Enasidenib Plus Azacitidine vs Azacitidine Monotherapy in m

Enasidenib Plus Azacitidine vs Azacitidine Monotherapy in m. IDH 2 Newly Diagnosed Acute Myeloid Leukemia CCO Independent Conference Coverage* Highlights of the 2020 ASCO Virtual Scientific Meeting, May 29 -31, 2020 *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is provided by Clinical Care Options, LLC Supported by educational grants from Abb. Vie, Inc. ; Astra. Zeneca; Bristol-Myers Squibb; Glaxo. Smith. Kline LLC; and Karyopharm Therapeutics Inc.
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

Enasidenib + AZA in Patients With Newly Diagnosed AML: Background § 8% to 19% of patients with AML have m. IDH 2; prevalence increases with age[1, 2] § m. IDH 2 result in accumulation of oncometabolite 2 -HG, which blocks differentiation, lowers apoptotic threshold, and alters hypoxic responses § Enasidenib is an oral small molecule inhibitor of m. IDH 2 proteins that promotes myeloid cell differentiation and blocks 2 -HG function[3, 4]; currently approved in US and conditionally approved in Australia and Canada for use in treatment of m. IDH 2 R/R AML ‒ Monotherapy with enasidenib or AZA induces ORR of ~ 30% and CR rates of ~ 20% in newly diagnosed AML § Combining enasidenib + AZA enhances cell differentiation in vitro; phase Ib results of current study suggested combination therapy was tolerable[3] § Current analysis reports results of randomized phase II of enasidenib + AZA vs AZA monotherapy trial in patients with newly diagnosed m. IDH 2 AML 1. Chan. Nature Med. 2015; 2: 178. 2. Heuser. Exp Hematol. 2015; 43: 685. 3. Stein. Blood. 2017; 130: 722. 4. Shih. Blood. 2014; 124: 437. Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com
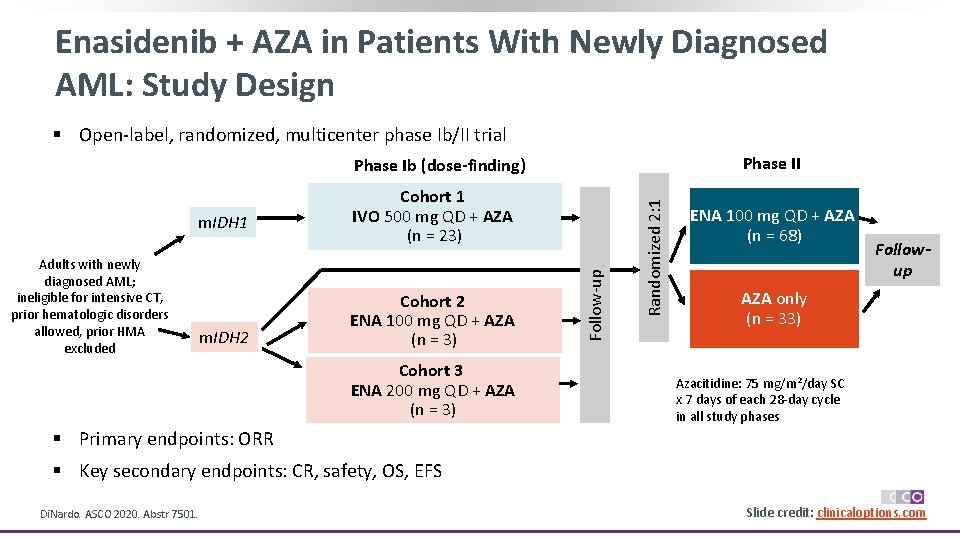
