Emission Sources Pollutant emission sources may be classified


Emission Sources Pollutant emission sources may be classified by: . 1 Spatial distribution 1. 1 point sources e. g. industrial chimneys 1. 2 line sources e. g. highways, airline routes 1. 3 area sources e. g. extensive urban, industrial complexes 2

. 2 Stationary and mobile sources. 3 The basis of the height of discharge 3. 1 street level 3. 2 stack level 3. 3 building level 3. 4 above the atmospheric boundary layer level 3

Types of emissions 1. Planned emissions 2. Fugitive emissions 3. Accidental emissions 4

. 1 Planned emissions arise when it is economically or technically impossible to completely remove all the contaminants in a discharge and hence the process operation allows pollutants to be discharged to the environment at known and controlled rates. . 2 Fugitive emissions arise when pollutants are released in an unplanned way, normally without first passing through the entire process. They generally originate from operations which are uneconomic or impractical to control, have poor physical arrangements for effluent control, or poorly maintained or managed. . 3 Accidental emissions result from plant failures. They can give rise to very high concentrations but they normally occur only infrequently. 5

Environmental Monitoring Strategies Objectives of monitoring -to gather information on the existence and concentration of substances in the environment, either naturally occurring or from anthropogenic sources 6

Monitoring may be carried out: . 1 To assess pollution effects on man and his environment and to identify any possible cause and effect relationship between pollutant concentrations and health effects or climate change. 2 To study and evaluate pollutant interactions and patterns 7

. 3 To assess the need for legislative controls on emissions of pollutants and ensure compliance with emission standards. 4 To active emergency procedures, in areas prone to acute pollution episodes 8
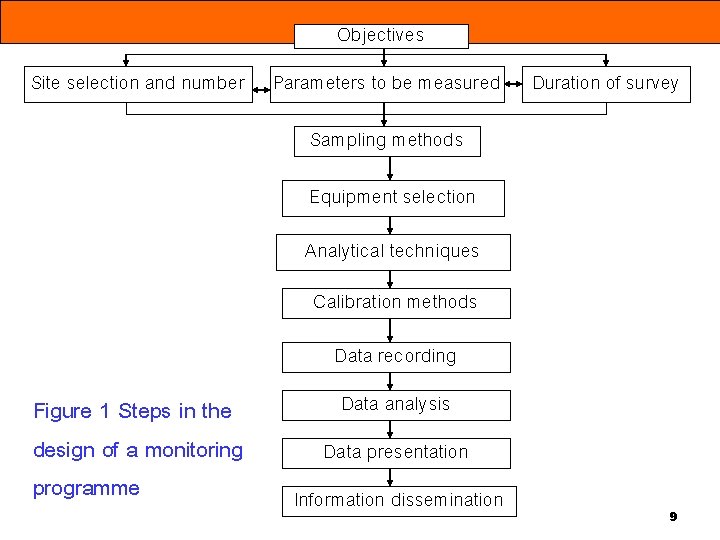
Objectives Site selection and number Parameters to be measured Duration of survey Sampling methods Equipment selection Analytical techniques Calibration methods Data recording Figure 1 Steps in the design of a monitoring programme Data analysis Data presentation Information dissemination 9

How different between air monitoring and air sampling Air monitoring n refers to methods and equipment designed to give real-time or near-immediate information on air or gas characteristics, especially pollutant concentrations Air sampling n the proper techniques to acquire a representative sample of possibly contaminated air or gas for later evaluation or analysis of the quantity of pollutant present. 10

Air sampling can be divided into 3 sampling categories: 1. การตรวจวดมลพษอากาศในบรรยากาศ (Ambient sampling) n the measurement of outdoor air pollutant level. 2. การตรวจวดมลพษอากาศในสถานประกอบการ (Industrial hygiene (workplace) air sampling) n usually indoor measurement in workplaces, either stationary or area monitoring or personal monitoring for worker protection 11

3. การตรวจวดมลพษอากาศจากปลองโรงงานอตสาหกรร ม (Source or emission sampling) n the measurement of gas flow rate, physical characteristics, and composition and pollutant concentration in exhaust gas streams leaving a process, factory, stack, or ventilation system and entering the atmosphere. 12
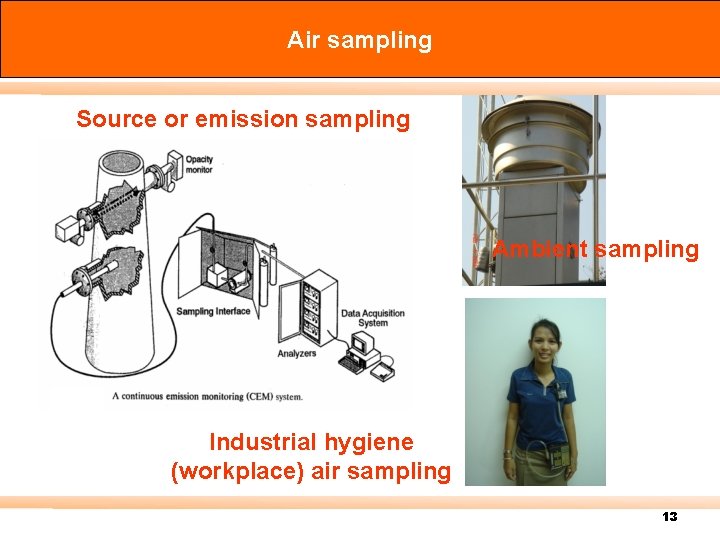
Air sampling Source or emission sampling Ambient sampling Industrial hygiene (workplace) air sampling 13

