Encuentro con el experto Trombosis Modelos predictivos de

Encuentro con el experto: Trombosis Modelos predictivos de riesgo de trombosis en el paciente oncológico Anna Falanga, MD Director Department of Immunohematology and Transfusion Medicine. Director Hemostasis and Thrombosis Center. Hospital Papa Giovanni XXIII. Bergamo, Italy
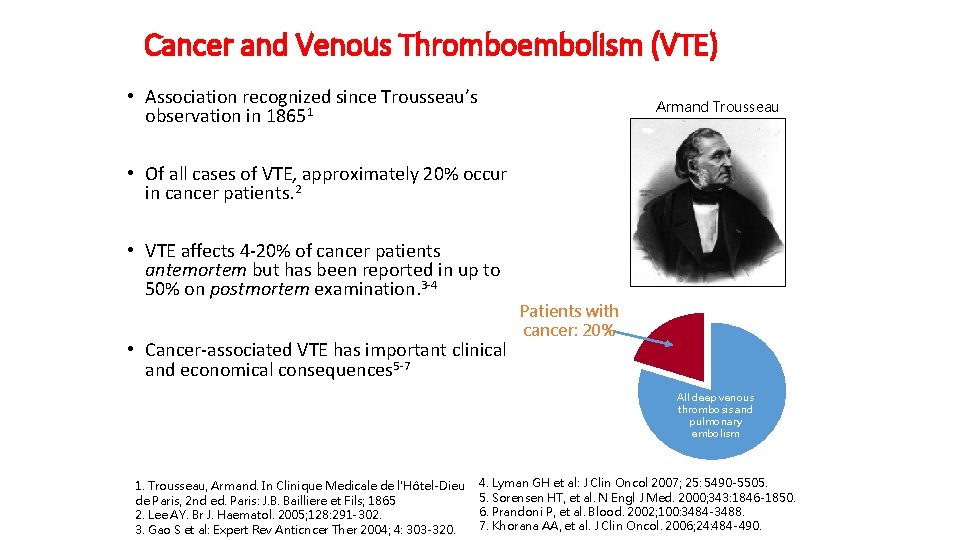
Cancer and Venous Thromboembolism (VTE) • Association recognized since Trousseau’s observation in 18651 Armand Trousseau • Of all cases of VTE, approximately 20% occur in cancer patients. 2 • VTE affects 4 -20% of cancer patients antemortem but has been reported in up to 50% on postmortem examination. 3 -4 • Cancer-associated VTE has important clinical and economical consequences 5 -7 Patients with cancer: 20% All deep venous thrombosis and pulmonary embolism 1. Trousseau, Armand. In Clinique Medicale de l'Hôtel-Dieu de Paris, 2 nd ed. Paris: J. B. Bailliere et Fils; 1865 2. Lee AY. Br J. Haematol. 2005; 128: 291 -302. 3. Gao S et al: Expert Rev Anticncer Ther 2004; 4: 303 -320. 4. Lyman GH et al: J Clin Oncol 2007; 25: 5490 -5505. 5. Sorensen HT, et al. N Engl J Med. 2000; 343: 1846 -1850. 6. Prandoni P, et al. Blood. 2002; 100: 3484 -3488. 7. Khorana AA, et al. J Clin Oncol. 2006; 24: 484 -490.
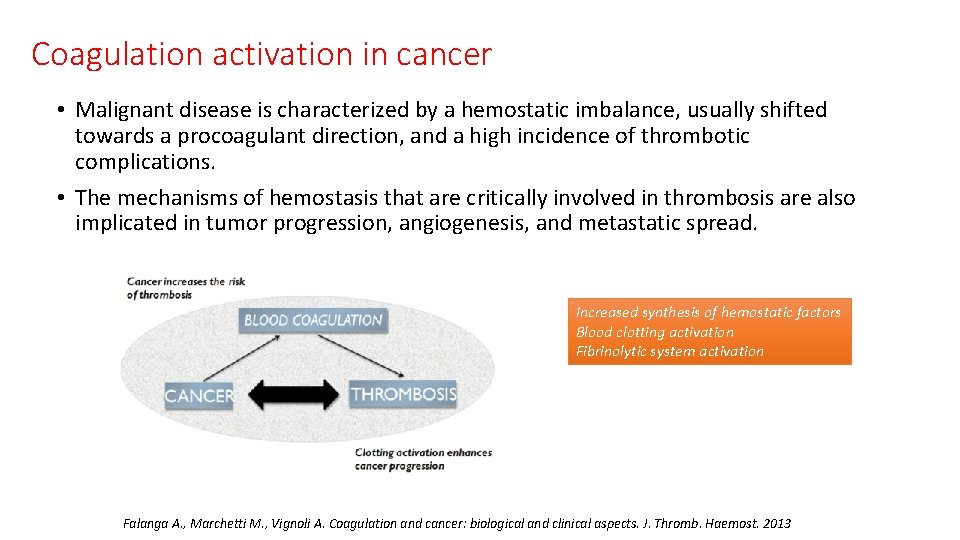
Coagulation activation in cancer • Malignant disease is characterized by a hemostatic imbalance, usually shifted towards a procoagulant direction, and a high incidence of thrombotic complications. • The mechanisms of hemostasis that are critically involved in thrombosis are also implicated in tumor progression, angiogenesis, and metastatic spread. Increased synthesis of hemostatic factors Blood clotting activation Fibrinolytic system activation Falanga A. , Marchetti M. , Vignoli A. Coagulation and cancer: biological and clinical aspects. J. Thromb. Haemost. 2013
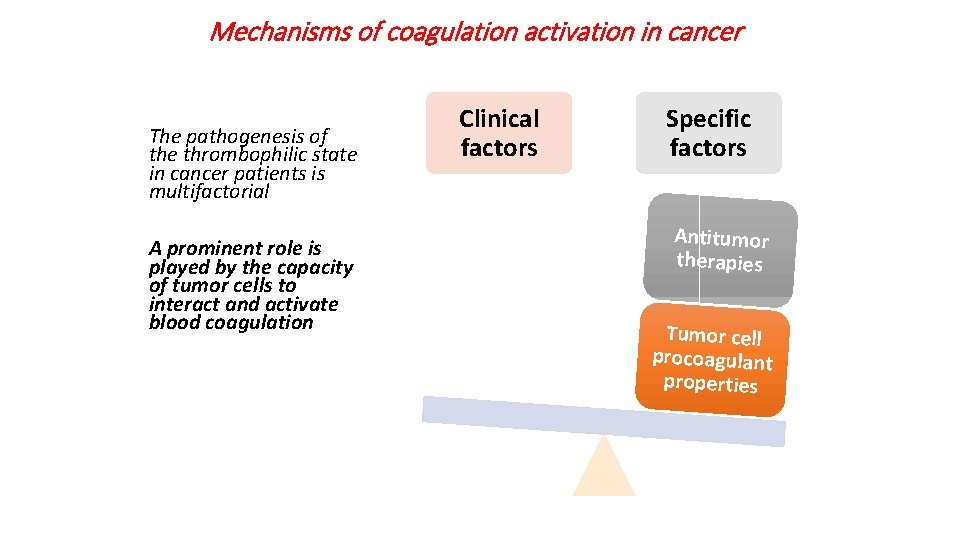
Mechanisms of coagulation activation in cancer The pathogenesis of the thrombophilic state in cancer patients is multifactorial A prominent role is played by the capacity of tumor cells to interact and activate blood coagulation Clinical factors Specific factors Antitumor therapies Tumor cell procoagulant properties

