Emory Clinical Biomarkers Laboratory Measuring the poise of

Emory Clinical Biomarkers Laboratory Measuring the poise of thiol/disulfide redox in vivo Dean P. Jones, Ph. D. Department of Medicine/Division of Pulmonary, Allergy and Critical Care Medicine Emory University, Atlanta
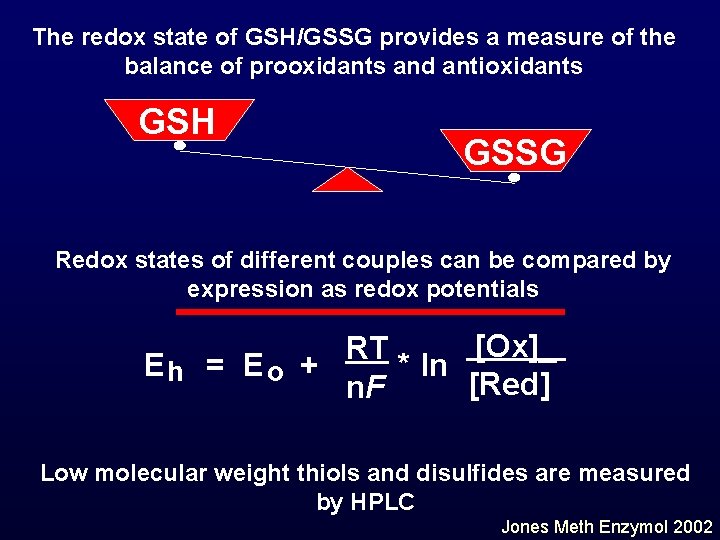
The redox state of GSH/GSSG provides a measure of the balance of prooxidants and antioxidants GSH GSSG Redox states of different couples can be compared by expression as redox potentials [Ox]_ RT Eh = E o + * ln [Red] n. F Low molecular weight thiols and disulfides are measured by HPLC Jones Meth Enzymol 2002
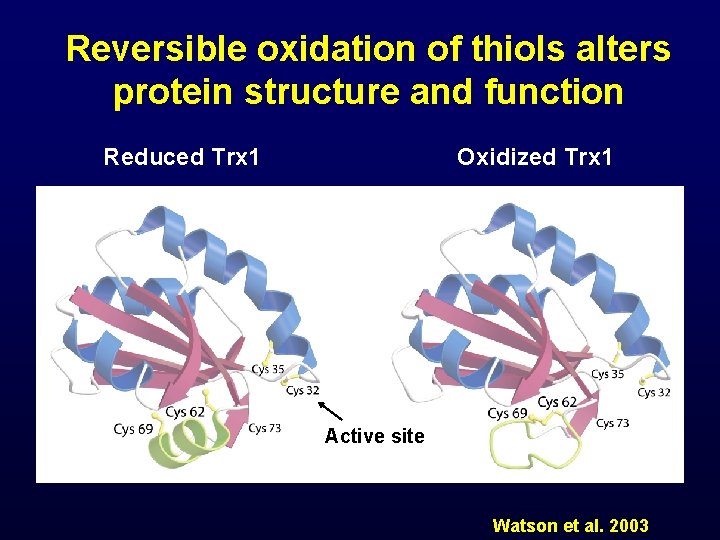
Reversible oxidation of thiols alters protein structure and function Reduced Trx 1 Oxidized Trx 1 Active site Watson et al. 2003
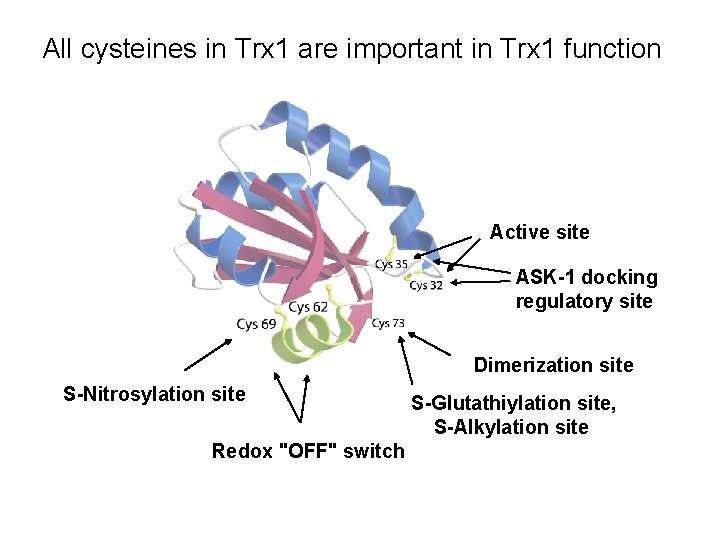
All cysteines in Trx 1 are important in Trx 1 function Active site ASK-1 docking regulatory site Dimerization site S-Nitrosylation site Redox "OFF" switch S-Glutathiylation site, S-Alkylation site
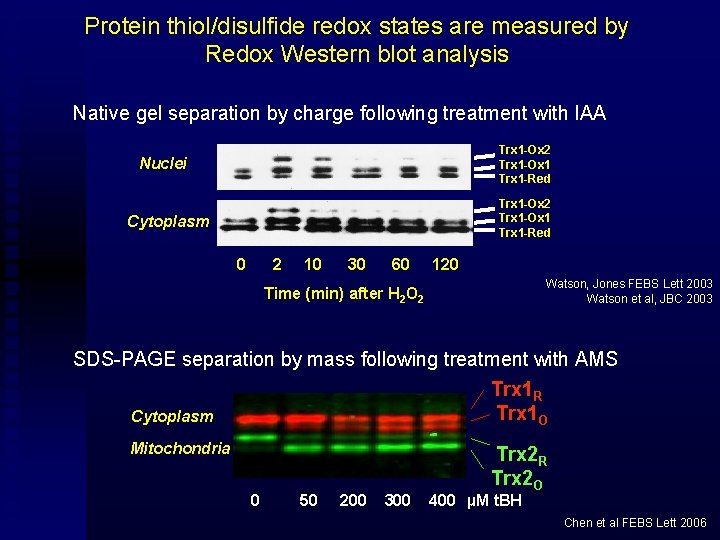
Protein thiol/disulfide redox states are measured by Redox Western blot analysis Native gel separation by charge following treatment with IAA Trx 1 -Ox 2 Trx 1 -Ox 1 Trx 1 -Red Nuclei Trx 1 -Ox 2 Trx 1 -Ox 1 Trx 1 -Red Cytoplasm 0 2 10 30 60 120 Watson, Jones FEBS Lett 2003 Watson et al, JBC 2003 Time (min) after H 2 O 2 SDS-PAGE separation by mass following treatment with AMS Trx 1 R Trx 1 O Cytoplasm Mitochondria 0 50 200 300 Trx 2 R Trx 2 O 400 µM t. BH Chen et al FEBS Lett 2006

