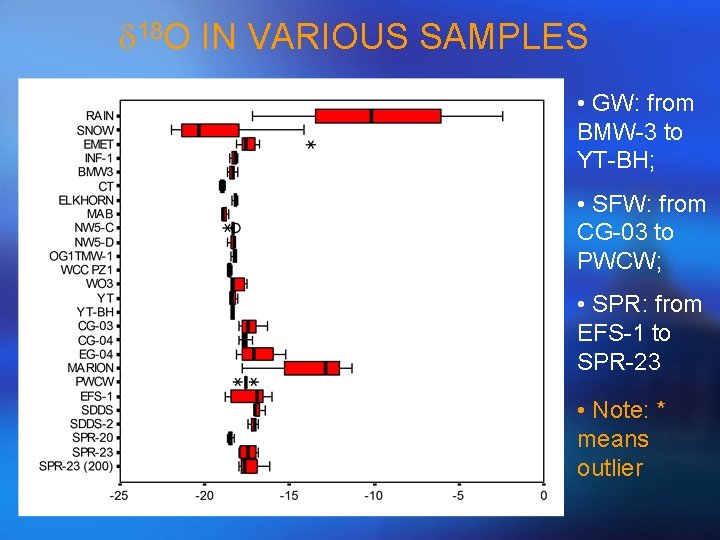
d 18 O IN VARIOUS SAMPLES • GW: from BMW-3 to YT-BH; • SFW: from CG-03 to PWCW; • SPR: from EFS-1 to SPR-23 • Note: * means outlier
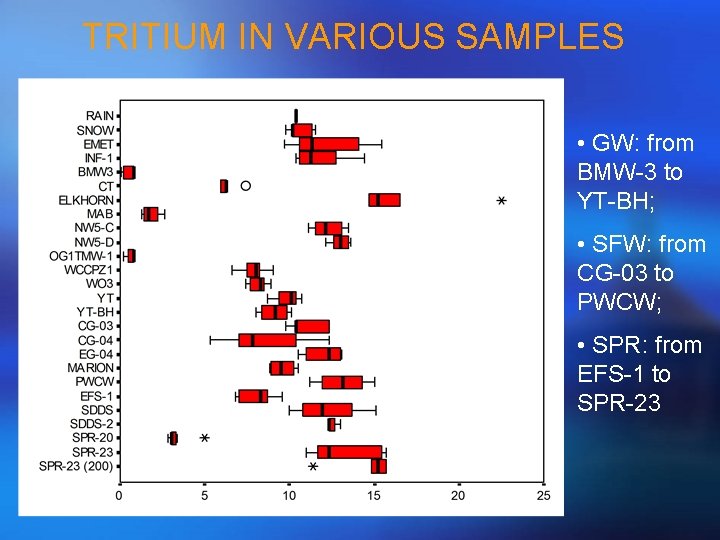
TRITIUM IN VARIOUS SAMPLES • GW: from BMW-3 to YT-BH; • SFW: from CG-03 to PWCW; • SPR: from EFS-1 to SPR-23
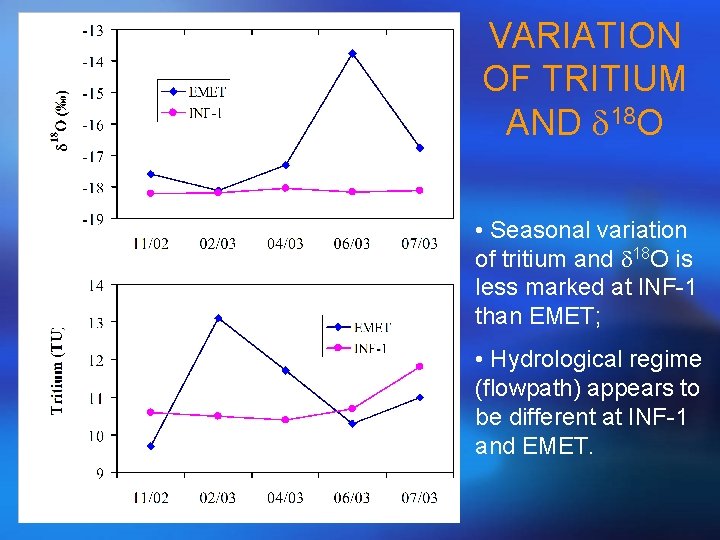
VARIATION OF TRITIUM AND d 18 O • Seasonal variation of tritium and d 18 O is less marked at INF-1 than EMET; • Hydrological regime (flowpath) appears to be different at INF-1 and EMET.