The Hypercoagulable state of Patients with cancer: A subclinical clotting activation determines • Patients with cancer present with a hypercoagulable state. • This is a condition characterized by the alterations of laboratory thrombotic markers, without overt thrombosis. • These clotting abnormalities increase with: • The start of chemotherapies • The progression of cancer
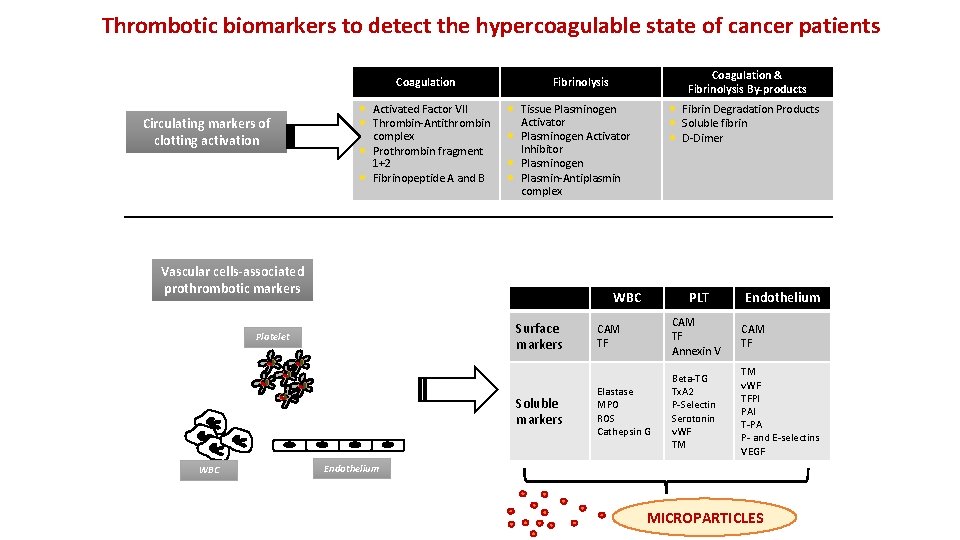
Thrombotic biomarkers to detect the hypercoagulable state of cancer patients Coagulation Circulating markers of clotting activation • • Activated Factor VII Thrombin-Antithrombin complex Prothrombin fragment 1+2 Fibrinopeptide A and B • • Tissue Plasminogen Activator Inhibitor Plasminogen Plasmin-Antiplasmin complex Vascular cells-associated prothrombotic markers WBC Surface markers Platelet Soluble markers WBC Coagulation & Fibrinolysis By-products Fibrinolysis Fibrin Degradation Products Soluble fibrin D-Dimer PLT Endothelium CAM TF Annexin V CAM TF Elastase MPO ROS Cathepsin G Beta-TG Tx. A 2 P-Selectin Serotonin v. WF TM TM v. WF TFPI PAI T-PA P- and E-selectins VEGF Endothelium MICROPARTICLES

Predictive value and diagnosis of VTE D-Dimer • Prospective studies have shown that D-dimer levels are predictive of VTE recurrence in patients without cancer. • Several studies investigate whether thrombotic markers, in particular D-dimer, are predictive of a first VTE event in patients with cancer. • High D-dimer levels are associated with VTE in several studies.
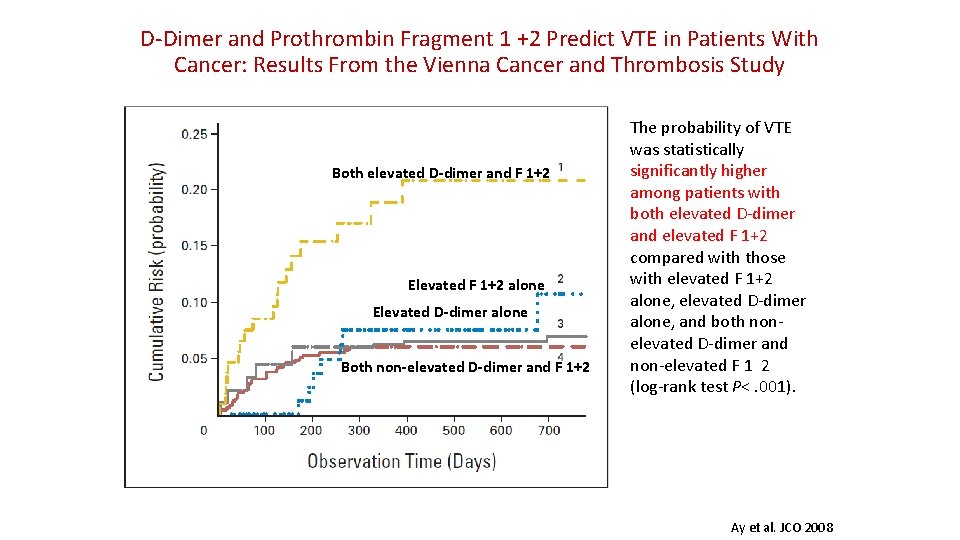
D-Dimer and Prothrombin Fragment 1 +2 Predict VTE in Patients With Cancer: Results From the Vienna Cancer and Thrombosis Study Both elevated D-dimer and F 1+2 Elevated F 1+2 alone Elevated D-dimer alone Both non-elevated D-dimer and F 1+2 The probability of VTE was statistically significantly higher among patients with both elevated D-dimer and elevated F 1+2 compared with those with elevated F 1+2 alone, elevated D-dimer alone, and both nonelevated D-dimer and non-elevated F 1 2 (log-rank test P<. 001). Ay et al. JCO 2008

