Emerging Percutaneous Treatment of Discogenic LBP AAPMR 2015

Emerging Percutaneous Treatment of Discogenic LBP AAPMR 2015 Annual Assembly Boston, MA

Disclosures • Co-investigator: Mesoblast; Spinal Restoration; ATRM/Depuy; Stryker Biotech; St Jude Medical; NIH funded LSS/ESI trial; SI Bone; Vertiflex; Halyard • SIS AUC Comm; NASS Clinical Guidelines Comm • Spine Section co-editor, Pain Medicine • Consultant: Vertiflex; Zyga; Biobridges • CAB: Mesoblast; Medtronic; Halyard

Disclosures • Discussed technologies not FDA approved

Michael J De. Palma, MD President, Medical Director, Interventional Spine Care Fellowship Virginia i. Spine Physicians, PC President, Chairman, Director of Research Virginia Spine Research Institute, Inc President, Director i. Spine Ingenuity, Inc

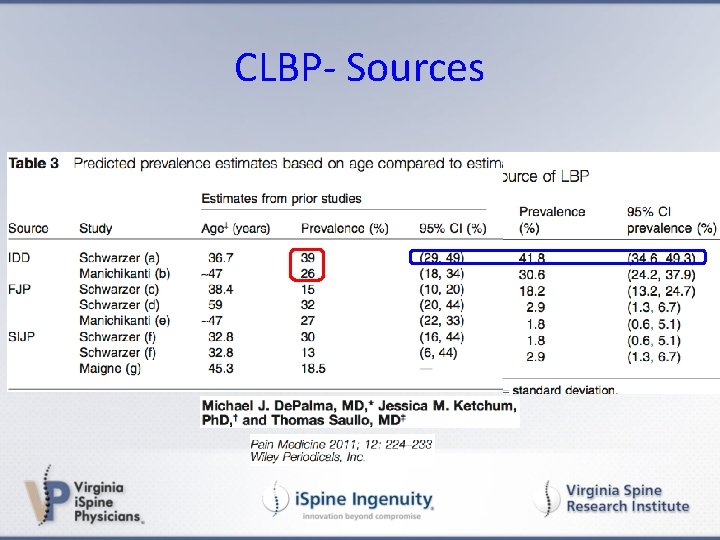
CLBP- Sources
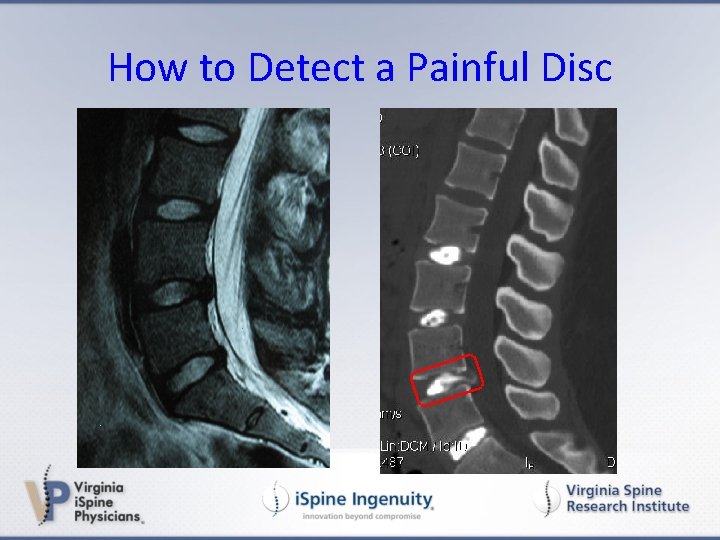
How to Detect a Painful Disc
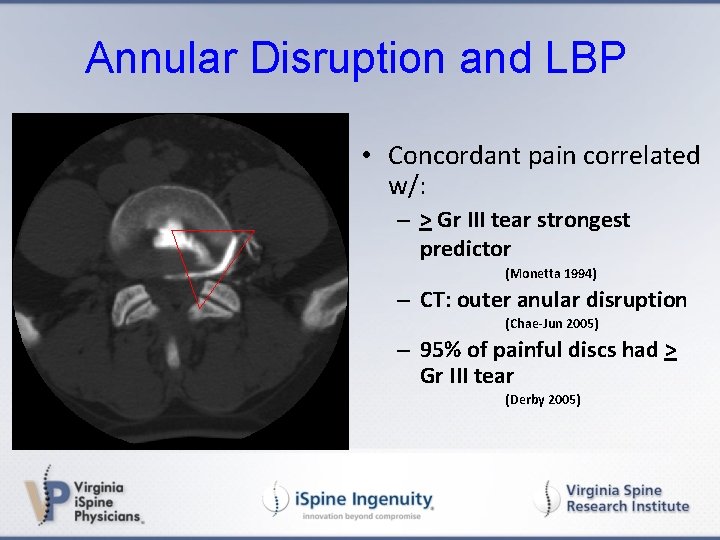
Annular Disruption and LBP • Concordant pain correlated w/: – > Gr III tear strongest predictor (Monetta 1994) – CT: outer anular disruption (Chae-Jun 2005) – 95% of painful discs had > Gr III tear (Derby 2005)