Quantification of thiol/disulfide redox in biologic systems has provided 3 general conclusions 1. At the cellular level, GSH redox becomes oxidized as cells progress through the life cycle, and cells regulate extracellular thiol/disulfide redox state 2. At the systemic level, plasma GSH redox becomes oxidized with oxidative stress and is oxidized in association with aging and chronic disease 3. In cells and plasma, GSH redox is NOT equilibrated with thioredoxin or Cys/Cy. SS, providing the basis to consider discrete redox circuits for redox signaling and control
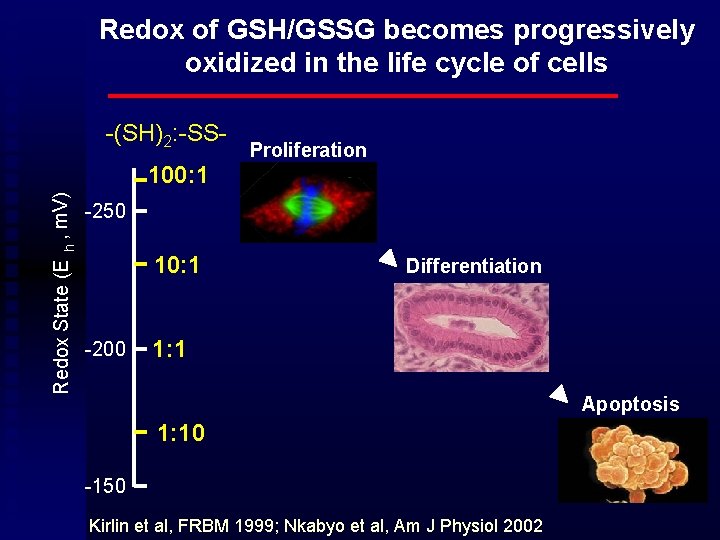
Redox of GSH/GSSG becomes progressively oxidized in the life cycle of cells -(SH)2: -SSRedox State (E h , m. V) 100: 1 Proliferation -250 10: 1 -200 Differentiation 1: 1 Apoptosis 1: 10 -150 Kirlin et al, FRBM 1999; Nkabyo et al, Am J Physiol 2002
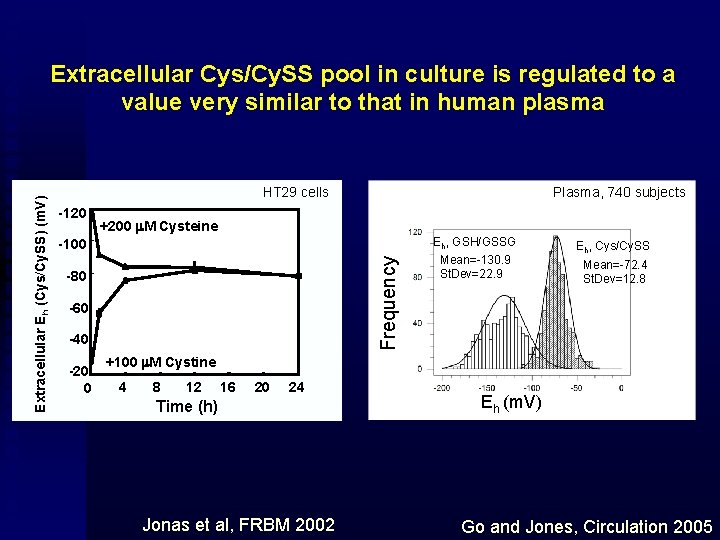
HT 29 cells -120 Plasma, 740 subjects +200 m. M Cysteine -100 Frequency Extracellular Eh (Cys/Cy. SS) (m. V) Extracellular Cys/Cy. SS pool in culture is regulated to a value very similar to that in human plasma -80 -60 -40 -20 0 Eh, GSH/GSSG Mean=-130. 9 St. Dev=22. 9 Eh, Cys/Cy. SS Mean=-72. 4 St. Dev=12. 8 +100 m. M Cystine 4 8 12 16 20 24 Time (h) Jonas et al, FRBM 2002 Eh (m. V) Go and Jones, Circulation 2005

Interorgan GSH/Cysteine balance Tissues Plasma -220 m. V -138 m. V Major pool Most reduced GSH/GSSG Cys/Cy. SS Major pool Most oxidized -80 m. V -150 m. V

