Emil Fischers proof of the structure of glucose



- Slides: 13

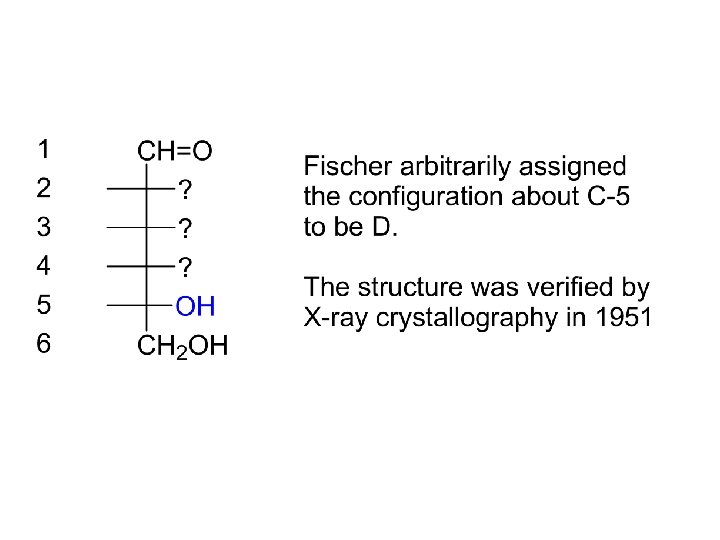
Emil Fischer’s proof of the structure of glucose. 1891 (Nobel Prize 1902) (+)-Glucose is an aldohexose * * CH 2 -CH-CH-CH=O OH OH OH Four chiral centers 24 = 16 stereoisomers


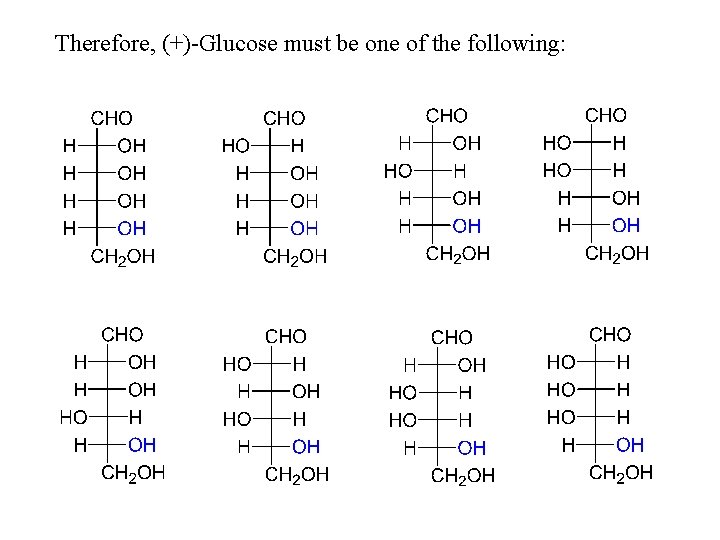
Therefore, (+)-Glucose must be one of the following:

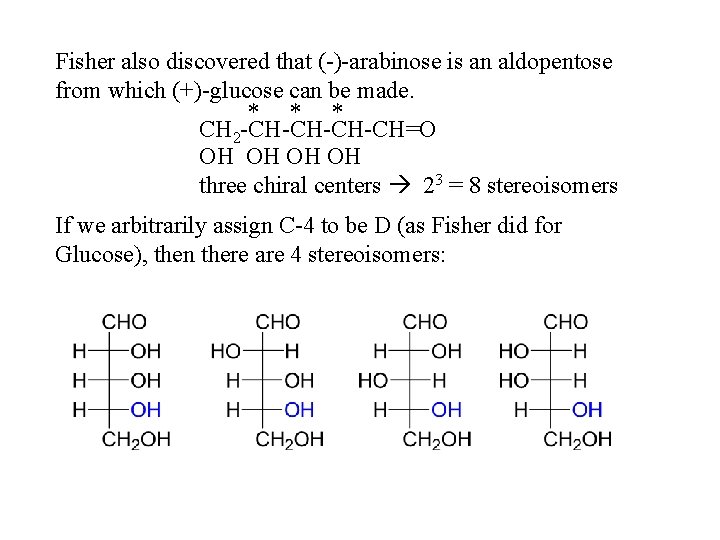
Fisher also discovered that (-)-arabinose is an aldopentose from which (+)-glucose can be made. * * * CH 2 -CH-CH=O OH OH three chiral centers 23 = 8 stereoisomers If we arbitrarily assign C-4 to be D (as Fisher did for Glucose), then there are 4 stereoisomers:

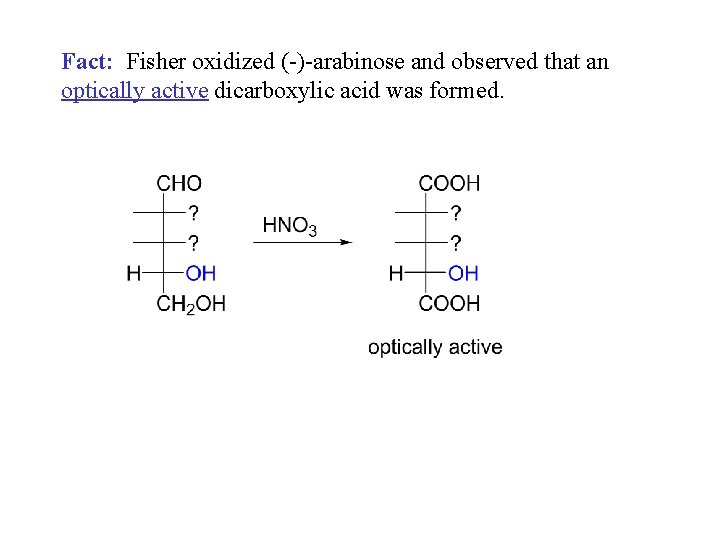
Fact: Fisher oxidized (-)-arabinose and observed that an optically active dicarboxylic acid was formed.

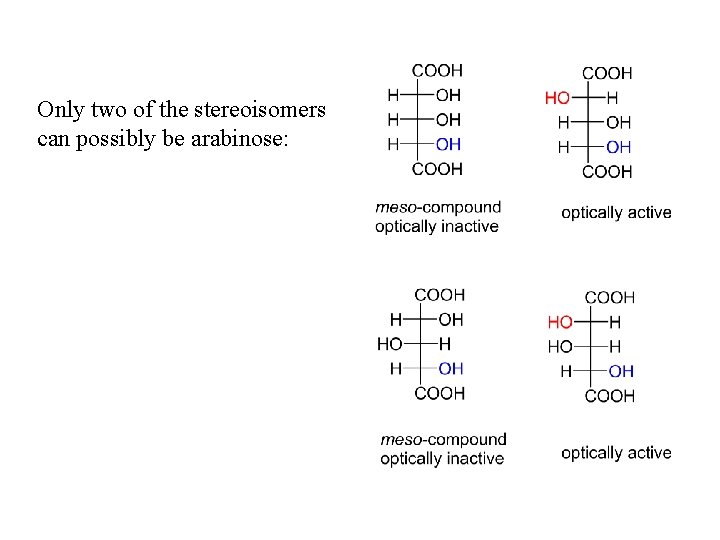
Only two of the stereoisomers can possibly be arabinose:

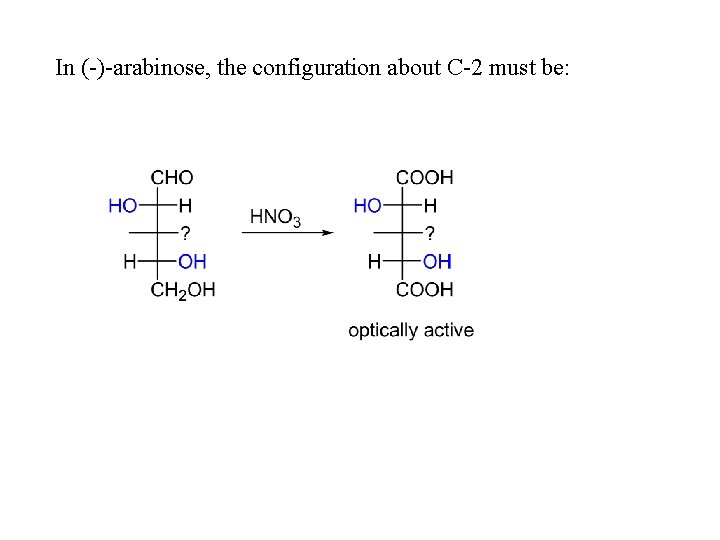
In (-)-arabinose, the configuration about C-2 must be:

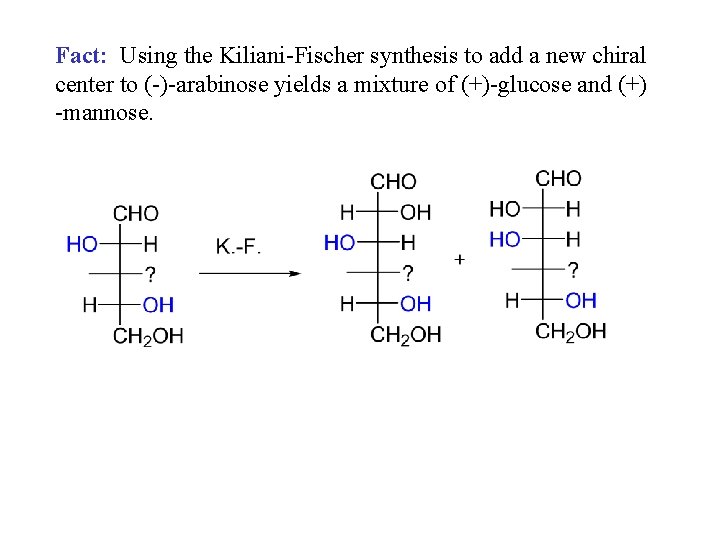
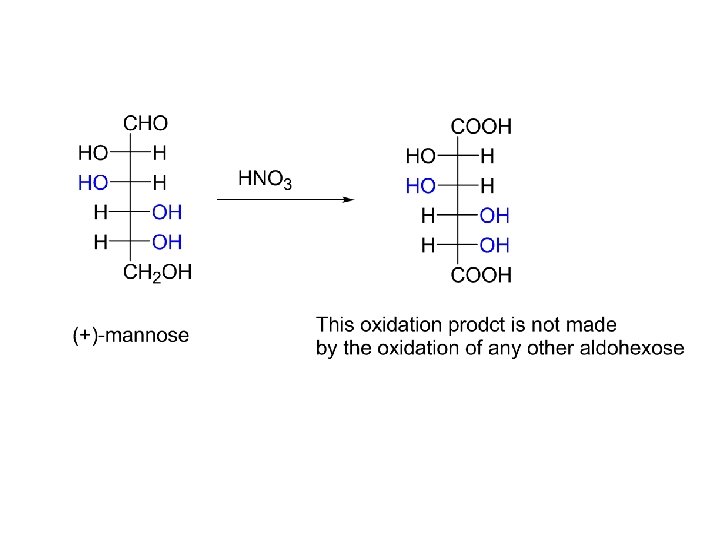
Fact: Using the Kiliani-Fischer synthesis to add a new chiral center to (-)-arabinose yields a mixture of (+)-glucose and (+) -mannose.

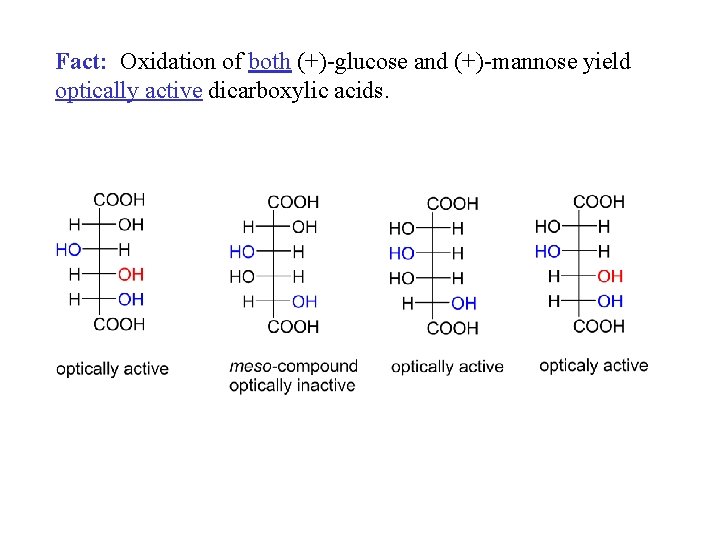
Fact: Oxidation of both (+)-glucose and (+)-mannose yield optically active dicarboxylic acids.

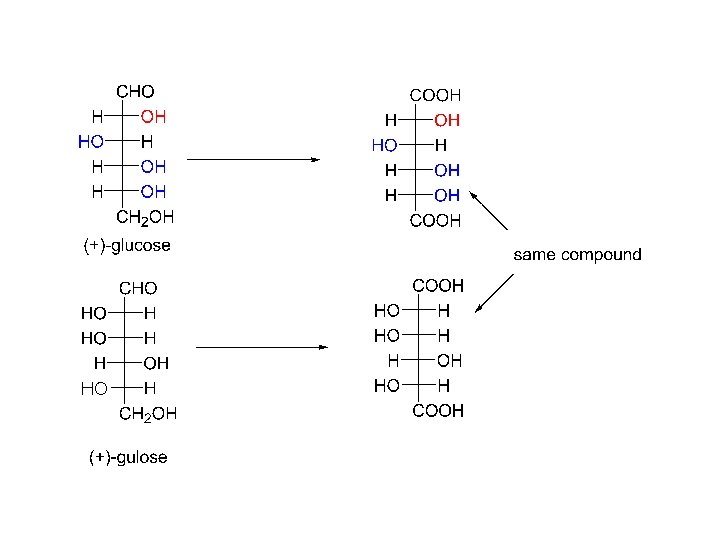
The only way that both (+)-glucose and (+)-mannose can give optically active dicarboxylic acids upon oxidation is if the configuration about C-4 is with the –OH on the right in the Fischer projection. But, which compound is (+)-glucose and which one is (+) -mannose? Fact: Oxidation of both (+)-glucose and (+)-gulose yield the same dicarboxylic acid. Fact: The dicarboxylic acid produced from (+)-mannose is not made by the oxidation of any other aldohexose.


HO

Therefore, (+)-glucose has the following configuration: C-5 was assigned arbitrarily by Fischer. C-3 is known from the observation that the oxidation product of (-) arabinose is optically active; and (+)-glucose and (+) -mannose can be made from (-)arabinose by the Kiliani-Fischer synthesis. C-4 is known from the fact that both (+) -glucose and (+)-mannose yield optically active dicarboxylic acids. C-2 is known from the fact that both (+) -glucose and (+)-gulose yield the same dicarboxylic acid.