Emerging trends in Synthesis involving Dimethyl Sulfoxide DMSO






![Things you might not know An ‘x’ is given when both [DAS] and [H Things you might not know An ‘x’ is given when both [DAS] and [H](https://slidetodoc.com/presentation_image_h/a61b01d8886c85a493626fd86ac4180c/image-11.jpg)



- Slides: 15

Emerging trends in Synthesis involving Dimethyl Sulfoxide DMSO ‘Tricks and Tips’ Gaylord Chemical Company L. L. C Preeminent Manufacturer of Dimethyl Sulfoxide (DMSO) Dimethyl Sulfide (DMS) and Procipient® www. Gaylord. Chemical. com Debra D. Dolliver, University of Alabama Hashim Javaid, IMCD Group Artie Mc. Kim, Gaylord Chemical Company Copyright 2016 Gaylord Chemical Company LLC

Who we are IMCD Group: • Gaylord’s exclusive Pan European distribution • Dedicated, market focused expertise Gaylord Chemical Company • Focused thiochemical producer since 1961 How is DMSO made? www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC

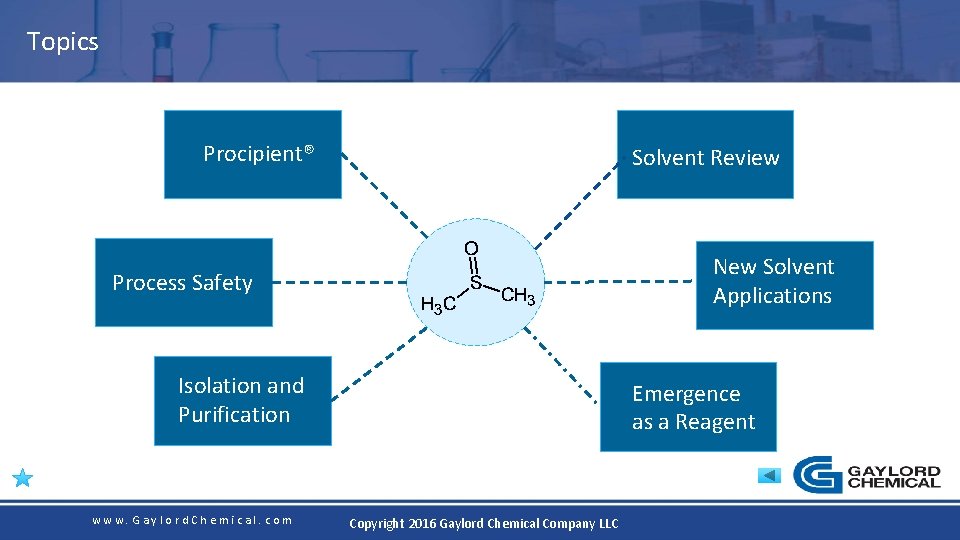
Topics Procipient® Solvent Review New Solvent Applications Process Safety Isolation and Purification www. Gaylord. Chemical. com Emergence as a Reagent Copyright 2016 Gaylord Chemical Company LLC

Solvent Review: The status of DAS The emerging consensus on dipolar aprotic solvent use is that: • They are here to stay! • The amide solvents pose significant health and regulatory risks • DMSO, while less toxic, can be reactive. ‘Solvent Guides’ for Discovery Groups largely Agree: • • GSK: Reactivity an issue; a preferred DAS for Safety, Health Sanofi: Compatibility, odor concerns. Pfizer: Only DMSO and Ac. N in the ‘usable’ category ACS Green Chemistry Institute: waste handling DMSO Tip: Low solubility for oxygen in DMSO can provide benefits air sensitive reactions, such as transition metal cross-couplings www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC Org. Process. Res. Dev. 2015, 19, 740747 Green Chem. , 2011, 13, 854 -862. J. Org. Process Res. Dev, 2013, 17, 1517 -1525 Green Chem. , 2008, 10, 31 -36 www. acs. org/gcipharmaroundtable Papageorgiou, C. D. OPRD presentation 2016