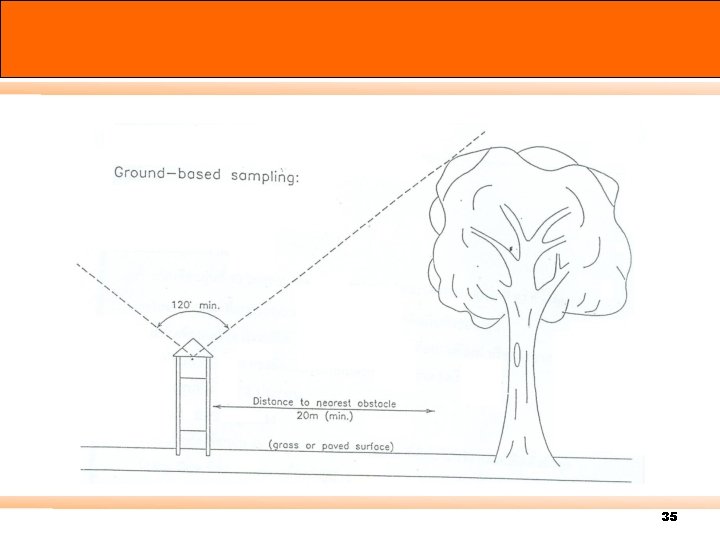
Air Sampling Method n can be divided into 2 sampling methods. 3. 1 Grab sampling 3. 2 Continuous Emission Monitoring Systems (CEMs) 14




















35

การเกบตวอยางอากาศ 1) Passive sampler 2) Active Sampler 36
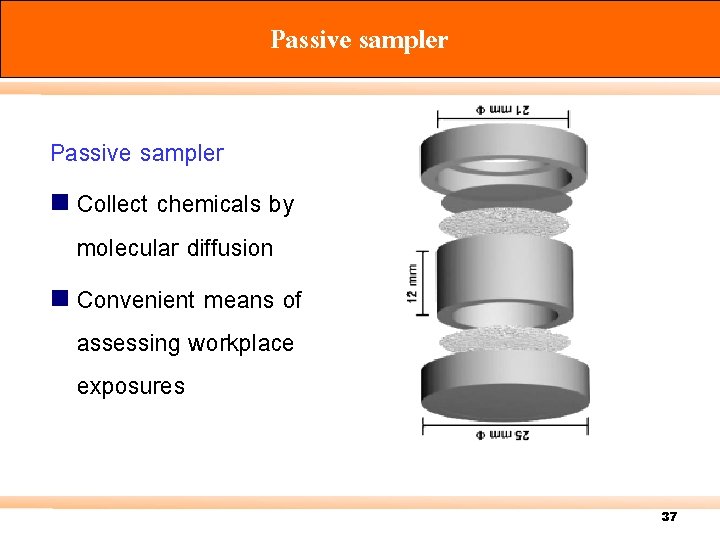
Passive sampler n Collect chemicals by molecular diffusion n Convenient means of assessing workplace exposures 37
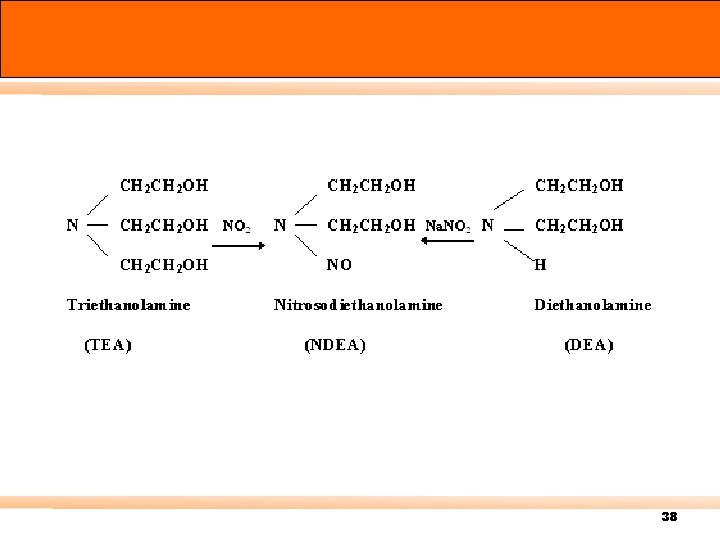
38