Anesthesia of Painful Annular Fissures De. Palma Pain Med 2009
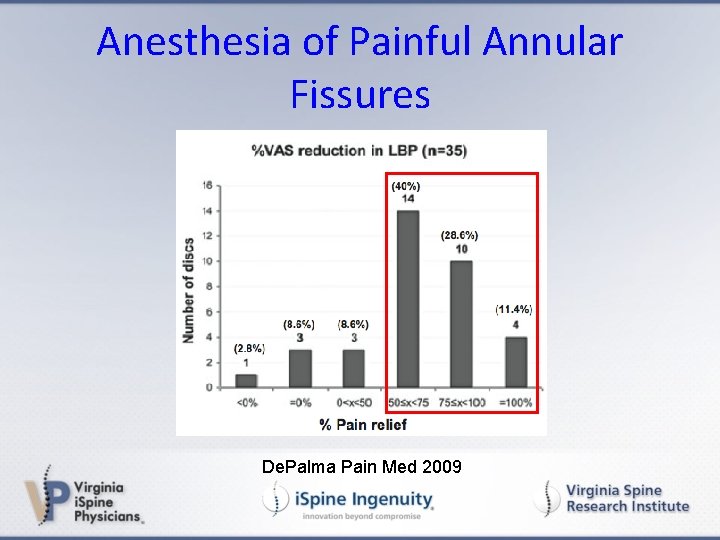
Anesthesia of Painful Annular Fissures De. Palma Pain Med 2009

The Intervertebral Disc is a Common Source of CLBP d/t Painful Annular Fissures
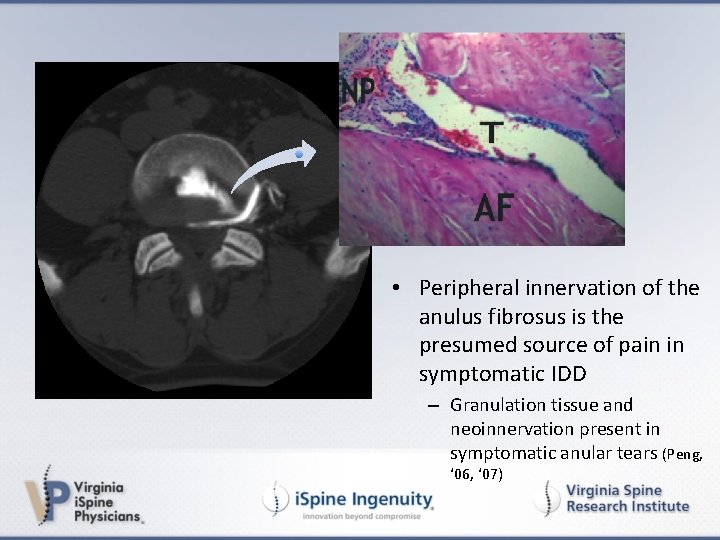
• Peripheral innervation of the anulus fibrosus is the presumed source of pain in symptomatic IDD – Granulation tissue and neoinnervation present in symptomatic anular tears (Peng, ‘ 06, ‘ 07)
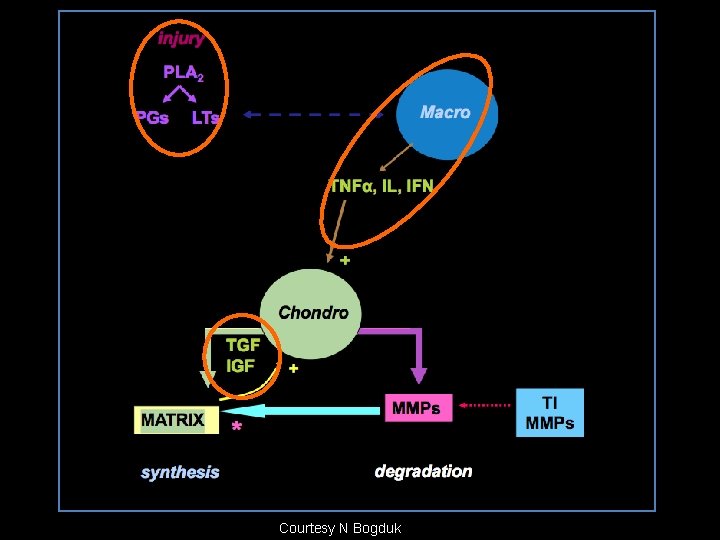
Courtesy N Bogduk