Quantification of thiol/disulfide redox in biologic systems has provided 3 conclusions 1. At the cellular level: Cells regulate extracellular thiol/disulfide redox state. Cellular GSH redox becomes oxidized as cells progress through the life cycle 2. At the systemic level: Plasma GSH redox becomes oxidized with oxidative stress. Plasma redox is oxidized with aging, nutritional deficiency, toxicity and chronic disease 3. Relationship of redox couples: GSH redox is NOT equilibrated with thioredoxin or Cys/Cy. SS. This provides the basis to consider discrete redox circuits for redox signaling and control
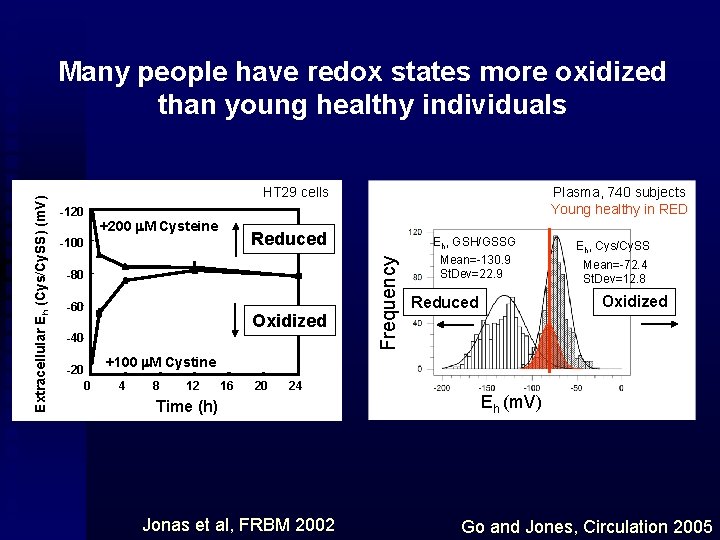
HT 29 cells -120 +200 m. M Cysteine Reduced -100 -80 -60 Oxidized -40 -20 0 Plasma, 740 subjects Young healthy in RED Frequency Extracellular Eh (Cys/Cy. SS) (m. V) Many people have redox states more oxidized than young healthy individuals Eh, GSH/GSSG Mean=-130. 9 St. Dev=22. 9 Eh, Cys/Cy. SS Mean=-72. 4 St. Dev=12. 8 Oxidized Reduced +100 m. M Cystine 4 8 12 16 20 24 Time (h) Jonas et al, FRBM 2002 Eh (m. V) Go and Jones, Circulation 2005
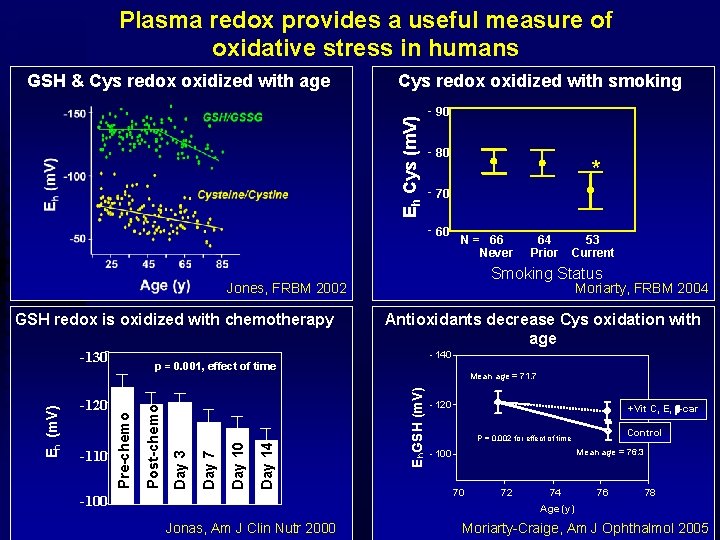
Plasma redox provides a useful measure of oxidative stress in humans Cys redox oxidized with smoking Eh Cys (m. V) GSH & Cys redox oxidized with age - 90 - 80 * - 70 - 60 N = 66 Never Antioxidants decrease Cys oxidation with age -140 --100 Mean age = 71. 7 E h. GSH (m. V) Day 14 Day 10 Day 7 Day 3 -110 Post-chemo -120 Moriarty, FRBM 2004 p = 0. 