Nitrosodiethanolamine is easily hydrolysed to form nitrite as H 3 PO 4 R 2 N-NO + H 2 O R 2 NH + HNO 2 Therefore NO 2 in the ambient is absorbed by TEA in the form of the nitrite ion and the factor of NO 2 converted to NO 2 - is 1. 0. These NO 2 - are analyzed by using the Saltzman method. 39
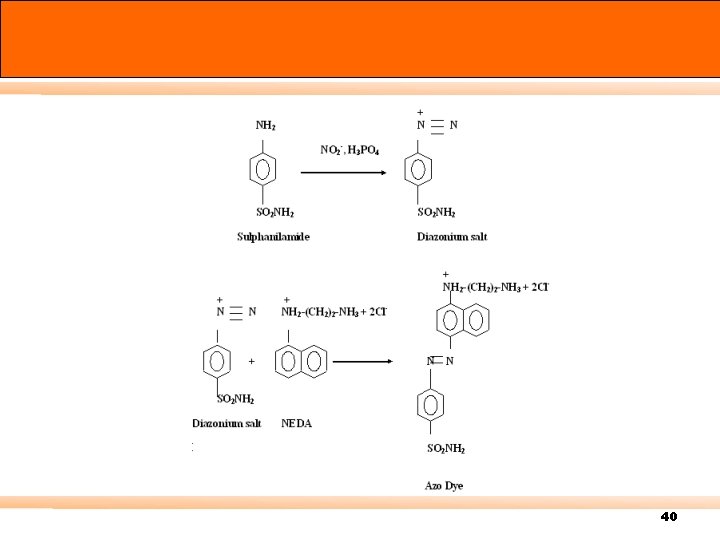
Saltzman method 40
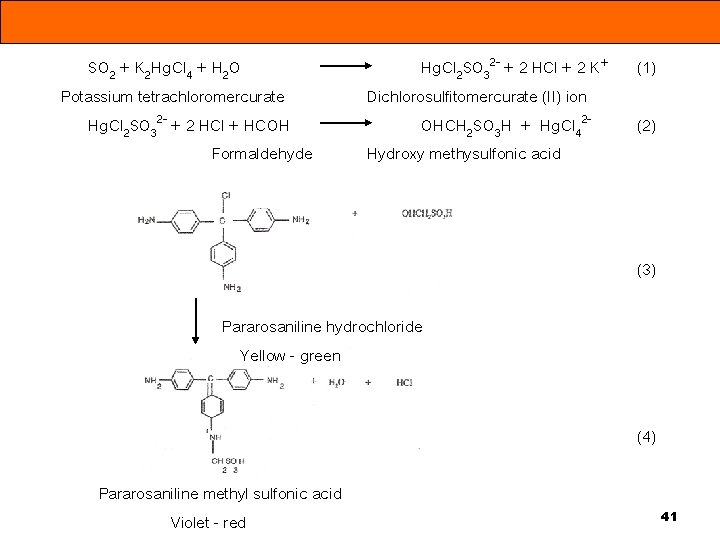
SO 2 + K 2 Hg. Cl 4 + H 2 O Potassium tetrachloromercurate Hg. Cl 2 SO 32 - + 2 HCl + HCOH Formaldehyde Hg. Cl 2 SO 32 - + 2 HCl + 2 K+ Dichlorosulfitomercurate (II) ion OHCH 2 SO 3 H + Hg. Cl 42 Hydroxy methysulfonic acid (1) (2) (3) Pararosaniline hydrochloride Yellow - green (4) Pararosaniline methyl sulfonic acid Violet - red 41

n วด NH 3 ในรป NH 4+ วด UV โดยใช Indophenol blue method 42

Fick’s law (Palmes et al, 1976) At constant temperature, the unidirectional flow of a gas 1 through a gas 2 can be described by Fick’s law J 1 = - D 12 dc 1 / dz J 1 = molal flux of gas 1 (moles/ cm 2/sec. ) D 12 = diffusion coefficient of gas 1 through gas 2 (cm 2/sec. ) c 1 = concentration of diffusing gas 1 (moles/ cm 3) z = distance in direction of diffusion (cm) 43

Q 1 = where Q 1 = A = t = c 1 -c 0 = J 1 A t = ( - D 12 (c 1 - c 0) / z ) A t quantity of gas 1 transferred (moles) cross - sectional area of tube (cm 2) time (seconds) concentration gradient along the cylinder of length z (moles/ cm 3) 44

Q 1 = ( - D 12 (c 1 - c 0) / z ) A t Q 1 = ( - D 12 c 1 / z ) r 2 t A = พท. วงกลม At one end the diffusion tube is closed and gas diffuses through the tube at the other end. If an efficient absorber is used to remove gas 1, then the concentration of gas 1 at the surface of absorber (c 0) can be assumed to be zero. The concentration of gas 1 at the side of the sampler in the diffusion tube (c 1) is equal to the concentration of gas 1 in the atmosphere. 45
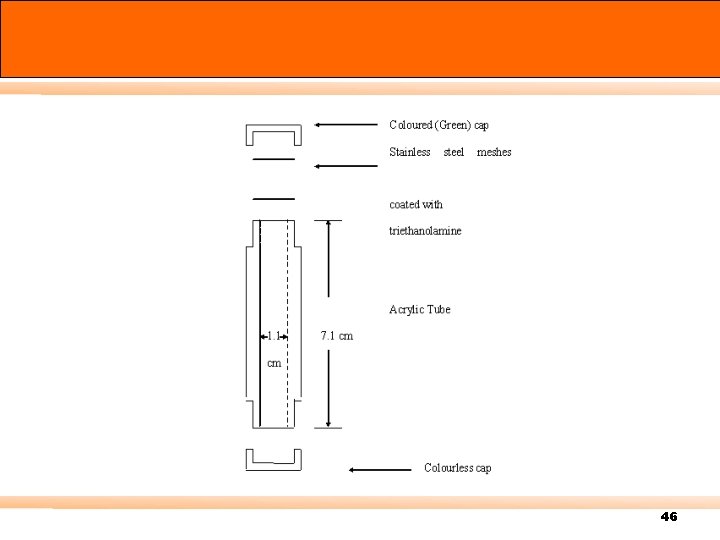
46

The diffusion coefficient for NO 2 in air (D 12) = 0. 154 cm 2/sec. Internal diameter of a diffusion tube = 1. 1 cm Length = 7. 1 cm Q = (-0. 154) c / 7. 1)(0. 55)2(3. 14) t = -0. 02 c t เปลยน time จาก seconds เปน hours Q = -0. 02 x 3600 c t = - 72 c t 47


Mathematical calculation Uptake rate = D x A / L D = Diffusion coefficient (cm 2/sec) Unique to gas/vapor being sampled A = Cross-sectional area of diffusion path (cm 2) L = Length of diffusion path (cm) Dependent on sampler geometry 49