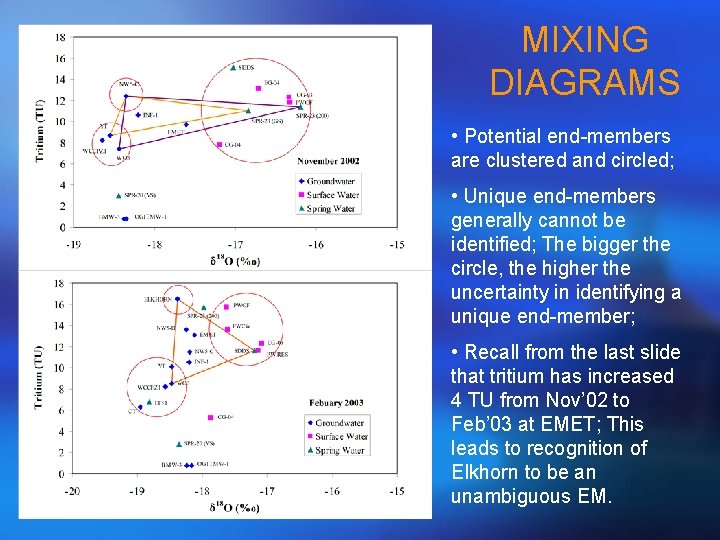
MIXING DIAGRAMS • Potential end-members are clustered and circled; • Unique end-members generally cannot be identified; The bigger the circle, the higher the uncertainty in identifying a unique end-member; • Recall from the last slide that tritium has increased 4 TU from Nov’ 02 to Feb’ 03 at EMET; This leads to recognition of Elkhorn to be an unambiguous EM.
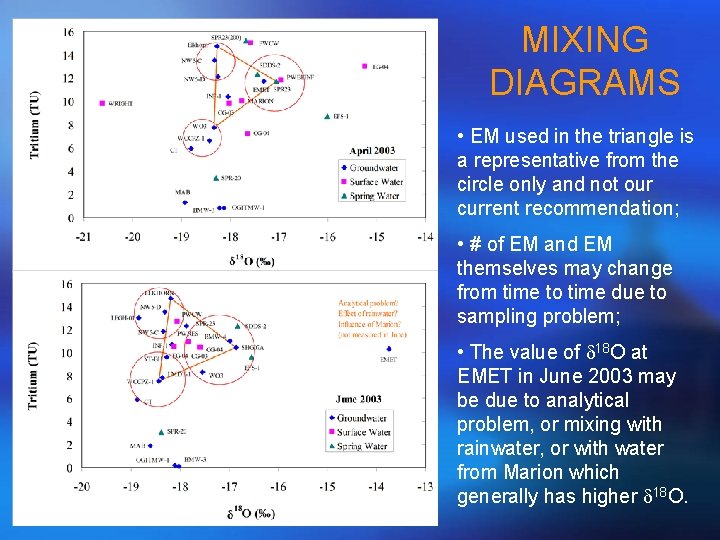
MIXING DIAGRAMS • EM used in the triangle is a representative from the circle only and not our current recommendation; • # of EM and EM themselves may change from time to time due to sampling problem; • The value of d 18 O at EMET in June 2003 may be due to analytical problem, or mixing with rainwater, or with water from Marion which generally has higher d 18 O.
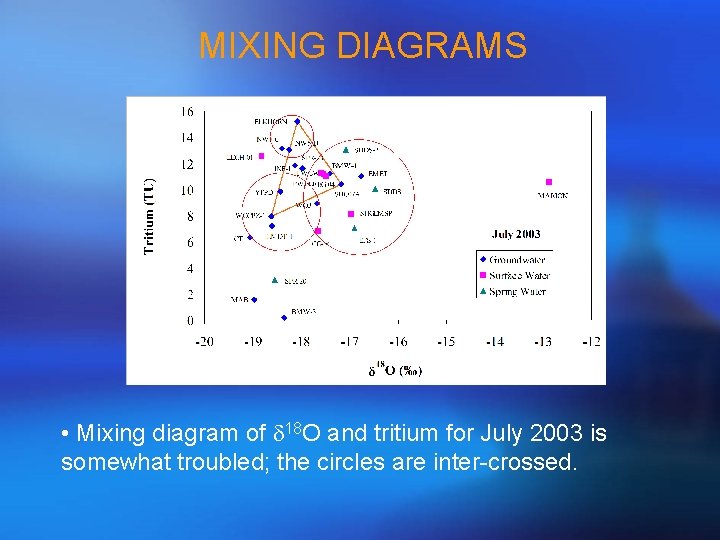
MIXING DIAGRAMS • Mixing diagram of d 18 O and tritium for July 2003 is somewhat troubled; the circles are inter-crossed.