Incidence of venous thromboembolism and the role of D-dimer as predictive marker in patients with advanced gastric cancer receiving chemotherapy: a prospective study • All consecutive chemotherapy-naïve patients with advanced gastric cancer who would receive palliative chemotherapy between November 2009 and April 2012 were recruited. • Their pre-treatment clinical and laboratory variables, including D-dimer, were recorded. • In multivariate analysis, pre-treatment D-dimer level was the only marginally significant risk factor for VTE development. • Considering the usefulness of D-dimer as a biomarker such as its ease of use and low cost, pretreatment D-dimer might be used as a risk stratification factor for VTE and in patient selection for thromboprophylaxis. Park et al. World J Gastrointest Oncol 2017

THE IMPORTANCE OF IDENTIFYING «HIGH RISK» PATIENTS in Ambulatory Cancer patients candidate to Primary thromboprophylaxis to prevent thrombosis during chemotherapy BACKGROUND

Thromboprophylaxis There is evidence that VTE occurs significantly in the outpatient setting. However, primary thromboprophylaxis is not indicated in all ambulatory cancer patients

Research Evidence • Large RCTs have been conducted to evaluate the impact of thromboprophylaxis with LMWH in ambulatory patients with solid tumours receiving systemic chemotherapy • These trials gave positive results in terms of efficacy and safety • However they did not impact very much on clinical practice due to the relatively high number of patients needed to treat and the fear of bleeding
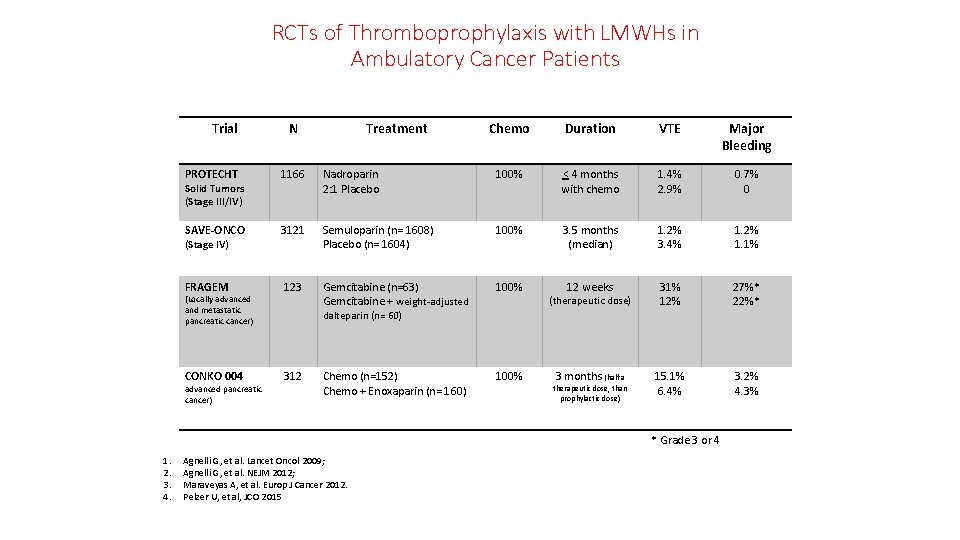
RCTs of Thromboprophylaxis with LMWHs in Ambulatory Cancer Patients Trial N Treatment Chemo Duration VTE Major Bleeding PROTECHT 1166 Nadroparin 2: 1 Placebo 100% < 4 months with chemo 1. 4% 2. 9% 0. 7% 0 SAVE-ONCO 3121 Semuloparin (n= 1608) Placebo (n= 1604) 100% 3. 5 months (median) 1. 2% 3. 4% 1. 2% 1. 1% FRAGEM 123 Gemcitabine (n=63) Gemcitabine + weight-adjusted 100% 12 weeks (therapeutic dose) 31% 12% 27%* 22%* Chemo (n=152) Chemo + Enoxaparin (n= 160) 100% 3 months (half a therapeutic dose, than prophylactic dose) 15. 1% 6. 4% 3. 2% 4. 3% Solid Tumors (Stage III/IV) (Stage IV) (Locally advanced and metastatic pancreatic cancer) CONKO 004 advanced pancreatic cancer) dalteparin (n= 60) 312 * Grade 3 or 4 1. 2. 3. 4. Agnelli G, et al. Lancet Oncol 2009; Agnelli G, et al. NEJM 2012; Maraveyas A, et al. Europ J Cancer 2012. Pelzer U, et al, JCO 2015

Importance of the Identification of high risk patients • The identification of ambulatory cancer patients who might benefit from primary thromboprophylaxis is still one of the most challenging areas • The rate of VTE differs widely among the cancer population, and the risk of VTE also must be weighed against the increased risk of major bleeding • The guidelines encouraged the use of risk assessment models (RAMs), like the Khorana score

How to identify high risk patients: Risk Assessment Models • One approach is the stratification of patients according to their risk of thrombosis, applying risk assessment models (RAMs) that include clinical parameters and biomarkers.

A number of patient-, treatment-, and tumor-related risk factors contribute to the overall VTE risk in cancer. Epidemiology, risk and outcomes of venous thromboembolism in cancer. Falanga; L. Russo - Hamostaseologie. 2012

Risk Factors for Cancer-associated Thrombosis Patient-related factors • Advanced age • Female gender • Prior VTE • Patient co-morbidities • Infection, obesity, anemia, pulmonary or renal disease • Prolonged immobilization • Inherited Thrombophilic factors Cancer-related factors • Site: brain, pancreas, kidney, stomach, lung, bladder, gynecologic, hematologic malignancies • Stage: advanced stage and initial period after diagnosis • Hospitalization • Surgery • Chemo- and hormonal therapy • Anti-angiogenic therapy • Erythropoiesis stimulating agents • Blood transfusions Candidate Biomarkers • Platelet count (>350, 000/µl) • Leukocyte count (> 11, 000/µl) • D-dimer • Tissue Factor expression by tumor cells • Circulating tissue factor (MPs) • Soluble P-selectin A. Falanga, L. Russo - Hamostaseologie. 2012 • C-reactive protein
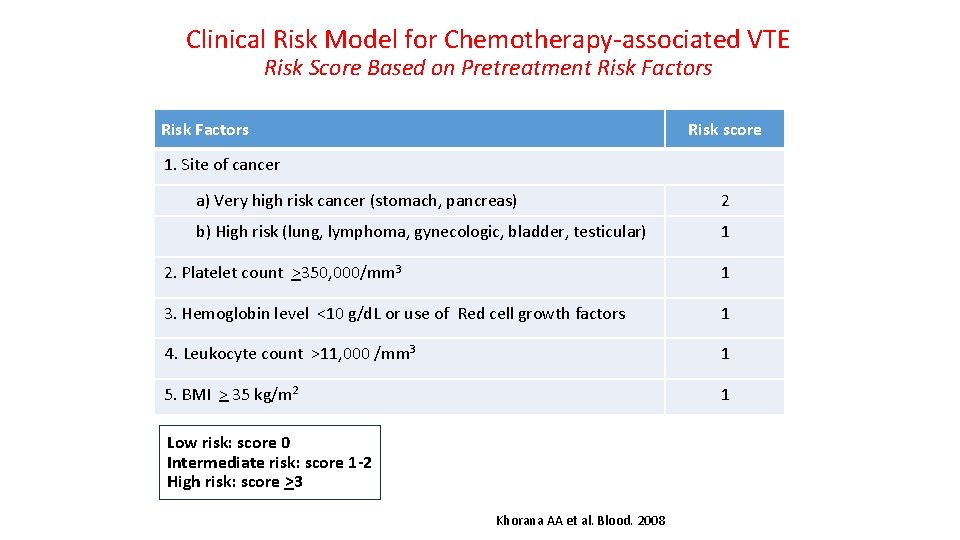
Clinical Risk Model for Chemotherapy-associated VTE Risk Score Based on Pretreatment Risk Factors Risk score 1. Site of cancer a) Very high risk cancer (stomach, pancreas) 2 b) High risk (lung, lymphoma, gynecologic, bladder, testicular) 1 2. Platelet count >350, 000/mm 3 1 3. Hemoglobin level <10 g/d. L or use of Red cell growth factors 1 4. Leukocyte count >11, 000 /mm 3 1 5. BMI > 35 kg/m 2 1 Low risk: score 0 Intermediate risk: score 1 -2 High risk: score >3 Khorana AA et al. Blood. 2008