Active sampler - Particulate Sampling & Instrumentation Total dust Respirable dust 50
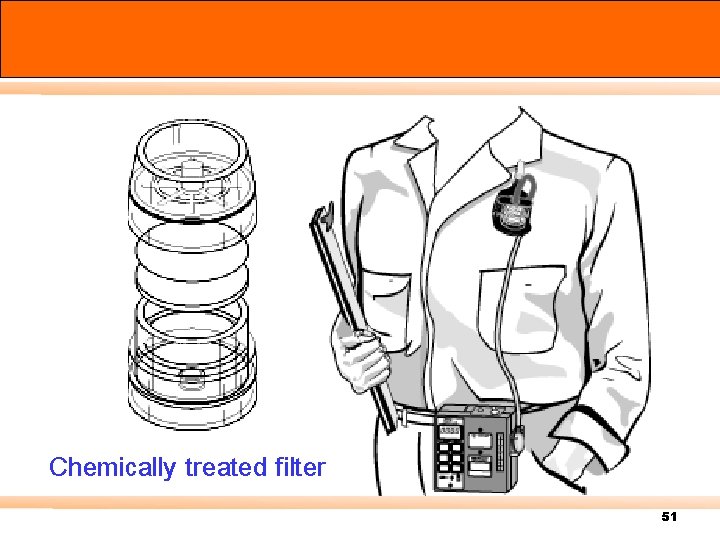
Chemically treated filter 51

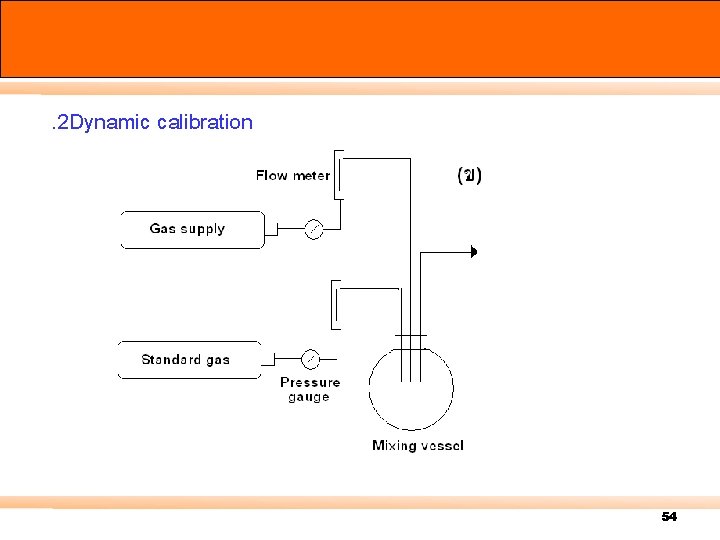
. 2 Dynamic calibration 54

การสอบเทยบเครองมอ Air or gas volume measuring devices There are three broad categories of air or gas volume measuring devices: • measure volume directly • measure rate of gas flow • measure velocity of gas Q = V/t where Q = the average flow rate V = the volume of gas t = the time period 55


Calibration There are three types of standard meter: Primary standard – Devices for which measuring volume can be accurately determined by measurement of internal dimensions alone (accuracy 0. 3% or more) e. g. spirometer, displacement bottle, soap-bubble meter, mercury-sealed piston. Intermediate standard – Devices that are more versatile than primary standards, but for which physical dimensions cannot be easily measured (accuracy 1 to 2%) e. g. Roots meter, Wet test meter (WTM), Dry gas meter (DGM). Secondary standard – typically more portable, rugged, and versatile (accuracy 5% or better) 57

Primary Standard Devices 58
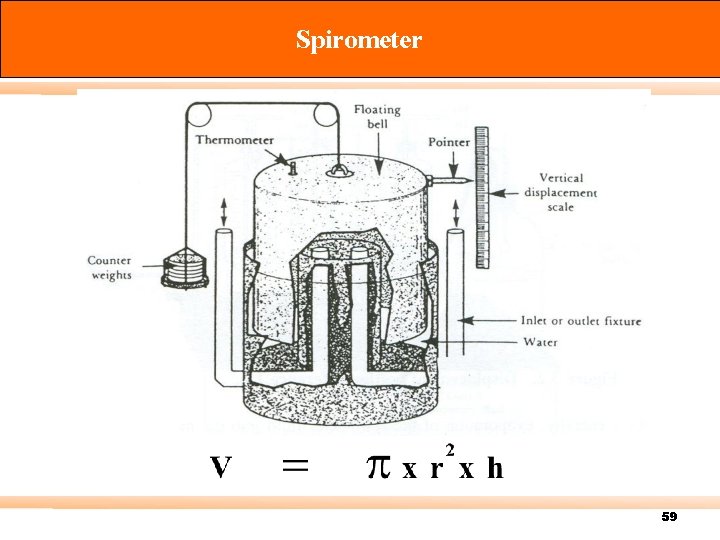
Spirometer 59
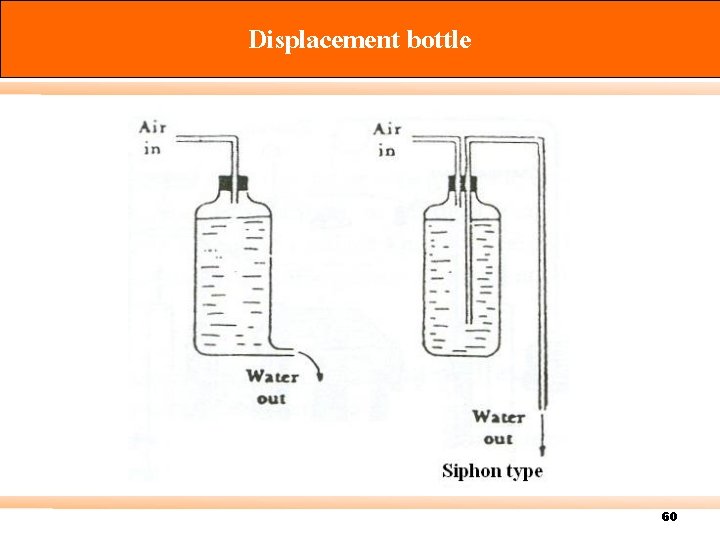
Displacement bottle 60
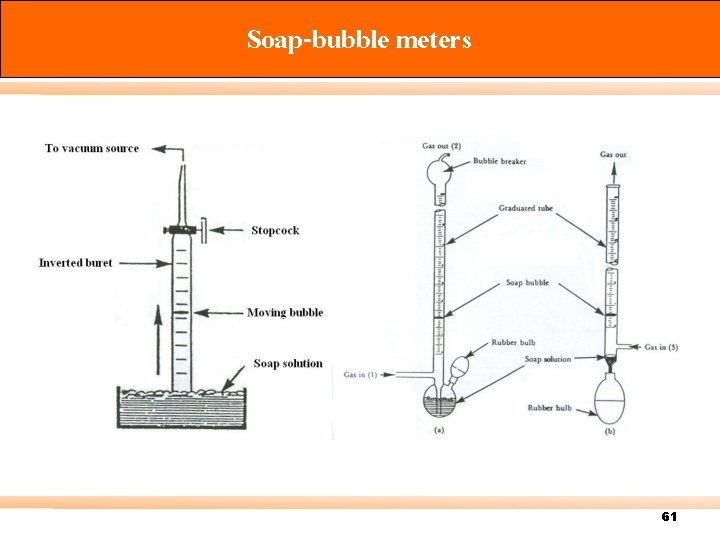
Soap-bubble meters 61
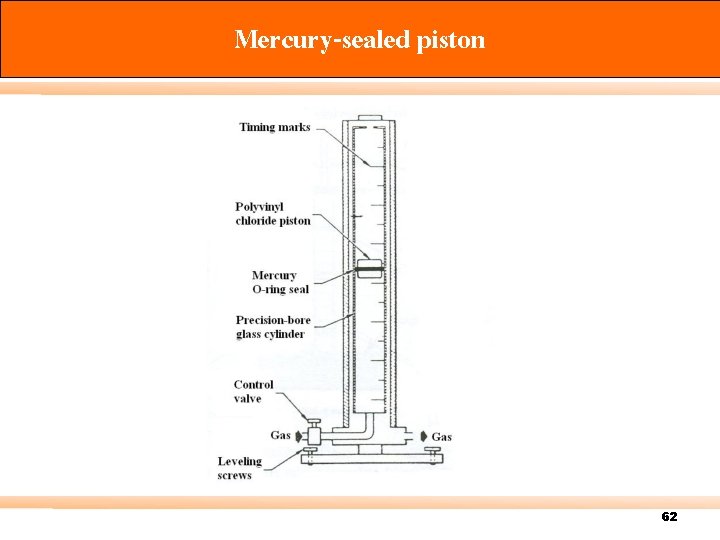
Mercury-sealed piston 62