Can Experimental Technology Reconstitute the Disc? Do these Changes Translate into Clinical Improvement?
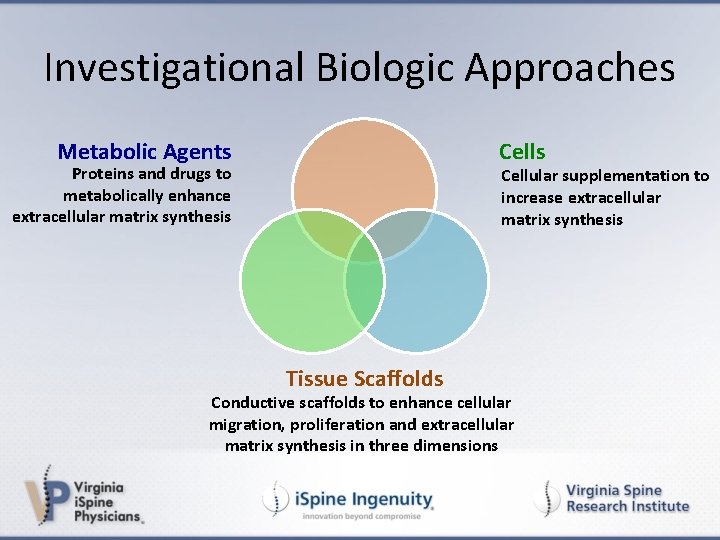
Investigational Biologic Approaches Metabolic Agents Cells Proteins and drugs to metabolically enhance extracellular matrix synthesis Cellular supplementation to increase extracellular matrix synthesis Tissue Scaffolds Conductive scaffolds to enhance cellular migration, proliferation and extracellular matrix synthesis in three dimensions

Investigational Biologic Approaches Intradiscal Metabolic Agents • Proteins and drugs to metabolically enhance extracellular matrix synthesis ADVANTAGES DISADVANTAGES • • • Anabolic benefit (inductive) Anti-catabolic Proliferative Extensive In vitro and animal preclinical evidence Viable cell content? Limited cellular nutrition? Vertebral osteolysis? Extradiscal leakage? Expense?
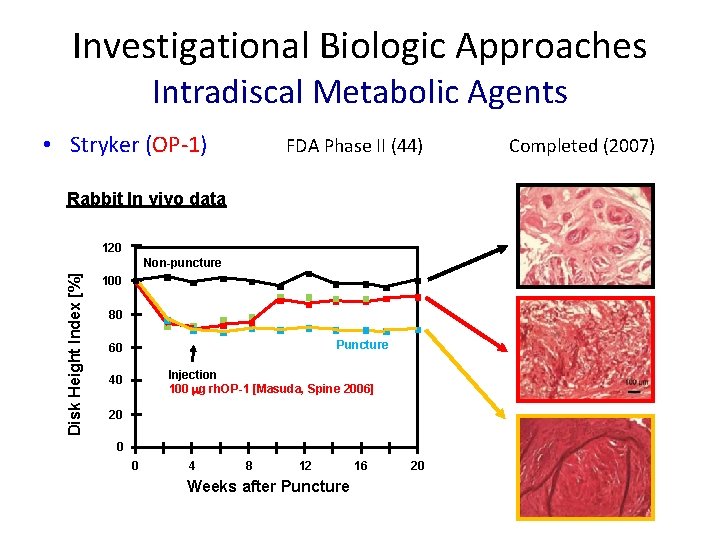
Investigational Biologic Approaches Intradiscal Metabolic Agents • Stryker (OP-1) FDA Phase II (44) Rabbit In vivo data 120 Disk Height Index [%] Non-puncture 100 80 Puncture 60 Injection 100 mg rh. OP-1 [Masuda, Spine 2006] 40 20 0 0 4 8 12 Weeks after Puncture 16 20 Completed (2007)

Investigational Biologic Approaches Intradiscal Metabolic Agents • Depuy (GDF-5) FDA Phase I/II (30) FDA Phase II (30) Rabbit In vivo data 120 Disk Height Index [%] Non-puncture 100 80 Puncture 60 Injection 100 mg rh. GDF-5 [Chujo, Spine 2006] 40 20 0 0 4 8 12 Weeks after Puncture 16 20 Enrolling (2012) Enrolling (2013)