SUMMARY FOR MIXING DIAGRAMS OF TRITIUM AND d 18 O • EMs may change from time to time within a water year; • Except for Elkhorn, unique EMs cannot be identified at this time; • However, EM clusters are usually consistent from time to time; • One cluster includes: WO 3, CT, YT, and WCCPZ-1; • The other cluster generally includes: SPR-23, PWBEINF, SDDS-2, SHG 07 A, EFS-1, BMW-4, CG-03, CG-04; • Particularly, some EMs could be excluded from a potential EM list: OG 1 TMW-1, BMW-3, MAB, and SPR-20.
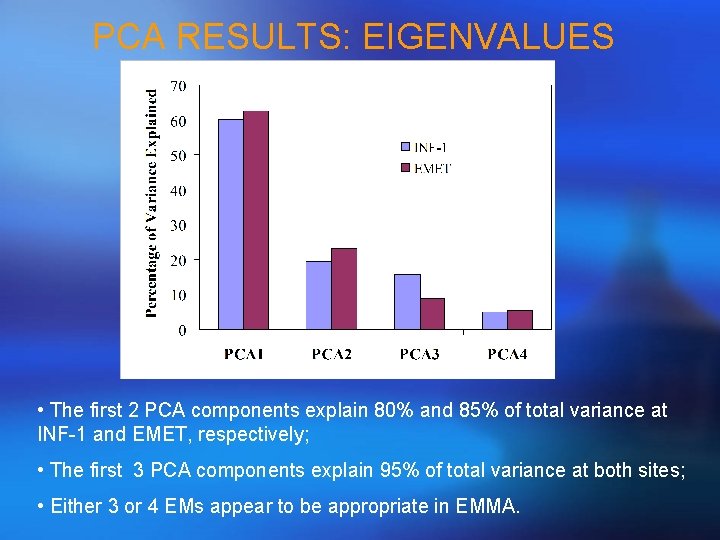
PCA RESULTS: EIGENVALUES • The first 2 PCA components explain 80% and 85% of total variance at INF-1 and EMET, respectively; • The first 3 PCA components explain 95% of total variance at both sites; • Either 3 or 4 EMs appear to be appropriate in EMMA.
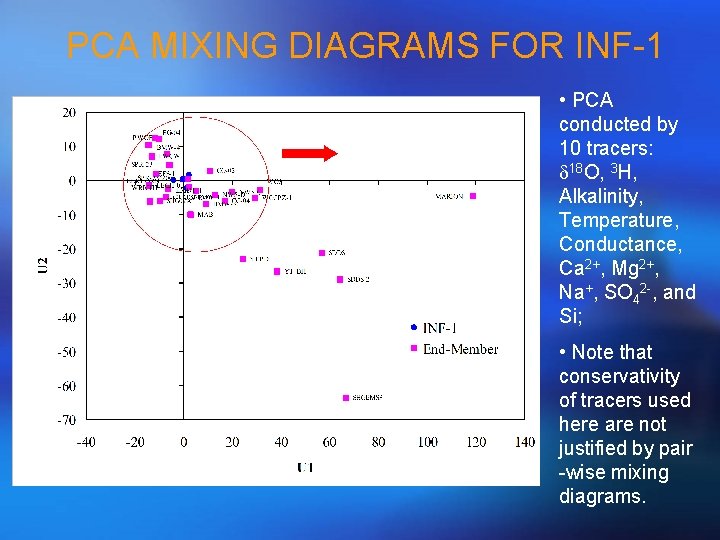
PCA MIXING DIAGRAMS FOR INF-1 • PCA conducted by 10 tracers: d 18 O, 3 H, Alkalinity, Temperature, Conductance, Ca 2+, Mg 2+, Na+, SO 42 -, and Si; • Note that conservativity of tracers used here are not justified by pair -wise mixing diagrams.