001, effect of time Pre-chemo Eh (m. V) -130 53 Current Smoking Status Jones, FRBM 2002 GSH redox is oxidized with chemotherapy 64 Prior -120 +Vit C, E, b-car Control P = 0. 002 for effect of time Mean age = 76. 3 -100 70 72 74 76 78 Age (y) Jonas, Am J Clin Nutr 2000 Moriarty-Craige, Am J Ophthalmol 2005
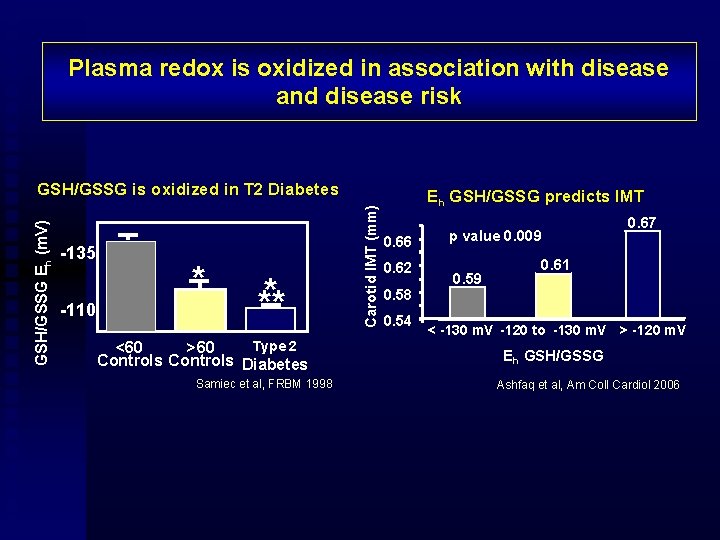
Plasma redox is oxidized in association with disease and disease risk -135 -110 * * ** Type 2 <60 >60 Controls Diabetes Samiec et al, FRBM 1998 Carotid IMT (mm) GSH/GSSG Eh (m. V) GSH/GSSG is oxidized in T 2 Diabetes Eh GSH/GSSG predicts IMT 0. 66 0. 62 0. 58 0. 54 p value 0. 009 0. 59 0. 67 0. 61 < -130 m. V -120 to -130 m. V > -120 m. V Eh GSH/GSSG Ashfaq et al, Am Coll Cardiol 2006
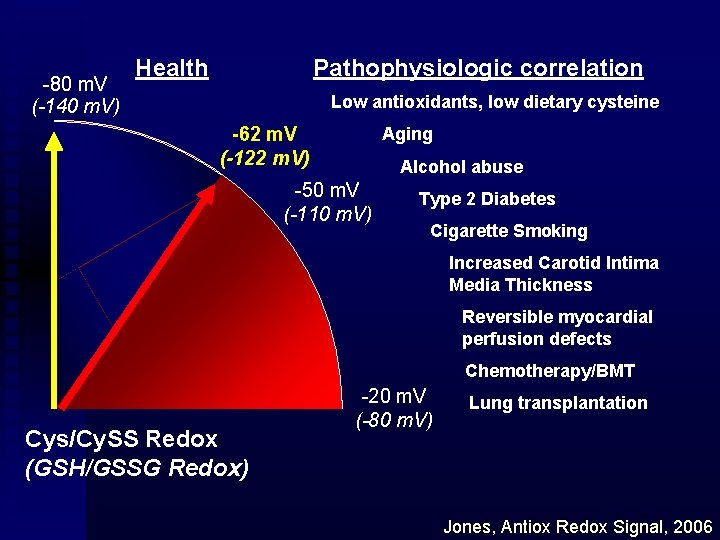
-80 m. V (-140 m. V) Health Pathophysiologic correlation Low antioxidants, low dietary cysteine -62 m. V (-122 m. V) Aging Alcohol abuse -50 m. V (-110 m. V) Type 2 Diabetes Cigarette Smoking Increased Carotid Intima Media Thickness Reversible myocardial perfusion defects Chemotherapy/BMT Cys/Cy. SS Redox (GSH/GSSG Redox) -20 m. V (-80 m. V) Lung transplantation Jones, Antiox Redox Signal, 2006

Quantification of thiol/disulfide redox in biologic systems has provided 3 general conclusions 1. At the cellular level, GSH redox becomes oxidized as cells progress through the life cycle, and cells regulate extracellular thiol/disulfide redox state 2. At the systemic level, plasma GSH redox becomes oxidized with oxidative