Risk model-based Clinical Trials of LMWH Study (Agent) PHACS* (LMWH x 12 wks) Micro. TEC (LMWH x 6 mos) Criteria for N inclusion* -Khorana Risk score 404 >=3 Endpoints -Lung, colon, pancreas -Metastatic or unresectable -Elevated TF MPs VTE 227 Studies enroll patients initiating a new chemotherapy regimen Both studies were underpowered for low rate recruitment: • PHACS enrolled 117 patients • Micro. TEC enrolled 66 patients Asymptomatic and symptomatic VTE *Khorana AA et al, Thromb Res 2017 **Zwicker J et al, Br J Haematol 2012

ASH 2018: RCTs EVALUATING DOACs IN PRIMARY PREVENTION OF VTE IN “HIGH-RISK” AMBULATORY CANCER PATIENTS Khorana risk score cut-off of ≥ 2 Study Indication Study design Apixaban for the prevention of VTE in cancer patients (AVERT)* VTE prevention in intermediatehigh risk (Khorana score ≥ 2) ambulatory cancer patients receiving chemotherapy Open label RCT of apixaban vs placebo Rivaroxaban VTE prophylaxis in ambulatory cancer participants (CASSINI)** VTE prevention in intermediatehigh risk (Khorana score ≥ 2) ambulatory cancer patients receiving chemotherapy Open label RCT of Rivaroxaban vs placebo *Carrier et al, published in NEJM online, in Dec. 2018 **Khorana et al, presented at ASH (LBA), in Dec. 2018 Both are pub. lished in paper in NEJM this week

The Khorana’s Score • This score was developed in a study population of 4, 066 cancer patients that were enrolled in the “Awareness of Neutropenia in Chemotherapy” (ANC) Study Group Registry and started a new chemotherapy regimen. Khorana AA et al. Blood. 2008
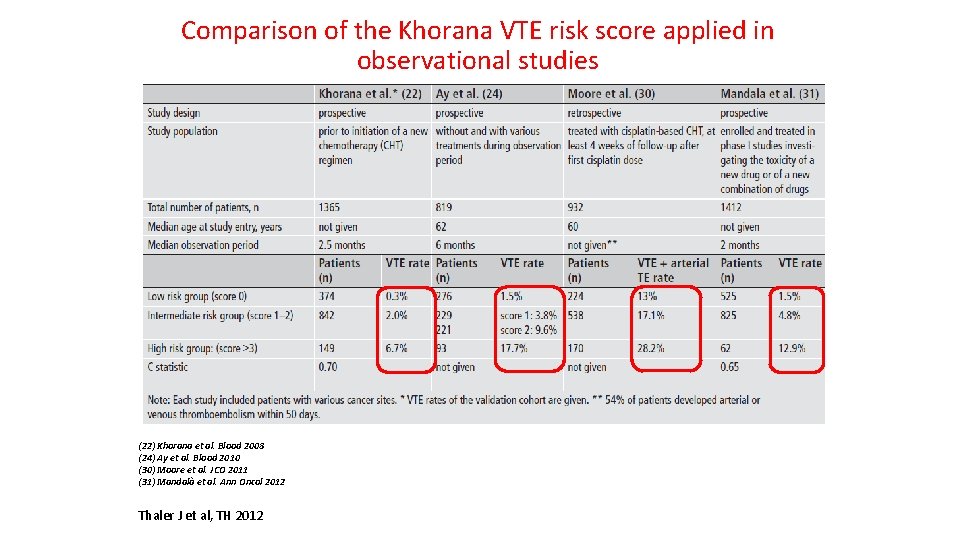
Comparison of the Khorana VTE risk score applied in observational studies (22) Khorana et al. Blood 2008 (24) Ay et al. Blood 2010 (30) Moore et al. JCO 2011 (31) Mandalà et al. Ann Oncol 2012 Thaler J et al, TH 2012
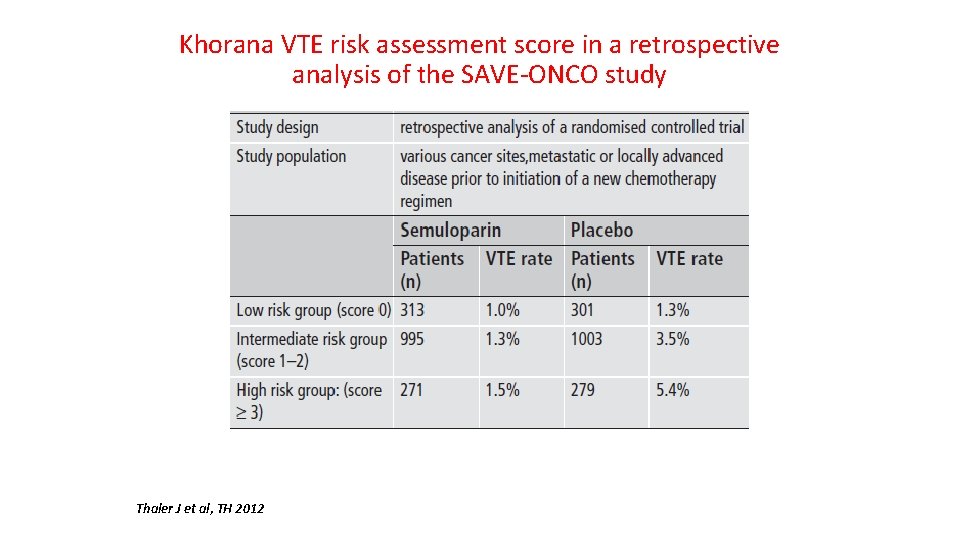
Khorana VTE risk assessment score in a retrospective analysis of the SAVE-ONCO study Thaler J et al, TH 2012
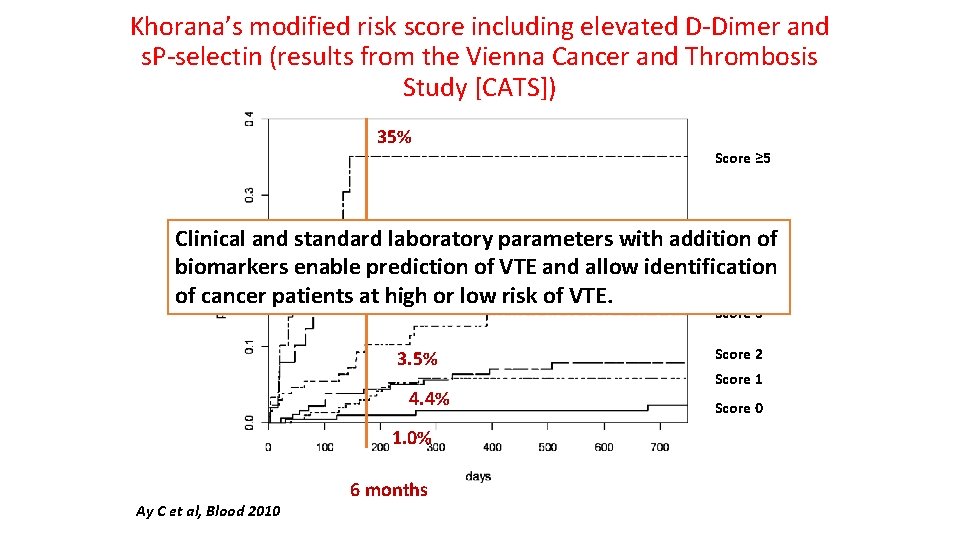
Khorana’s modified risk score including elevated D-Dimer and s. P-selectin (results from the Vienna Cancer and Thrombosis Study [CATS]) 35% Score ≥ 5 Score 4 Clinical and standard 20. 3% laboratory parameters with addition of biomarkers enable prediction of VTE and allow identification of cancer patients at 10. 3% high or low risk of VTE. Score 3 3. 5% 4. 4% 1. 0% Ay C et al, Blood 2010 6 months Score 2 Score 1 Score 0