Intermediate Standard Devices 63
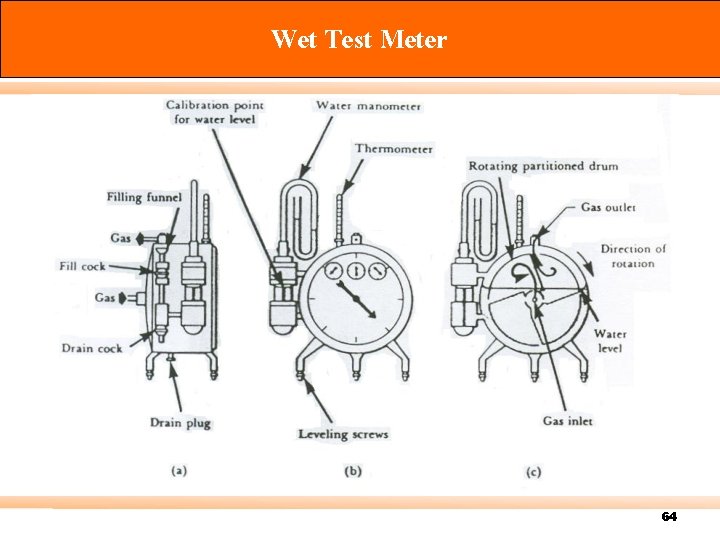
Wet Test Meter 64