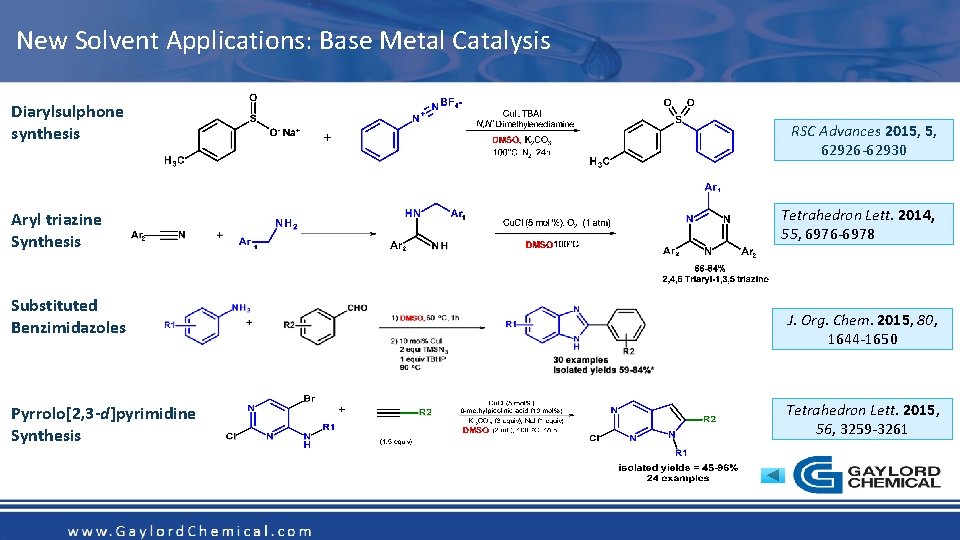
New Solvent Applications: Base Metal Catalysis Diarylsulphone synthesis RSC Advances 2015, 5, 62926 -62930 Tetrahedron Lett. 2014, 55, 6976 -6978 Aryl triazine Synthesis Substituted Benzimidazoles J. Org. Chem. 2015, 80, 1644 -1650 Tetrahedron Lett. 2015, 56, 3259 -3261 Pyrrolo[2, 3 -d]pyrimidine Synthesis www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC

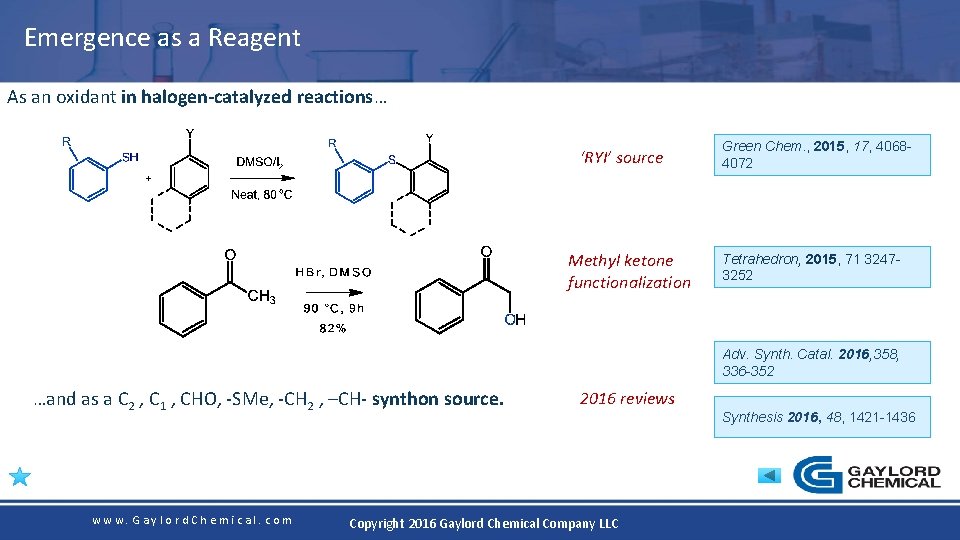
Emergence as a Reagent As an oxidant in halogen-catalyzed reactions… ‘RYI’ source Methyl ketone functionalization Green Chem. , 2015, 17, 40684072 Tetrahedron, 2015, 71 32473252 Adv. Synth. Catal. 2016, 358, 336 -352 …and as a C 2 , C 1 , CHO, -SMe, -CH 2 , –CH- synthon source. www. Gaylord. Chemical. com 2016 reviews Copyright 2016 Gaylord Chemical Company LLC Synthesis 2016, 48, 1421 -1436