Protecht* predictive score • Adding to the score based on the five predictive variables of the Khorana score: • treatment with cisplatin or carboplatin-based chemotherapy or gemcitabine = 1 point • In association = 2 points *Outpatients with metastatic or locally advanced solid tumors were randomized in a 2: 1 ratio to receive either subcutaneous injections of LMWH (3800 anti-Xa IU once daily) or placebo. Study treatment was started on the same day as chemotherapy and was given for the duration of chemotherapy or up to a maximum of 120 days (+/- 10 days). Verso M et al, Intern Emerg Med 2012
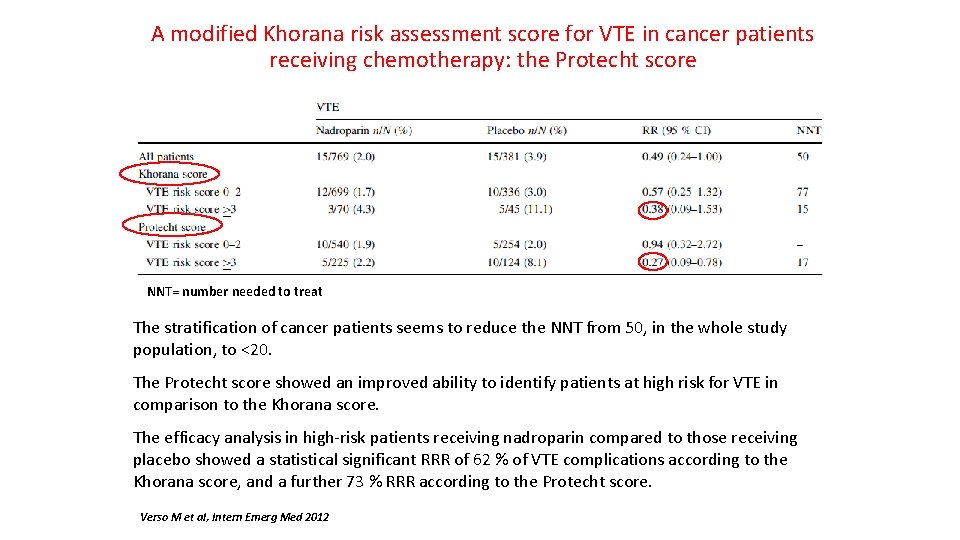
A modified Khorana risk assessment score for VTE in cancer patients receiving chemotherapy: the Protecht score NNT= number needed to treat The stratification of cancer patients seems to reduce the NNT from 50, in the whole study population, to <20. The Protecht score showed an improved ability to identify patients at high risk for VTE in comparison to the Khorana score. The efficacy analysis in high-risk patients receiving nadroparin compared to those receiving placebo showed a statistical significant RRR of 62 % of VTE complications according to the Khorana score, and a further 73 % RRR according to the Protecht score. Verso M et al, Intern Emerg Med 2012