stress and is oxidized in association with aging and chronic disease 3. In cells and plasma, GSH redox is NOT equilibrated with thioredoxin or Cys/Cy. SS, providing the basis to consider discrete redox circuits for redox signaling and control

GSH, Trx and Cys redox systems are not in redox equilibrium in cells -300 Trx Proliferation Differentiation Redox State (Eh , m. V) Apoptosis -250 Proliferation GSH -200 -150 Differentiation Cys Proliferation Apoptosis Differentiation Jones et al FASEB J 2004
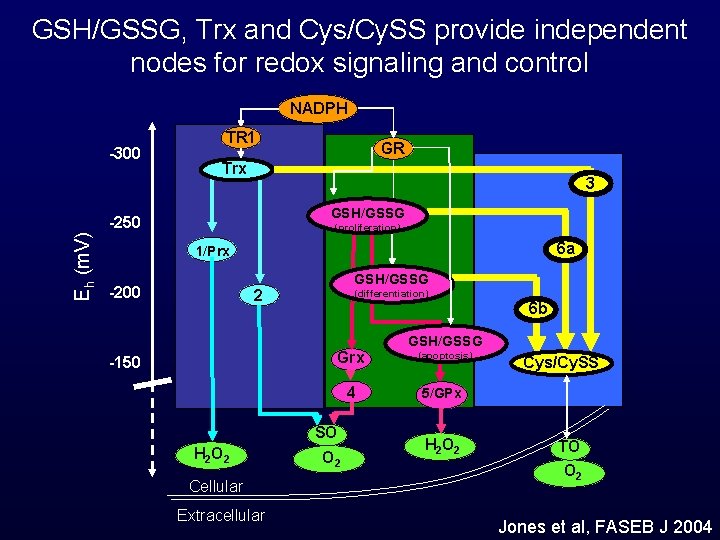
GSH/GSSG, Trx and Cys/Cy. SS provide independent nodes for redox signaling and control NADPH -300 TR 1 Trx 3 GSH/GSSG -250 Eh (m. V) GR (proliferation) 6 a 1/Prx -200 GSH/GSSG 2 (differentiation) 6 b GSH/GSSG Grx -150 4 SO H 2 O 2 Cellular Extracellular O 2 (apoptosis) Cys/Cy. SS 5/GPx H 2 O 2 TO O 2 Jones et al, FASEB J 2004
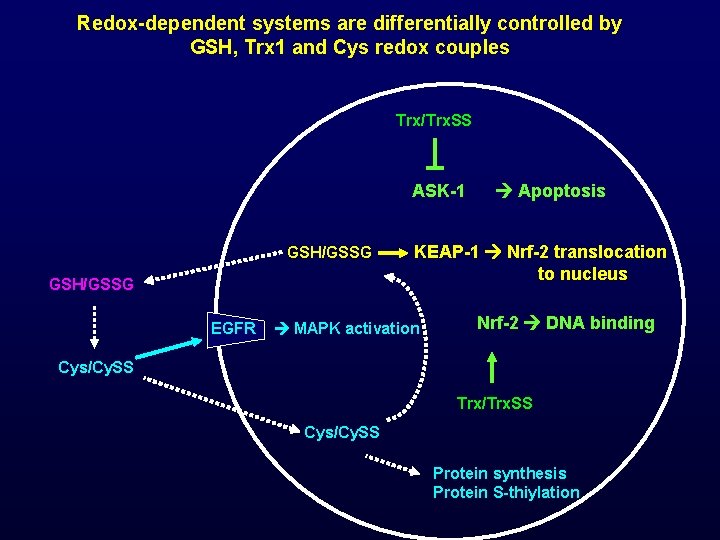
Redox-dependent systems are differentially controlled by GSH, Trx 1 and Cys redox couples Trx/Trx. SS ASK-1 GSH/GSSG EGFR Apoptosis KEAP-1 Nrf-2 translocation to nucleus MAPK activation Nrf-2 DNA binding Cys/Cy. SS Trx/Trx. SS Cys/Cy. SS Protein synthesis Protein S-thiylation
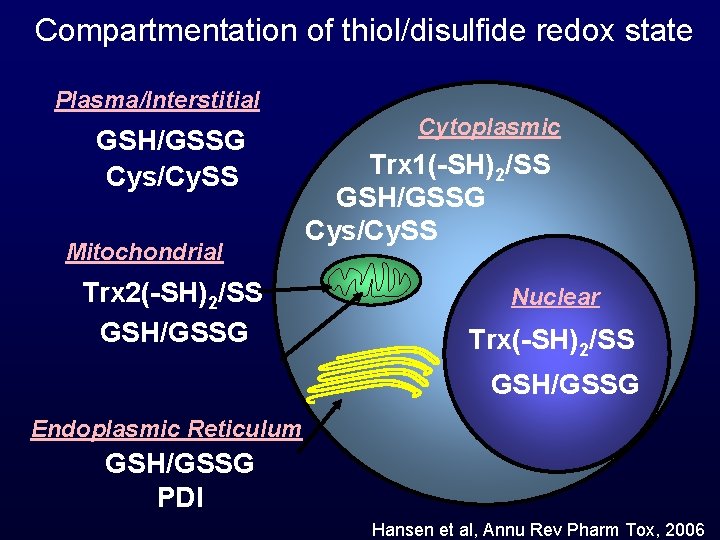
Compartmentation of thiol/disulfide redox state Plasma/Interstitial GSH/GSSG Cys/Cy. SS Mitochondrial Trx 2(-SH)2/SS GSH/GSSG Cytoplasmic Trx 1(-SH)2/SS GSH/GSSG Cys/Cy. SS Nuclear Trx(-SH)2/SS GSH/GSSG Endoplasmic Reticulum GSH/GSSG PDI Hansen et al, Annu Rev Pharm Tox, 2006