Secondary Standard Devices 65

Dry Test Meter 66


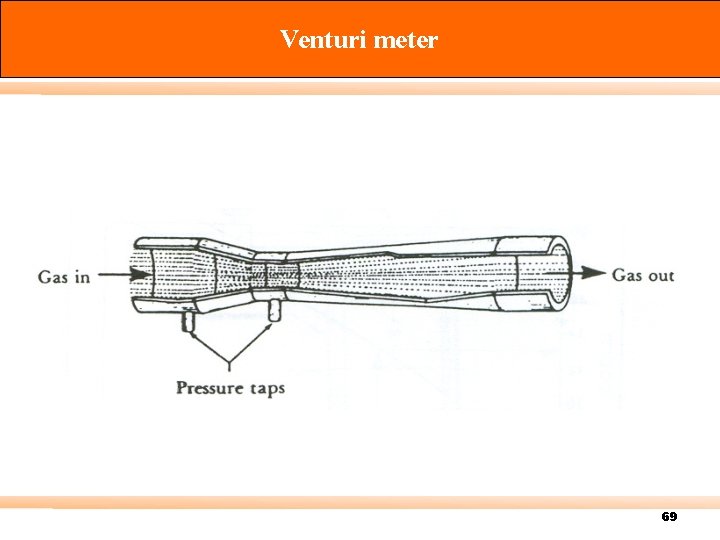
Venturi meter 69
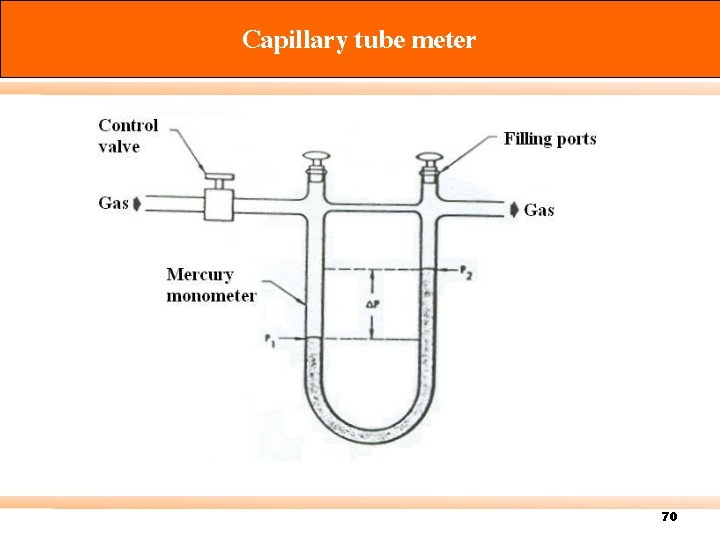
Capillary tube meter 70
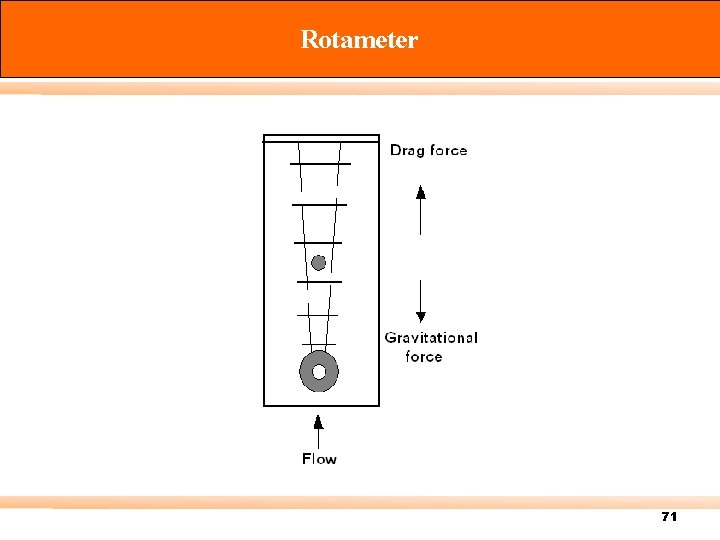
Rotameter 71

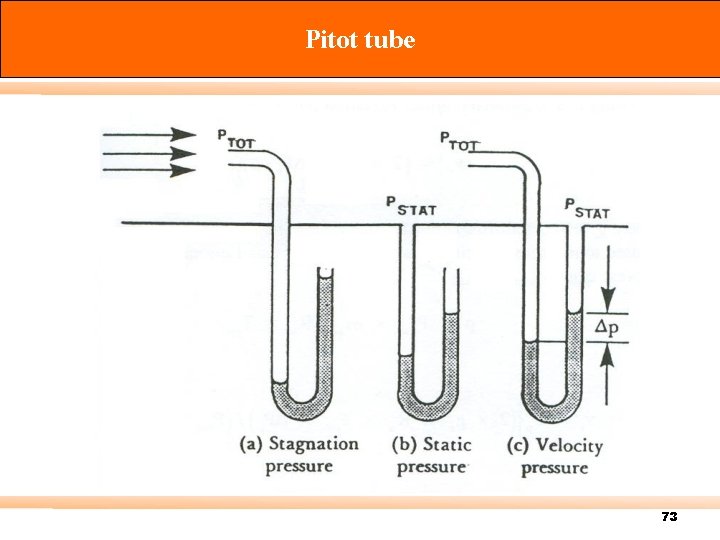
Pitot tube 73
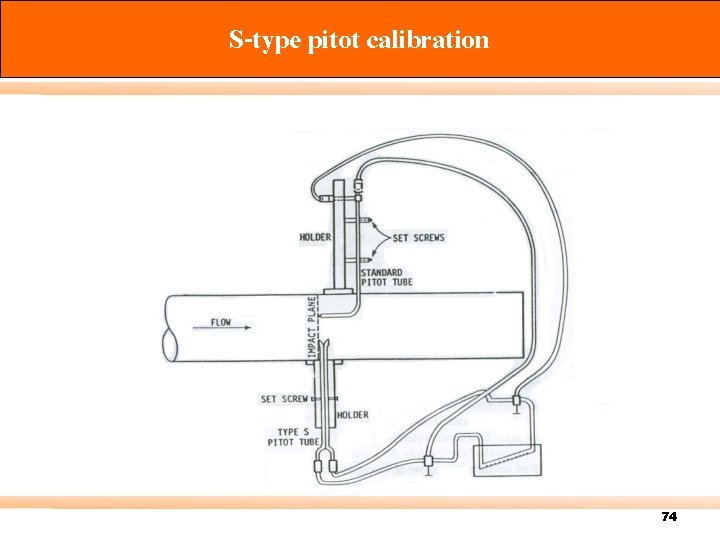
S-type pitot calibration 74
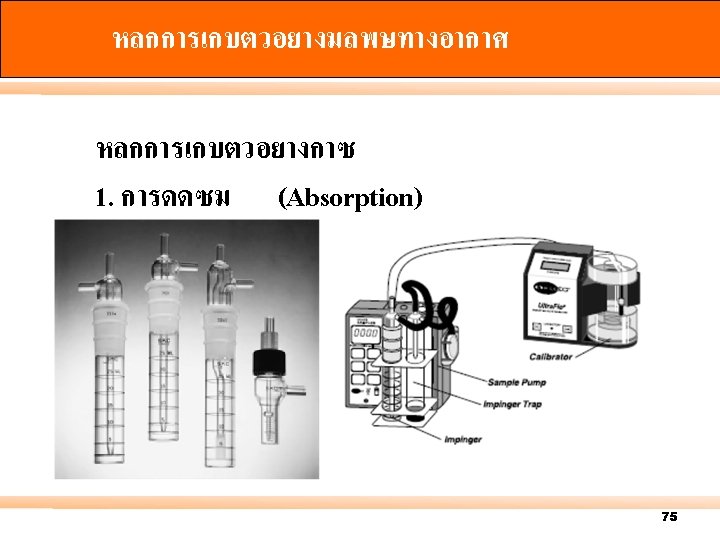