Enasidenib + AZA in Patients With Newly Diagnosed AML: Study Design § Open-label, randomized, multicenter phase Ib/II trial Phase II Adults with newly diagnosed AML; ineligible for intensive CT, prior hematologic disorders allowed, prior HMA excluded m. IDH 2 Cohort 2 ENA 100 mg QD + AZA (n = 3) Cohort 3 ENA 200 mg QD + AZA (n = 3) Follow-up m. IDH 1 Cohort 1 IVO 500 mg QD + AZA (n = 23) Randomized 2: 1 Phase Ib (dose-finding) ENA 100 mg QD + AZA (n = 68) Followup AZA only (n = 33) Azacitidine: 75 mg/m 2/day SC x 7 days of each 28 -day cycle in all study phases § Primary endpoints: ORR § Key secondary endpoints: CR, safety, OS, EFS Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com
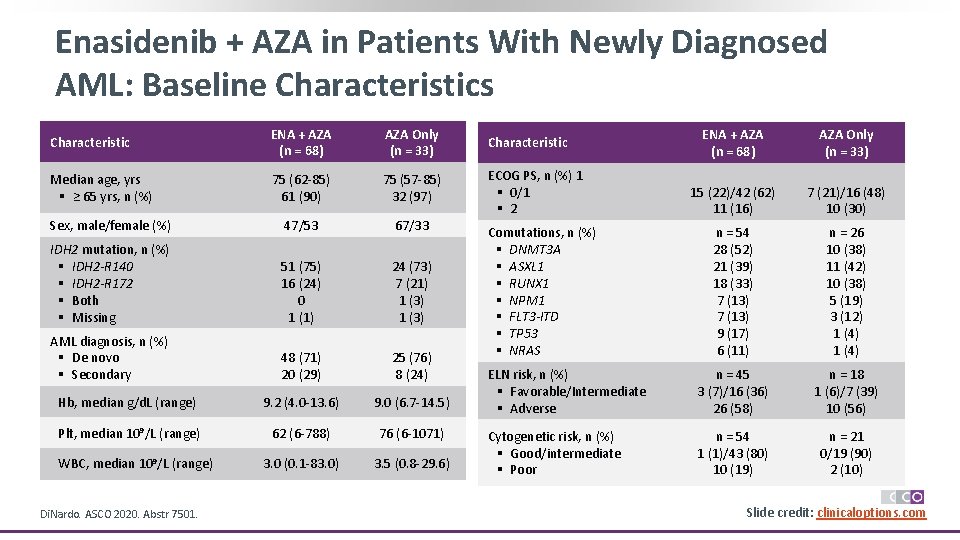
Enasidenib + AZA in Patients With Newly Diagnosed AML: Baseline Characteristics Characteristic ENA + AZA (n = 68) AZA Only (n = 33) Characteristic Median age, yrs § ≥ 65 yrs, n (%) 75 (62 -85) 61 (90) 75 (57 -85) 32 (97) Sex, male/female (%) 47/53 67/33 ECOG PS, n (%) 1 § 0/1 § 2 IDH 2 mutation, n (%) § IDH 2 -R 140 § IDH 2 -R 172 § Both § Missing 51 (75) 16 (24) 0 1 (1) 24 (73) 7 (21) 1 (3) AML diagnosis, n (%) § De novo § Secondary 48 (71) 20 (29) 25 (76) 8 (24) Hb, median g/d. L (range) 9. 2 (4. 0 -13. 6) 9. 0 (6. 7 -14. 5) Plt, median 109/L (range) 62 (6 -788) 76 (6 -1071) 3. 0 (0. 1 -83. 0) 3. 5 (0. 8 -29. 6) WBC, median 109/L (range) Di. Nardo. ASCO 2020. Abstr 7501. ENA + AZA (n = 68) AZA Only (n = 33) 15 (22)/42 (62) 11 (16) 7 (21)/16 (48) 10 (30) n = 54 28 (52) 21 (39) 18 (33) 7 (13) 9 (17) 6 (11) n = 26 10 (38) 11 (42) 10 (38) 5 (19) 3 (12) 1 (4) ELN risk, n (%) § Favorable/Intermediate § Adverse n = 45 3 (7)/16 (36) 26 (58) n = 18 1 (6)/7 (39) 10 (56) Cytogenetic risk, n (%) § Good/intermediate § Poor n = 54 1 (1)/43 (80) 10 (19) n = 21 0/19 (90) 2 (10) Comutations, n (%) § DNMT 3 A § ASXL 1 § RUNX 1 § NPM 1 § FLT 3 -ITD § TP 53 § NRAS Slide credit: clinicaloptions. com
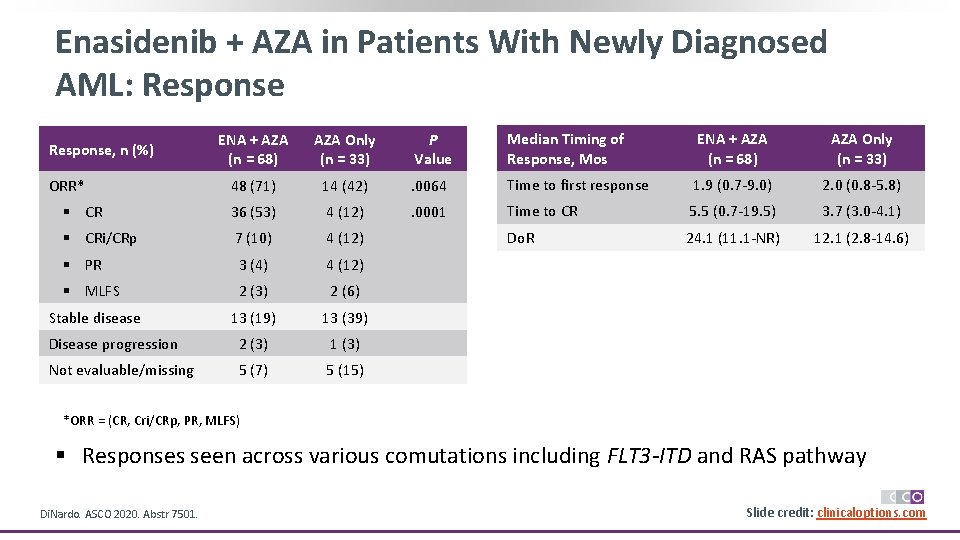