Isolation and Purification Workup protocols for DMSO For nonpolar products in DMSO For polar products in DMSO New impurity Rule of Thumb For 5 m. L of DMSO use: Cold Diethyl ether (5 x 5 ml) Then wash organic phases 5 x 15 ml of water to wash out all DMSO Dry – Organic phase & Evaporate Final ‘plug wash’ through silica gel for 100% DMSO removal. DMSO Pressure distillation / Reduced New impurity concentration. New impurity Followed extraction into MTBE, water washes. Final ‘plug wash’ silica gel to After second Afterthrough first cycle (~ 10 L) (~20 L) for 100% DMSO removal. [impurity] = 237 [impurity] = 124 Starting material (~ 40 L) [impurity] = 58 ppm DMSO Tip: When dry, DMSO has an avidity for aromatic hydrocarbons over aliphatics. www. Gaylord. Chemical. com New impurity Copyright 2016 Gaylord Chemical Company LLC ppm After third cycle (~ 1 L) [impurity] = 1408 ppm

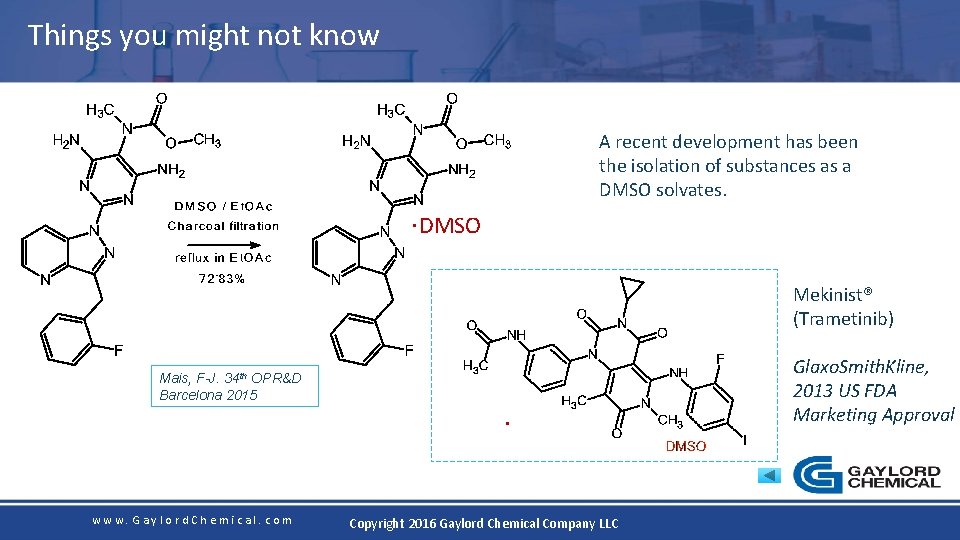
Things you might not know A recent development has been the isolation of substances as a DMSO solvates. . DMSO Mekinist® (Trametinib) Mais, F-J. 34 th OPR&D Barcelona 2015 www. Gaylord. Chemical. com . Copyright 2016 Gaylord Chemical Company LLC Glaxo. Smith. Kline, 2013 US FDA Marketing Approval

Things you might not know DMSO as a reaction solvent was compared to other dipolar aprotic solvents (DAS) in extractive work up to determine: DAS 1. How much of the DAS ends up in the extraction solvent? 2. How much water ends up in the extraction solvent? DMSO DMF NMP DMAc TMU DMI 1, 4 -dioxane diglyme acetonitrile Extraction solvent Aqueous solvent Toluene Ac. OEt Ac. Oi. Pr 1 -chlorobutane heptanes H 2 O Na. Cl/H 20 (10 cg/g) Organic Process Research & Development 2007, 11, 160 -164 Delhaye, L. ; Ceccato, A. ; Jacobs, P. ; Kottgen, C. ; Merschaert, A. Organic Process Research & Development 2007, 11, 160 -164 www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC

Experimental details Equal volumes of DAS, extraction solvent, and aqueous solvent were stirred at 23 o. C for 45 minutes. Organic and aqueous layers were separated. Volumes and densities of both layers determined. Water in the extraction solvent was measured by Karl Fischer titration. DAS in the extraction solvent was measured by GC-FID. Delhaye, L. ; Ceccato, A. ; Jacobs, P. ; Kottgen, C. ; Merschaert, A. Organic Process Research & Development 2007, 11, 160 -164 www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC
![Things you might not know An x is given when both DAS and H Things you might not know An ‘x’ is given when both [DAS] and [H](https://slidetodoc.com/presentation_image_h/a61b01d8886c85a493626fd86ac4180c/image-11.jpg)
Things you might not know An ‘x’ is given when both [DAS] and [H 2 O] are < 10 cg / g in the extraction solvent toluene Ac. OEt Ac. Oi. Pr 1 -Cl-butane heptanes DMSO X X X DMF X X NMP X X X DMAc X X X X TMU DMI THF 1, 4 -dioxane diglyme acetonitile X DMSO Tip: There actually some advantages to DMSO use in LLE. Delhaye, L. ; Ceccato, A. ; Jacobs, P. ; Kottgen, C. ; Merschaert, A. Organic Process Research & Development 2007, 11, 160 -164 www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC

Procipient® (Dimethyl Sulfoxide USP, Ph. Eur) DMSO of extremely high purity achieved through Patented Procipient® Technology Regulatory Details Water-clear and Odorless Conforms to USP and the European Pharmacopoeia (Ph. Eur. ) Contained in registered products- N. America and Europe – Excipient and Active www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC Precedence of Use: Topical Drugs Precedence of Use: Parenteral Drugs

Process Safety There is an increased appreciation of process safety considerations under certain conditions. Appropriate hazard studies should always be done when scaling up reactions using DMSO. General Summary Historical review: Industrial Accidents OPRD 2012, San Francisco meeting J. Therm Anal Calorim. 2006, 1, 25 -30 Org. Process Res. Dev. 2014, 1836 -1842 Org. Process Res. Dev. 2012, 16, 1994 -2000 General Summary J. Therm. Anal. Calorim. 2015, 121, 295 -301 halogens High / low p. H Thermal decomposition Miscellaneous www. Gaylord. Chemical. com Org. Process Res. Dev. 2005, 9, 1003 -1008 J. Therm. Anal. Calorim. (2014) 188: 793 -399 Copyright 2016 Gaylord Chemical Company LLC AIChe 2015 11 th Global Congress on Process Safety 2015 Org. Process Res. Dev. 2006, 10, 1299 -1312

Recommendations for process safety Always distill under vacuum to minimize still temperature. Always cut off heating if vacuum source fails. Avoid distillation in the presence of halides (particularly Br). Avoid distillation at low p. H. Stop distillation if pot temperature exceeds 160°C. Prepare Dimsyl ion solution using Na. H (instead of Na). Avoid strong oxidants in combination with DMSO (POCl 3, thionyl chloride, acid chlorides, perchloric acid, acid anhydrides) Perform process safety testing: DSC, ARC, RADEX www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC

Thank you! Hashim Javaid, Ph. D International Technical Manager Regulated Synthesis IMCD Group Hashim. Javaid@imcd. co. uk Artie S. Mc. Kim, Ph. D Vice President, Technology Gaylord Chemical Company LLC amckim@gaylordchem. com Debra D. Dolliver, Ph. D University of Alabama Tuscaloosa, AL, USA Visit the DMSO ‘Synthesis Corner’ www. gaylordchem. com These materials comprise a review of the recent chemical literature. It was assembled for the educational purposes and is being shared as a service to others. www. Gaylord. Chemical. com Copyright 2016 Gaylord Chemical Company LLC