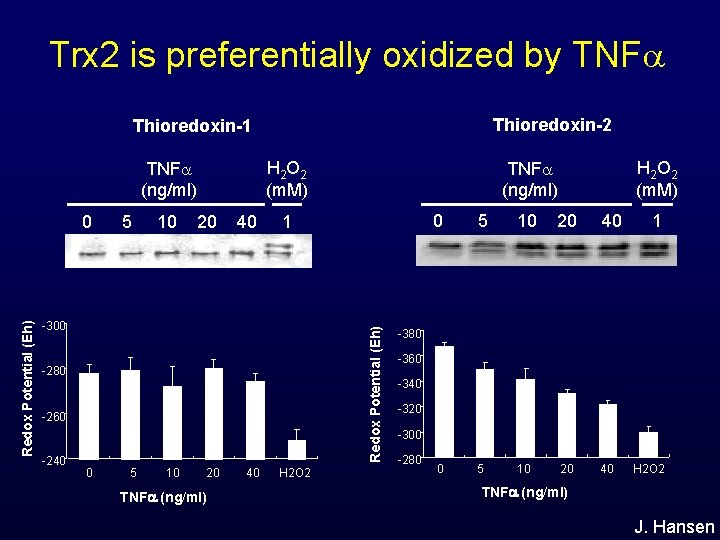
Trx 2 is preferentially oxidized by TNF Thioredoxin-2 Thioredoxin-1 H 2 O 2 (m. M) TNF (ng/ml) 5 10 20 40 1 -300 Redox Potential (Eh) 0 -280 -260 -240 0 5 10 20 TNF (ng/ml) 40 H 2 O 2 H 2 O 2 (m. M) TNF (ng/ml) 0 5 10 20 40 1 -380 -360 -340 -320 -300 -280 H 2 O 2 TNF (ng/ml) J. Hansen
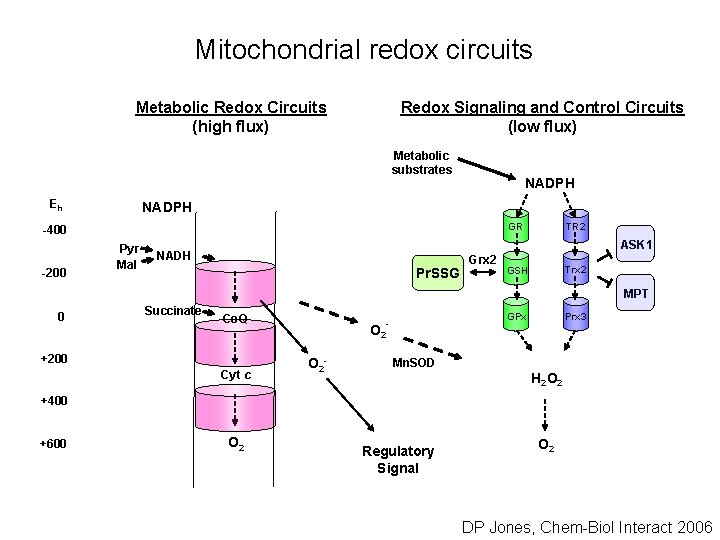
Mitochondrial redox circuits Metabolic Redox Circuits (high flux) Redox Signaling and Control Circuits (low flux) Metabolic substrates Eh NADPH GR -400 -200 Pyr Mal TR 2 ASK 1 NADH Pr. SSG Grx 2 Trx 2 GSH MPT 0 Succinate Co. Q +200 Cyt c GPx - O 2 - Prx 3 Mn. SOD H 2 O 2 +400 +600 O 2 Regulatory Signal O 2 DP Jones, Chem-Biol Interact 2006

Summary: Trx 2 in Mitochondrial Compartment 1. Mitochondrial Trx 2 has a more reduced redox state than cytoplasmic or nuclear Trx 1 or cellular GSH 2. Mitochondrial Trx 2 is more susceptible to oxidation than the cytoplasmic Trx 1 3. Redox western blot analysis of mitochondrial Trx 2 provides a useful approach to measure mitochondrial oxidative stress

GSH is difficult to measure in nuclei Cotgreave, 2003 Bellomo, 1992 Voehringer, 1998
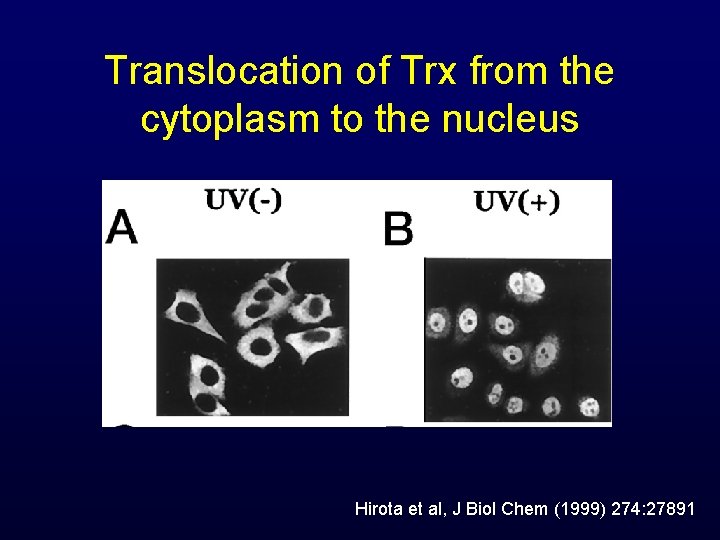
Translocation of Trx from the cytoplasm to the nucleus Hirota et al, J Biol Chem (1999) 274: 27891
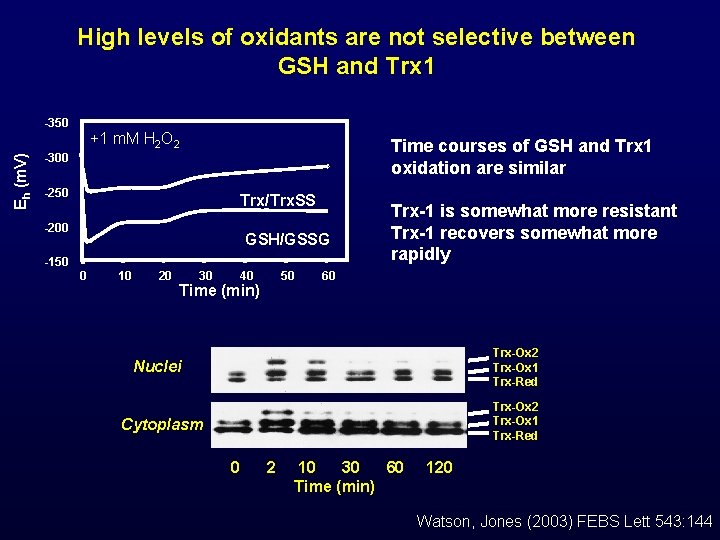