Enasidenib + AZA in Patients With Newly Diagnosed AML: Response ENA + AZA (n = 68) AZA Only (n = 33) P Value Median Timing of Response, Mos ENA + AZA (n = 68) AZA Only (n = 33) 48 (71) 14 (42) . 0064 Time to first response 1. 9 (0. 7 -9. 0) 2. 0 (0. 8 -5. 8) § CR 36 (53) 4 (12) . 0001 Time to CR 5. 5 (0. 7 -19. 5) 3. 7 (3. 0 -4. 1) § CRi/CRp 7 (10) 4 (12) Do. R 24. 1 (11. 1 -NR) 12. 1 (2. 8 -14. 6) § PR 3 (4) 4 (12) § MLFS 2 (3) 2 (6) Stable disease 13 (19) 13 (39) Disease progression 2 (3) 1 (3) Not evaluable/missing 5 (7) 5 (15) Response, n (%) ORR* *ORR = (CR, Cri/CRp, PR, MLFS) § Responses seen across various comutations including FLT 3 -ITD and RAS pathway Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com

Enasidenib + AZA in Patients With Newly Diagnosed AML: Safety § Common TEAEs with ENA + AZA and AZA only: thrombocytopenia (62% and 44%), nausea (69% and 38%), anemia (53% and 44%), vomiting (49% and 47%) § Cytopenias and IDH-DS were common txrelated grade 3 -4 TEAEs in ENA + AZA cohort ‒ IDH-DS at median 28. 5 days in 12 patients (18%) of ENA + AZA cohort § Tx-related grade 3/4 infection: ENA + AZA, 18%; AZA only, 31% § Patients who died: ENA + AZA, 43%; AZA only, 44% ENA + AZA (n = 68) AZA Only (n = 33) Any grade 3/4 50 (75) 20 (63) Thrombocytopenia 25 (37) 6 (19) Neutropenia 24 (35) 7 (22) Anemia 13 (19) 7 (22) Febrile neutropenia 10 (15) 5 (16) IDH-DS 7 (10) 0 ENA + AZA (n = 68) AZA Only (n = 33) 29 (43) 15 (22) 14 (21) 14 (44) 2 (6) 12 (38) 5 (7) 1 (3) TEAE, n (%) Deaths § While on tx § After tx d/c 60 -day mortality rate Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com
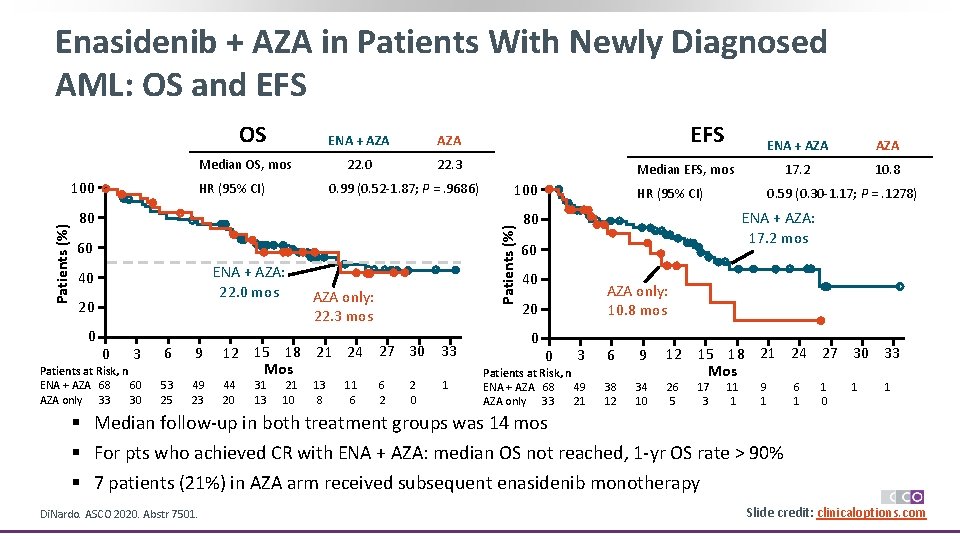
Enasidenib + AZA in Patients With Newly Diagnosed AML: OS and EFS Median OS, mos Patients (%) 100 HR (95% CI) ENA + AZA 22. 0 22. 3 0. 99 (0. 52 -1. 87; P =. 9686) 80 60 ENA + AZA: 22. 0 mos 40 20 0 0 3 6 9 AZA only: 22. 3 mos 12 15 18 21 24 27 30 33 Mos Patients at Risk, n ENA + AZA 68 60 53 49 44 31 21 13 11 6 2 1 AZA only 33 30 25 23 20 13 10 8 6 2 0 EFS ENA + AZA 17. 2 10. 8 Median EFS, mos 100 Patients (%) OS HR (95% CI) ENA + AZA: 17. 2 mos 80 60 40 AZA only: 10. 8 mos 20 0 0. 59 (0. 30 -1. 17; P =. 