Lancet Haematol 2018; 5: e 289– 98 Development Study: Vienna Cancer and Thrombosis Study (CATS) cohort (n=1423) to select prognostic variables for inclusion in the model. Validation Study: Multinational Cohort Study to Identify Cancer Patients at High Risk of Venous Thromboembolism (MICA) cohort (n=832). Numerous clinical variables and biomarkers were considered, but ONLY TWO included in the final Development model: • tumour-site category (the most important component of the Khorana score) • D-dimer concentrations. Pabinger I, et al. Lancet Haematol 2018
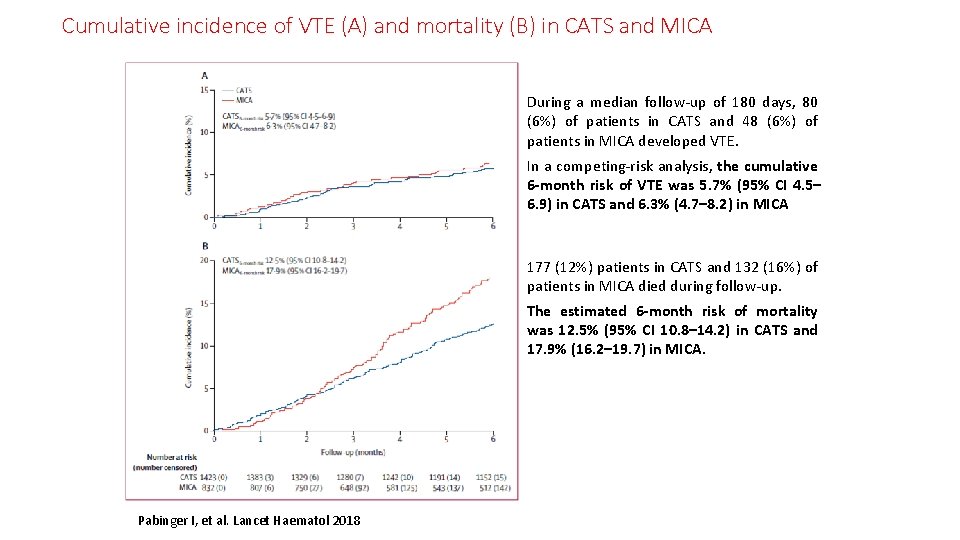
Cumulative incidence of VTE (A) and mortality (B) in CATS and MICA During a median follow-up of 180 days, 80 (6%) of patients in CATS and 48 (6%) of patients in MICA developed VTE. In a competing-risk analysis, the cumulative 6 -month risk of VTE was 5. 7% (95% CI 4. 5– 6. 9) in CATS and 6. 3% (4. 7– 8. 2) in MICA 177 (12%) patients in CATS and 132 (16%) of patients in MICA died during follow-up. The estimated 6 -month risk of mortality was 12. 5% (95% CI 10. 8– 14. 2) in CATS and 17. 9% (16. 2– 19. 7) in MICA. Pabinger I, et al. Lancet Haematol 2018

With inclusion of one clinical factor (tumour-site risk category) and one biomarker (D-dimer), this model was able to discriminate between patients at low and high risk of venous thromboembolism. This clinical prediction model could outperform previous clinical prediction scores in predicting those patients at high risk of developing VTE. Additionally, this model may have the potential for use in selection of patients with cancer who might benefit from thromboprophylaxis. Pabinger I, et al. Lancet Haematol 2018

Prospective Comparison of Methods for thromboembolic risk assessment with clinical Perceptions and Awarene. SS in real life patients - Cancer Associated Thrombosis (COMPASS–CAT ) This multicenter, prospective, longitudinal, non-interventional study was undertaken in outpatients suffering from breast, colon, lung, or ovarian cancer. Aim of the study was to identify the most relevant risk factors for symptomatic VTE and to develop a RAM applicable to patients after the initiation of anticancer treatment.
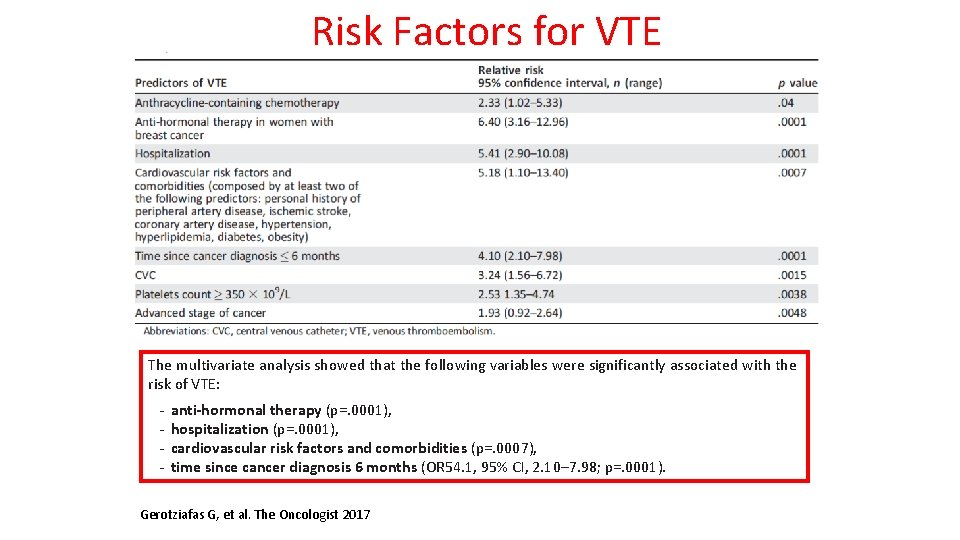
Risk Factors for VTE The multivariate analysis showed that the following variables were significantly associated with the risk of VTE: - anti-hormonal therapy (p=. 0001), hospitalization (p=. 0001), cardiovascular risk factors and comorbidities (p=. 0007), time since cancer diagnosis 6 months (OR 54. 1, 95% CI, 2. 10– 7. 98; p=. 0001). Gerotziafas G, et al. The Oncologist 2017

Simplified COMPASS–CAT Score for VTE prediction in ambulatory patients with common cancers on anticancer therapy Gerotziafas G, et al. The Oncologist 2017
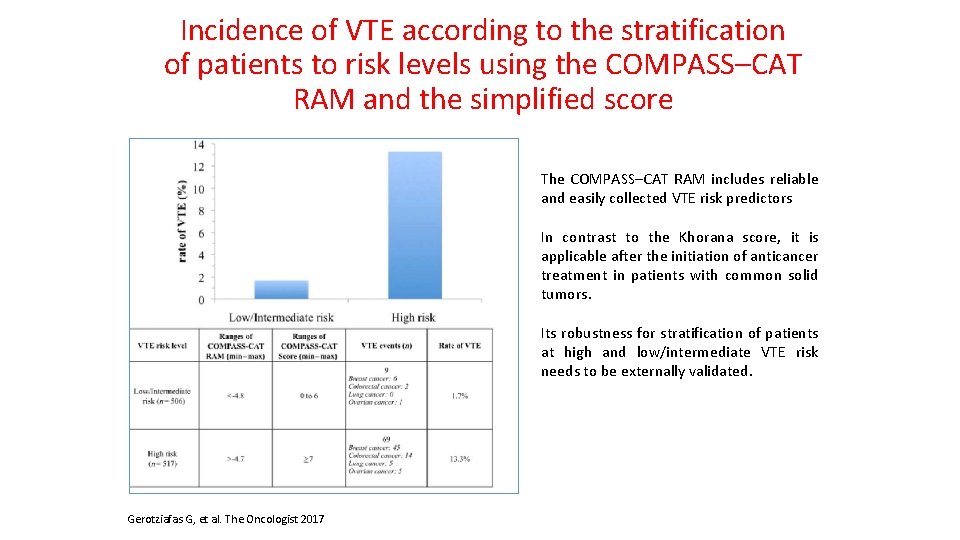
Incidence of VTE according to the stratification of patients to risk levels using the COMPASS–CAT RAM and the simplified score The COMPASS–CAT RAM includes reliable and easily collected VTE risk predictors In contrast to the Khorana score, it is applicable after the initiation of anticancer treatment in patients with common solid tumors. Its robustness for stratification of patients at high and low/intermediate VTE risk needs to be externally validated. Gerotziafas G, et al. The Oncologist 2017