2. การดดซบ (Adsorption) 76

Cold Trap for Thermal Desorption 77





Thermal Desoption-GC-MS 82

Scott Specialty Gas Standards 83
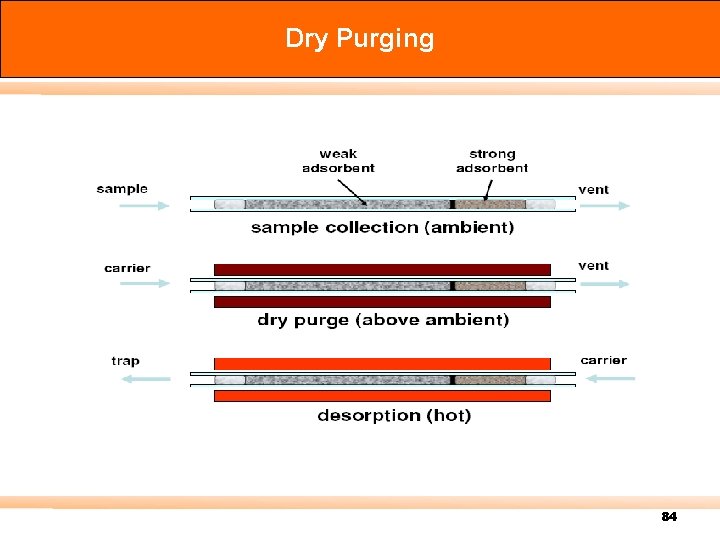
Dry Purging 84
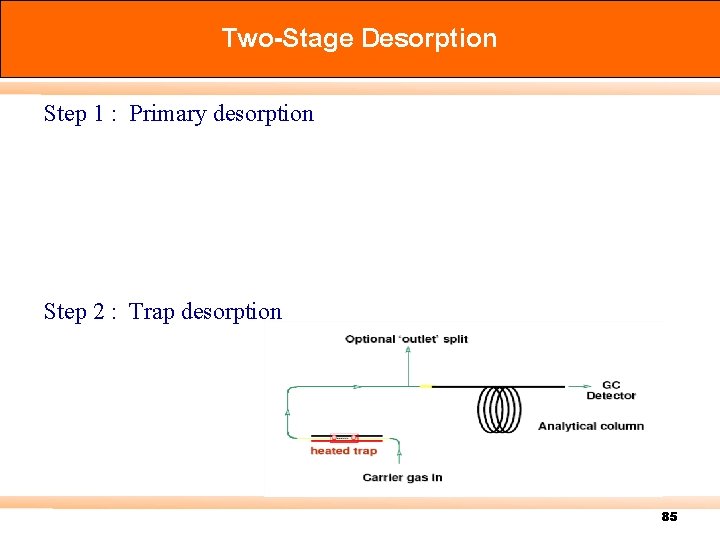
Two-Stage Desorption Step 1 : Primary desorption Step 2 : Trap desorption 85

86
3. การควบแนน (Condensation หรอ Cryogenic Sampling) 87

4. Grab Sampling Gas and vapor collection Tedlar bag Canister 88

Canister การเกบตวอยางอากา ศ โดยใช Canister n Grab sampling n 24 hour air sampling 89