1278) 0 3 6 9 12 15 18 21 24 27 30 33 Mos Patients at Risk, n ENA + AZA 68 49 38 34 26 17 11 9 6 1 1 AZA only 33 21 12 10 5 3 1 1 0 § Median follow-up in both treatment groups was 14 mos § For pts who achieved CR with ENA + AZA: median OS not reached, 1 -yr OS rate > 90% § 7 patients (21%) in AZA arm received subsequent enasidenib monotherapy Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com
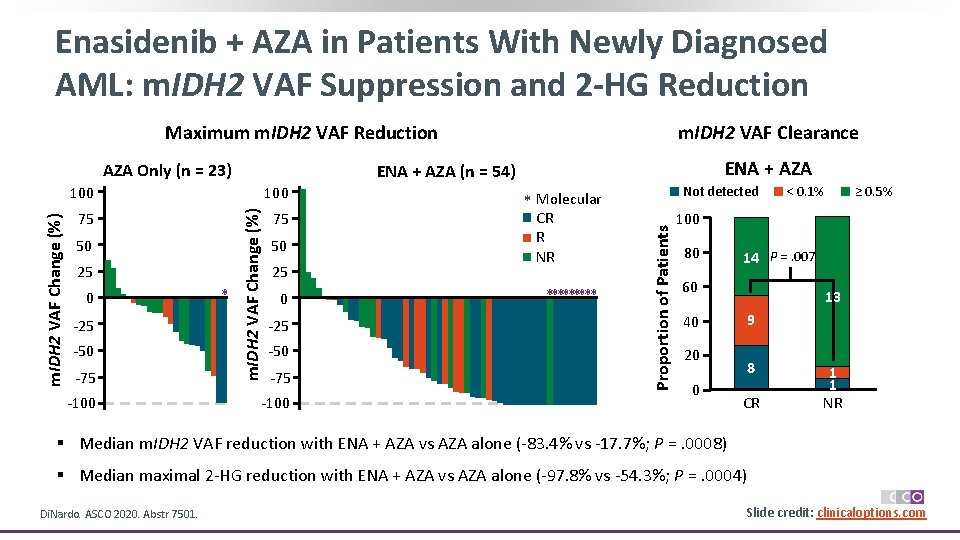
Enasidenib + AZA in Patients With Newly Diagnosed AML: m. IDH 2 VAF Suppression and 2 -HG Reduction Maximum m. IDH 2 VAF Reduction AZA Only (n = 23) 0 -25 -50 -75 -100 * 75 50 25 0 -25 -50 -75 -100 Not detected * Molecular CR R NR ***** Proportion of Patients 25 m. IDH 2 VAF Change (%) 100 50 ENA + AZA (n = 54) 100 75 m. IDH 2 VAF Clearance < 0. 1% ≥ 0. 5% 100 80 14 P =. 007 60 40 20 0 13 9 8 CR 1 1 NR § Median m. IDH 2 VAF reduction with ENA + AZA vs AZA alone (-83. 4% vs -17. 7%; P =. 0008) § Median maximal 2 -HG reduction with ENA + AZA vs AZA alone (-97. 8% vs -54. 3%; P =. 0004) Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com

Enasidenib + AZA in Patients With Newly Diagnosed AML: Conclusions § Among patients with newly diagnosed AML, significant improvement of ORR (the primary endpoint) with ENA + AZA vs AZA alone ‒ ORR (71% vs 42%; P =. 0064) ‒ CR rate (53% vs 12%; P =. 0001) § Reduction in 2 -HG concentrations indicating on-target activity and decreased m. IDH 2 with ENA + AZA § Safety profile of ENA + AZA combination similar to individual monotherapy § Investigators conclude that combining targeted therapy with ENA + AZA was safe and effective Di. Nardo. ASCO 2020. Abstr 7501. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASCO 2020! Short slideset summaries and additional CME-certified analyses with expert faculty commentary on key studies in: § Breast cancer § Gynecologic cancers § Gastrointestinal cancers § Hematologic malignancies § Genitourinary cancers § Lung cancer clinicaloptions. com/oncology
- Slides: 11