Study design • Aim: to evaluate the performance of the Khorana score in predicting VTE in ambulatory cancer patients. • Embase and MEDLINE were searched from January 2008 to June 2018 for studies which evaluated the Khorana score. • Additional data on the 6 -month incidence of venous thromboembolism were sought by contacting corresponding authors. • A total of 45 articles and 8 abstracts were included, comprising 55 cohorts enrolling 34, 555 ambulatory cancer patients. • The incidence in each Khorana score risk group was estimated with random effects meta-analysis.

RESULTS • Overall, 19% of patients had a Khorana score of 0 points, 64% a score of 1 or 2 points, and 17% a score of 3 or more points. • The incidence of VTE in the first 6 months was: • 5. 1% (95% CI 3. 9 -6. 5) in patients with a low-risk Khorana score (0 points), • 6. 6% (95% CI 5. 6 -7. 7) in those with an intermediate-risk Khorana score (1 or 2 points), and • 11. 0% (95% CI 8. 8 -13. 8) in those with a high-risk Khorana score (3 points or higher). • Of the patients with VTE in the first 6 months, 23. 4% (95% CI 18. 4 to 29. 4) had been classified as ‘high risk’ according to the Khorana score. • In conclusion, the Khorana score can be used to select ambulatory cancer patients at high risk of VTE for thromboprophylaxis, however most events occur outside this high-risk group. Frits I. Mulder , et al. Haematologica 2019

Under active investigation is also The predictive value of hypercoagulation markers in cancer prognosis and diagnosis

Why we need new tumor biomarkers? Remarkable advances in tumor biology research and in "omics" technologies Only few tumor biomarkers have been adopted successfully into routine clinical care of oncologic patients in the last 30 years. personalized medicine growing of therapeutic armamentarium early cancer detection does improve both survival and quality of life Mordente A, et al. Cancer Biomarkers Discovery and Validation: State of the Art, Problems and Future Perspectives, Advances in experimental medicine and biology 867 (2015) 9 -26. Diamandis E. P. . The failure of protein cancer biomarkers to reach the clinic: why, and what can be done to address the problem? , BMC medicine 10 (2012) 87.
Cancer and Thrombosis: A two-way relation Cancer increases the risk of thrombosis CANCER THROMBOSIS Thrombosis increases the risk of cancer
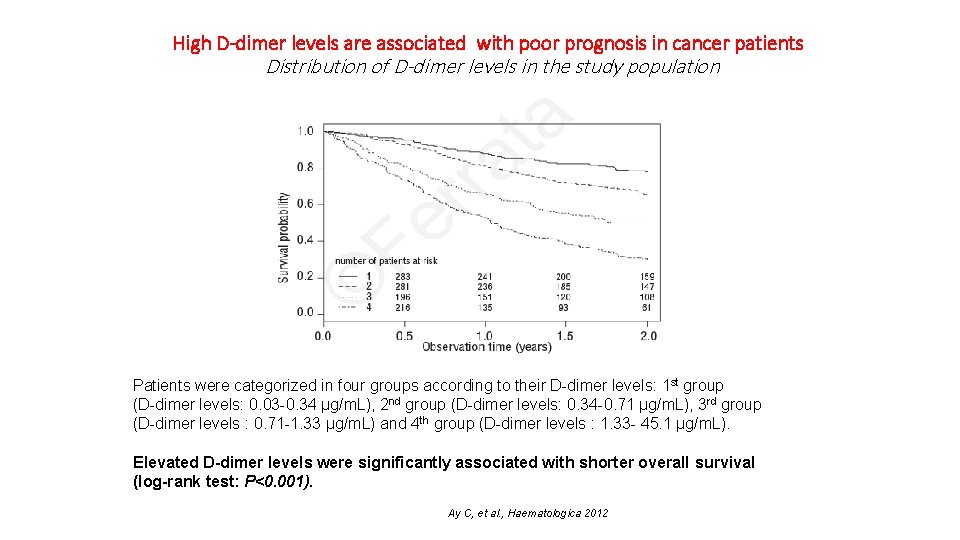
High D-dimer levels are associated with poor prognosis in cancer patients Distribution of D-dimer levels in the study population Patients were categorized in four groups according to their D-dimer levels: 1 st group (D-dimer levels: 0. 03 -0. 34 μg/m. L), 2 nd group (D-dimer levels: 0. 34 -0. 71 μg/m. L), 3 rd group (D-dimer levels : 0. 71 -1. 33 μg/m. L) and 4 th group (D-dimer levels : 1. 33 - 45. 1 μg/m. L). Elevated D-dimer levels were significantly associated with shorter overall survival (log-rank test: P<0. 001). Ay C, et al. , Haematologica 2012