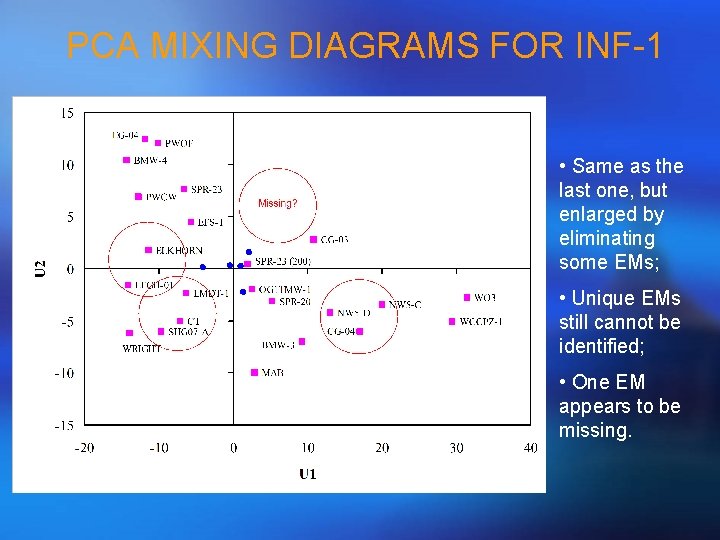
PCA MIXING DIAGRAMS FOR INF-1 • Same as the last one, but enlarged by eliminating some EMs; • Unique EMs still cannot be identified; • One EM appears to be missing.
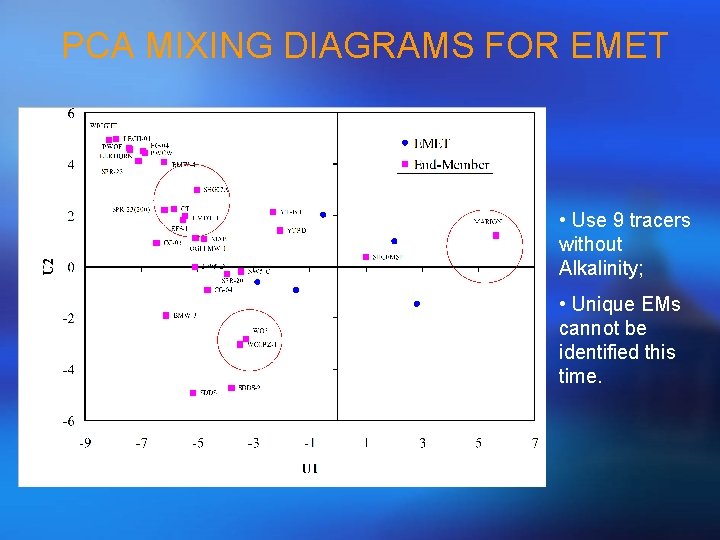
PCA MIXING DIAGRAMS FOR EMET • Use 9 tracers without Alkalinity; • Unique EMs cannot be identified this time.

SUMMARY FOR PCA AND EMMA • Unique EMs cannot be identified at this time; • However, some potential end-members are consistent with the mixing diagrams of tritium and d 18 O such as Elkhorn, CT, and CG-03; • Future work is needed to plot mixing diagrams for all tracers so that non-conservative tracers can be eliminated;

IMPLICATION FOR FUTURE SAMPLING SCHEME • Monthly or bi-monthly sampling scheme does capture seasonal signal within a water year; • But this scheme may miss temporal variation within all seasons; • Hydrological regime may change from season to season and within seasons; • So, temporally intensive sampling scheme may be needed to capture within-season variation in order to unanimously identify EMs using EMMA.

SUMMARY: MIXING MODEL VS EMMA General Mixing Model ¡ Easy to understand manipulate! ¡ Doable with limited measurements of solutes! ¡ But different tracers may yield different results! EMMA ¡ Use more tracers than necessary to lead to consistent results; ¡ Provide a framework for analyzing watershed chemical data sets; ¡ Generate testable hypotheses that focus future field efforts!

REDERENCES ¡ Hooper, R. , 2001, http: //www. cof. orst. edu/cof/fe/watershed/shortcourse/schedule. htm ¡ Christophersen, N. , C. Neal, R. P. Hooper, R. D. Vogt, and S. Andersen, Modeling stream water chemistry as a mixture of soil water end-members – a step towards second-generation acidification models, Journal of Hydrology, 116, 307 -320, 1990. ¡ Christophersen, N. and R. P. Hooper, Multivariate analysis of stream water chemical data: the use of principal components analysis for the endmember mixing problem, Water Resources Research, 28(1), 99 -107, 1992. ¡ Hooper, R. P. , N. Christophersen, and N. E. Peters, Modeling stream water chemistry as a mixture of soil water end-members – an application to the Panola mountain catchment, Georgia, U. S. A. , Journal of Hydrology, 116, 321 -343, 1990. ¡ Liu, F. , M. Williams, and N. Caine, in review, Source waters and flowpaths in a seasonally snow-covered catchment, Colorado Front Range, USA, Water Resources Research, 2003.
- Slides: 50