High levels of oxidants are not selective between GSH and Trx 1 Eh (m. V) -350 +1 m. M H 2 O 2 Time courses of GSH and Trx 1 oxidation are similar -300 -250 Trx/Trx. SS -200 GSH/GSSG -150 0 10 20 30 40 50 Time (min) Trx-1 is somewhat more resistant Trx-1 recovers somewhat more rapidly 60 Trx-Ox 2 Trx-Ox 1 Trx-Red Nuclei Trx-Ox 2 Trx-Ox 1 Trx-Red Cytoplasm 0 2 10 30 60 Time (min) 120 Watson, Jones (2003) FEBS Lett 543: 144
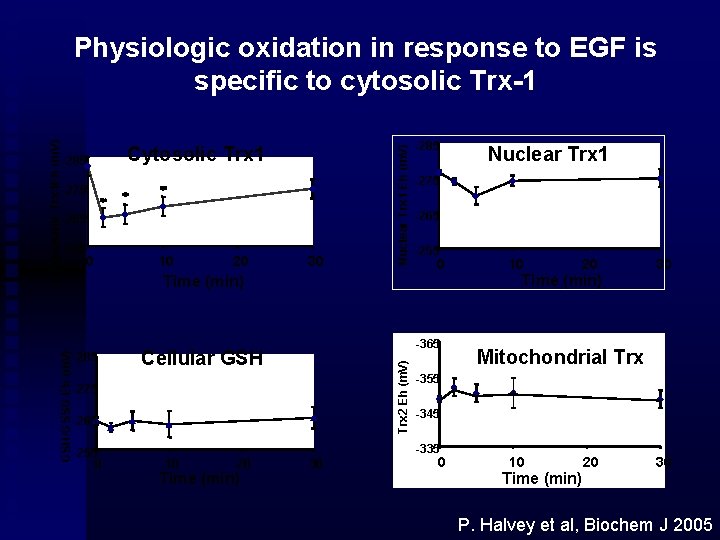
Cytosolic Trx 1 -275 -265 -255 0 10 20 30 Nuclear Trx 1 Eh (m. V) -285 -265 -255 0 -365 Cellular GSH -275 -265 -255 0 20 Time (min) 30 10 20 Time (min) 30 Mitochondrial Trx -355 -345 -335 10 Nuclear Trx 1 -275 Time (min) Trx 2 Eh (m. V) GSH/GSSG Eh (m. V) Cytoplasmic Trx 1 Eh (m. V) Physiologic oxidation in response to EGF is specific to cytosolic Trx-1 0 10 Time (min) 20 30 P. Halvey et al, Biochem J 2005

Nuclei contain less protein-SH per mg protein than cytoplasm Trx 1 and Pr. SH/Pr. SSG are more reduced in nuclei Nuclear Trx 1 and Pr. SH/Pr. SSG are more resistant to oxidation than cytoplasmic pools
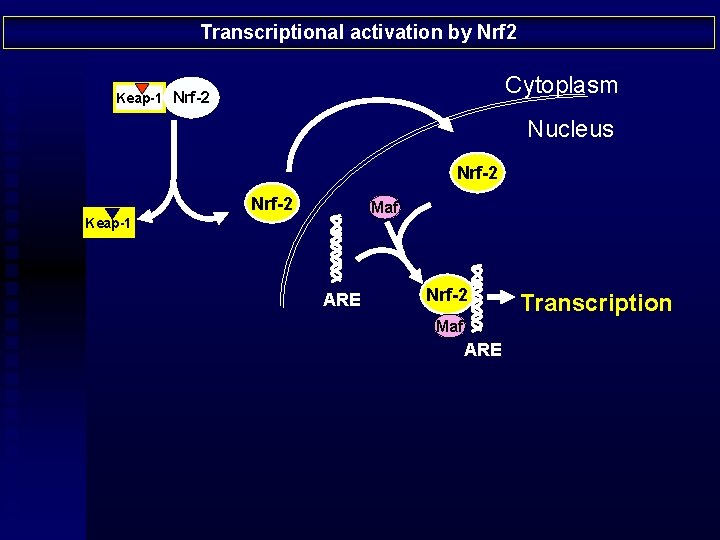
Transcriptional activation by Nrf 2 Cytoplasm Keap-1 Nrf-2 Nucleus Nrf-2 Maf Keap-1 ARE Nrf-2 Maf ARE Transcription
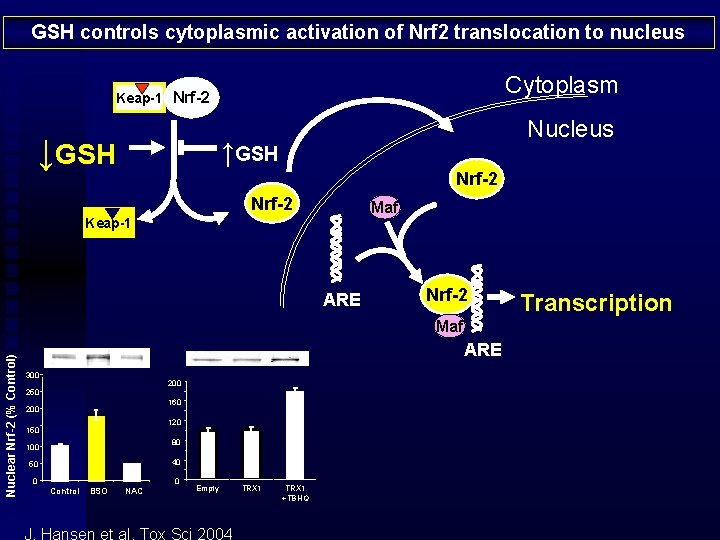
GSH controls cytoplasmic activation of Nrf 2 translocation to nucleus Cytoplasm Keap-1 Nrf-2 ↓GSH Nucleus ↑GSH Nrf-2 Maf Keap-1 ARE Nrf-2 Nuclear Nrf-2 (% Control) Maf ARE 300 250 160 200 120 150 80 100 40 50 0 Control BSO NAC 0 Empty J. Hansen et al, Tox Sci 2004 TRX 1 +TBHQ Transcription
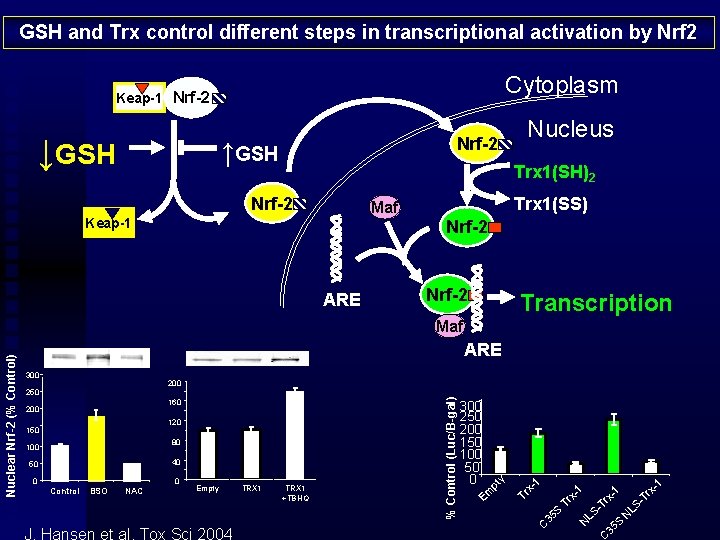