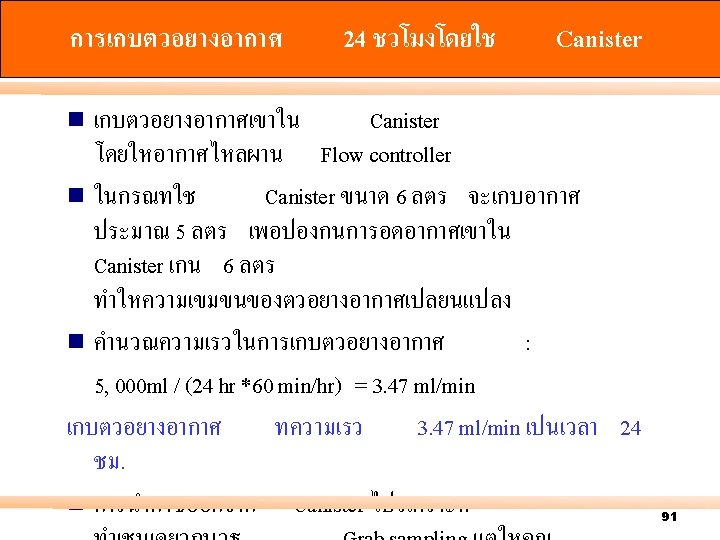


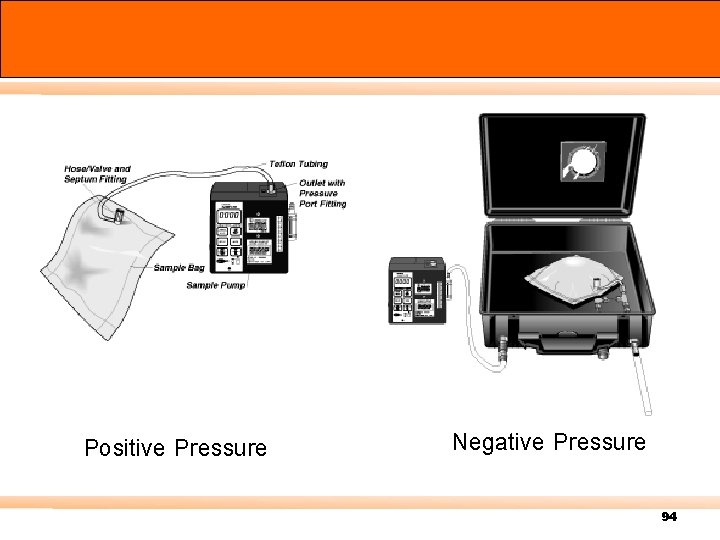
Positive Pressure Negative Pressure 94

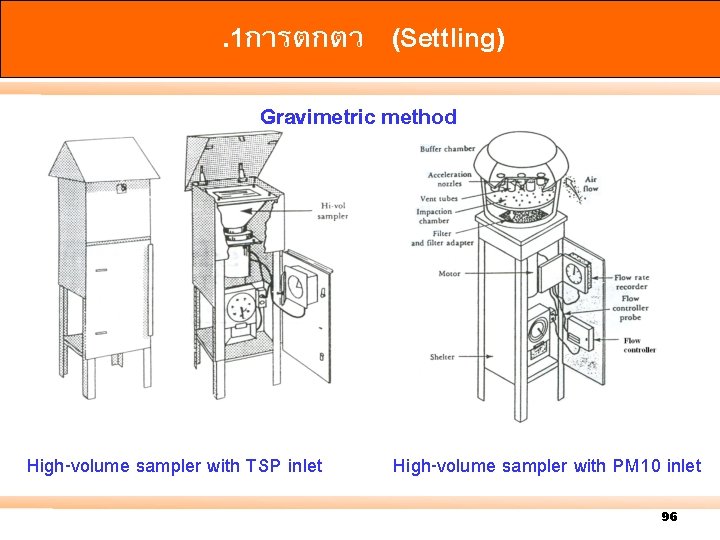
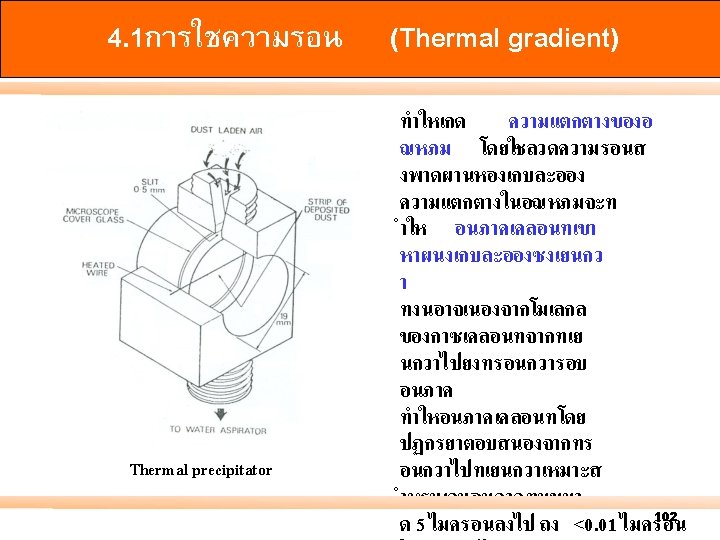
. 1การตกตว (Settling) Gravimetric method High-volume sampler with TSP inlet High-volume sampler with PM 10 inlet 96



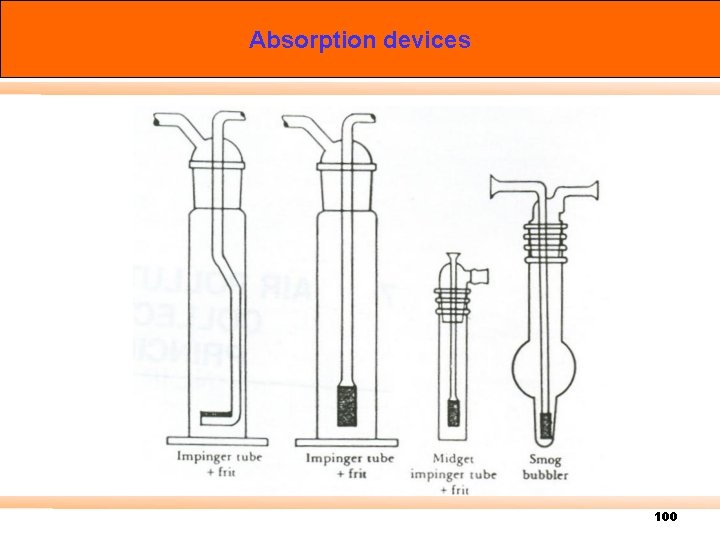
Absorption devices 100

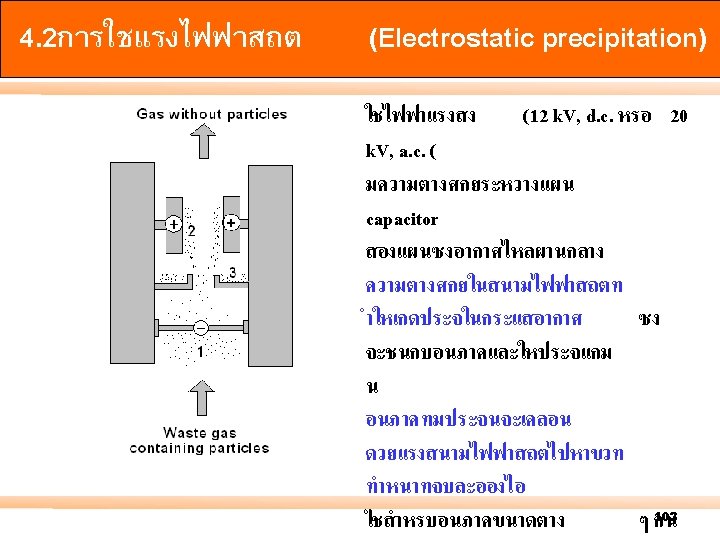


- Slides: 105