D-Dimer and survival in cancer • Circulating D-dimer levels are better predictors of overall survival and disease progression than carcinoembryonic antigen levels in patients with metastatic colorectal carcinoma. Blackwell K, et al. Cancer 2004 • Is circulating D-dimer level a better prognostic indicator than CEA in resectable colorectal cancer? Our experience on 199 cases. Perazzani et al. Int J Biol Markers. 2010 • The association with age, human tissue kallikreins 6 and 10 and hemostatic markers for survival outcome from epithelial ovarian cancer. Koh SC et al. Arch Gynecol Obstet 2011
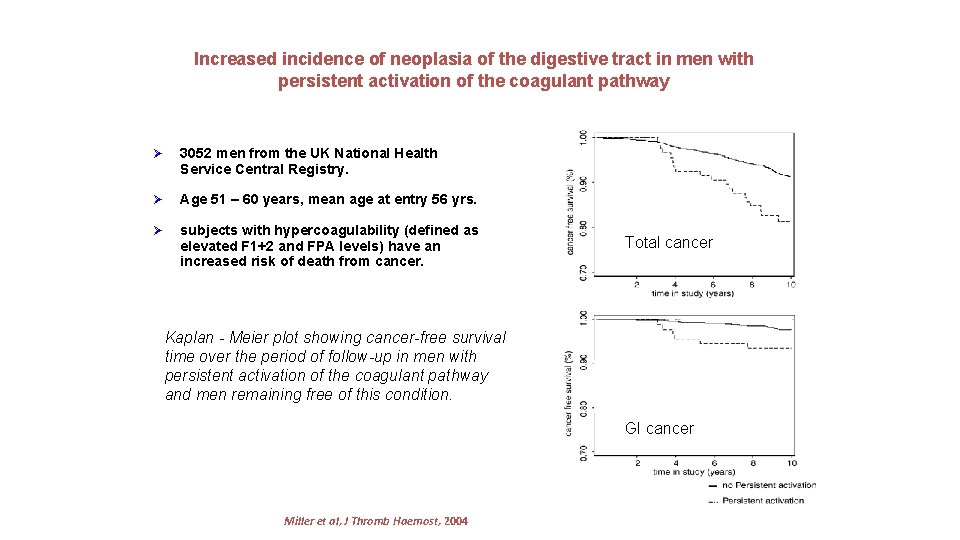
Increased incidence of neoplasia of the digestive tract in men with persistent activation of the coagulant pathway Ø 3052 men from the UK National Health Service Central Registry. Ø Age 51 – 60 years, mean age at entry 56 yrs. Ø subjects with hypercoagulability (defined as elevated F 1+2 and FPA levels) have an increased risk of death from cancer. total cancer Total cancer Kaplan - Meier plot showing cancer-free survival time over the period of follow-up in men with persistent activation of the coagulant pathway and men remaining free of this condition. GI cancer of the digestive tract Miller et al, J Thromb Haemost, 2004

Project AIRC 5 xmille – PI A. Falanga The HYPERCAN STUDY A PROSPECTIVE OBSERVATIONAL MULTICENTER ITALIAN STUDY • Hypercoagulation screening as an innovative tool for risk assessment, early diagnosis and prognosis in cancer

BACKGROUND OF THE STUDY Relevant to this Project is the concept that activation of blood coagulation results in an advantage for cancer cells

OBJECTIVES OF THE PROJECT To establish whether the persistence of a hypercoagulable state IN HEALTHY SUBJECTS may be associated to an increased risk of having a cancer diagnosis. To assess whether the occurrence of a hypercoagulable state IN PATIENTS WITH CANCER may be a marker of cancer prognosis in patients with four types of cancers (breast, lung, gastric, and colo-rectal). Falanga et al, Thromb Res, 2016

The HYPERCAN STUDY Ongoing Italian prospective, multicenter, observational study, composed of 2 main Projects: PROJECT 1. Assessment of thrombotic markers as a tool for cancer risk prediction in healthy subjects PROJECT 2. Evaluation of thrombotic markers in patients with NEWLY DIAGNOSED cancer in relation with the disease prognosis and VTE Falanga et al, Thromb Res 2016 HYPERCAN STUDY (AIRC 5 x 1000 grant #12237) Registered in Clinical. Trials. gov, Identification Number: NCT 02622815 Evaluation of thrombotic markers in patients with cancer in relation with prognosis and VTE

PROJECT 1. Assessment of thrombotic markers as a tool for cancer risk prediction in healthy subjects Healthy blood donors from Bergamo and Milan areas enrolled after written informed consent: • • Administration of a questionnaire on lifestyle and personal history; Blood samples collection: 1) at enrollement; and 2) after 6 -10 months; Follow up: 5 years; Enrollment start up: November 2012; • Subjects enrolled at January 2019 = 9, 000 INCLUSION CRITERIA • • • Blood donors men/women 35 -65 years old Good health* Signed informed consent EXCLUSION CRITERIA • Inflammations/Infections/Fever • Recent Vaccinations • Anticoagulant therapy ongoing • Recent Surgery • blood donors are periodically screened for a series of serological, biochemical and clinical parameters, and tested for viral infections

PROJECT 2. Evaluation of thrombotic markers in patients with NEWLY DIAGNOSED cancer in relation with prognosis and VTE Tumor type: Breast, NSCLC, CRC, and gastric cancers Outcomes: Overall Survival (OS), Progression free Survival (PFS), Disease free Survival (DFS), and VTE free survival (VFS). Blood sample schedule LIMITED RESECTED DISEASE q Enrollment, after surgery and before starting adjuvant CT q 1 year q 2 years q 3 years q 4 years q at recurrence METASTATIC DISEASE q Enrollment, before starting curative CT q 3 chemotherapy cycles q 6 chemotherapy cycles q end of treatment q at progression At each time point, information on treatment, recurrence, metastasis, survival and VTE are documented. Samples are stored at the Biobank of Papa Giovanni XXIII Hospital - BERGAMO (Italy)

PROJECT 2. Evaluation of thrombotic markers in patients with NEWLY DIAGNOSED cancer in relation with prognosis and VTE Enrolled: • ~4, 000 cancer patients (NSCLC, Breast, CRC, and Gastric): Ø 64% with limited disease Ø 36% with metastatic disease Update: May 2019

HYPERCAN INVESTIGATORS COORDINATING CENTER Department of Immunohematology and Transfusion Medicine & Thrombosis and Hemostasis Center Hospital Papa Giovanni XXIII, Bergamo (ITALY) PARTICIPATING CENTERS - Clinical Institute Humanitas, Rozzano, Milan (A. Santoro) - IRCCS National Cancer Institute, Milan (F. De Braud) - Hospital Papa Giovanni XXIII, Bergamo (R Labianca) - ASL Roma 1, Rome (G. Gasparini), Hospital San Giovanni Addolorata, Rome (E. Minelli) - IRCCS National Cancer Institute Giovanni Paolo II, Bari (F. Giuliani) - Policlinico San Marco, Zingonia Bergamo (A. D’Alessio) - Hospital Treviglio-Caravaggio, Treviglio , Bergamo (F. Petrelli) -Neuromed Institute, Pozzilli, Isernia (L. Iacoviello) STATISTICAL UNIT D. Spinelli, P. Malighetti HTH - University of Bergamo EPIDEMIOLOGICAL UNIT G. Sanpietro Public Health Agency - Bergamo EXTERNAL ADVISORY BOARD FR Rickles (Washington DC, USA) PH Reitsma (Leiden, NL) Supported by the Italian Cancer Research Association (AIRC)

CONCLUSIONS • Biomarkers of clotting activation are promising in predicting VTE. Their incorporation in Risk assessment models (RAMs) improves the identification of patients at high thrombotic risk. • The research is very active in this field and new RAMs with additional simple variables are coming. • For the time being the Khorana score can be helpful in selecting patients for thromboprophylaxis. • Further prospective studies involving large numbers of patients with specific cancer types are warranted to show the predictive value of these biomarkers not only for VTE, but also for cancer survival.

- Slides: 52