GSH and Trx control different steps in transcriptional activation by Nrf 2 Cytoplasm Keap-1 Nrf-2 ↓GSH Nrf-2 ↑GSH Nucleus Trx 1(SH)2 Nrf-2 Trx 1(SS) Maf Keap-1 Nrf-2 ARE Nrf-2 Transcription ARE 300 Tr x 1 -1 S N LS - -T rx TRX 1 +TBHQ 35 J. Hansen et al, Tox Sci 2004 TRX 1 LS Empty 1 0 N NAC Tr x 1 BSO S Control 35 0 C 40 50 Tr x- 80 100 pt 120 150 300 250 200 150 100 50 0 Em 160 200 y 200 250 % Control (Luc/B-gal) Nuclear Nrf-2 (% Control) Maf

Cytoplasmic activation of Nrf 2 is dependent upon GSH/GSSG Nuclear activity of Nrf 2 is dependent upon Trx 1
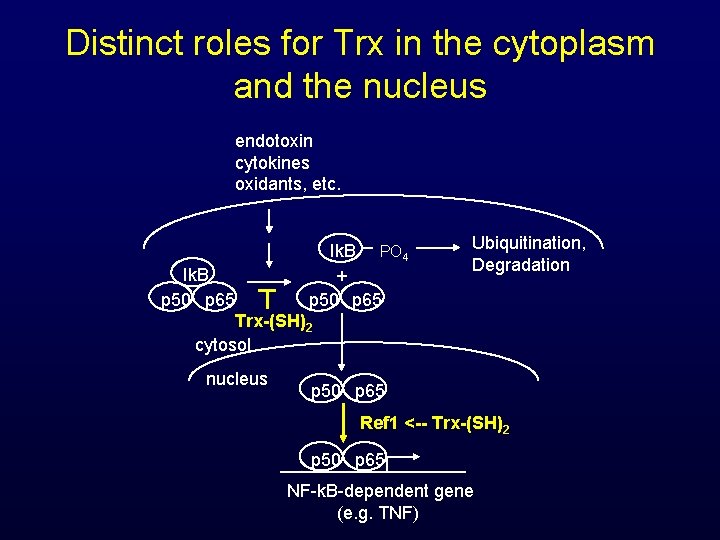
Distinct roles for Trx in the cytoplasm and the nucleus endotoxin cytokines oxidants, etc. Ik. B p 50 p 65 Ik. B PO 4 + p 50 p 65 Ubiquitination, Degradation Trx-(SH)2 cytosol nucleus p 50 p 65 Ref 1 <-- Trx-(SH)2 p 50 p 65 NF-k. B-dependent gene (e. g. TNF)
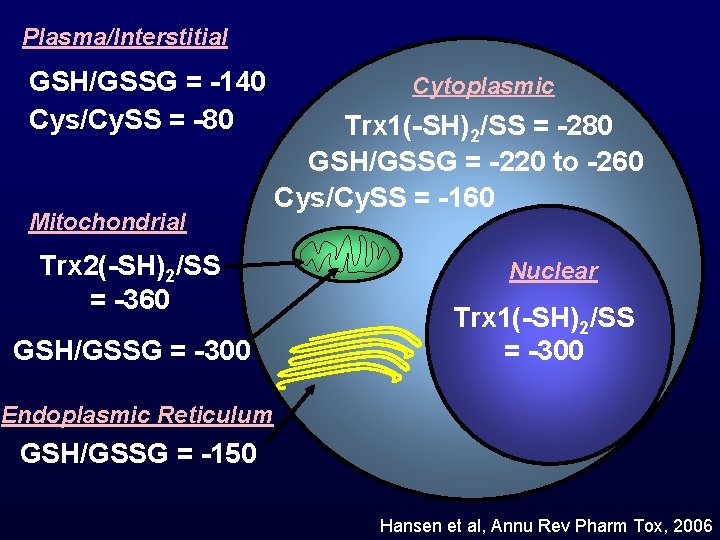
Plasma/Interstitial GSH/GSSG = -140 Cys/Cy. SS = -80 Mitochondrial Trx 2(-SH)2/SS = -360 GSH/GSSG = -300 Cytoplasmic Trx 1(-SH)2/SS = -280 GSH/GSSG = -220 to -260 Cys/Cy. SS = -160 Nuclear Trx 1(-SH)2/SS = -300 Endoplasmic Reticulum GSH/GSSG = -150 Hansen et al, Annu Rev Pharm Tox, 2006

Summary 1. Redox signaling and control involves discrete redox circuitry 2. The mitochondrial compartment is most reduced and most susceptible to oxidation 3. Nuclei are more reduced than cytoplasm and contain special mechanisms to protect against oxidative stress 4. Analytic methods are available to elucidate the redox circuitry and compartmentation of oxidative stress
- Slides: 34