Investigational Biologic Approaches Intradiscal Cells • Cellular supplementation to increase extracellular matrix synthesis – Juvenile chondrocytes [ISTO Technologies] – Meshenchymal Presursor cells [Mesoblast] – Progenitor cells [Discgenics] ADVANTAGES DISADVANTAGES • Anabolic benefit • Anti-catabolic (stem cells) • Extensive In vitro and animal preclinical evidence • • • Cell viability? Limited cellular nutrition? Differentiation signals? ECM similarity? Expense?
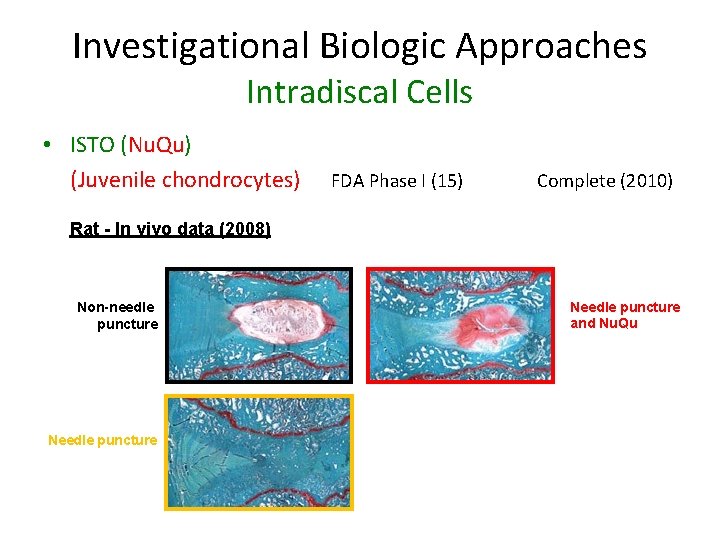
Investigational Biologic Approaches Intradiscal Cells • ISTO (Nu. Qu) (Juvenile chondrocytes) FDA Phase I (15) Complete (2010) Rat - In vivo data (2008) Non-needle puncture Needle puncture and Nu. Qu
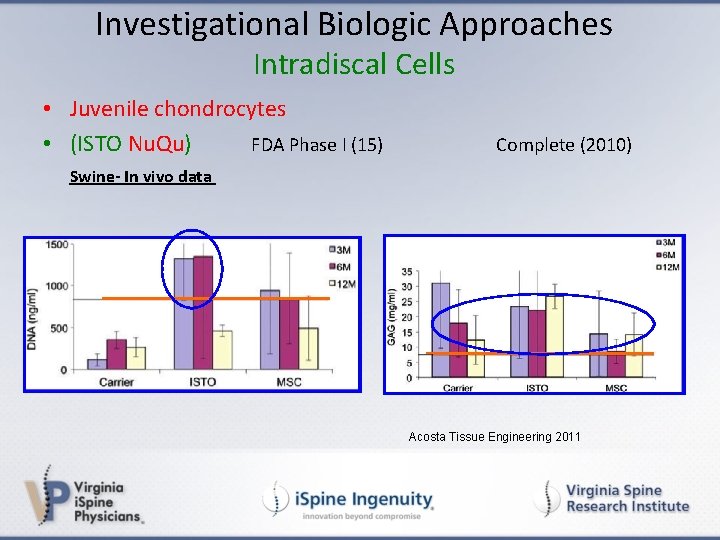
Investigational Biologic Approaches Intradiscal Cells • Juvenile chondrocytes • (ISTO Nu. Qu) FDA Phase I (15) Complete (2010) Swine- In vivo data Acosta Tissue Engineering 2011

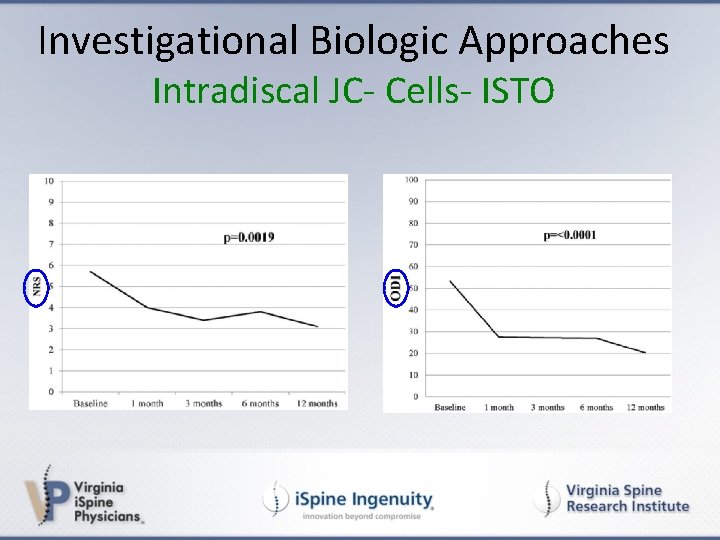
Investigational Biologic Approaches Intradiscal JC- Cells- ISTO • • 15 patient pilot, 2 site pilot study 1 year outcome Primary endpoint= safety Secondary endpoint= effectiveness (VAS, ODI, SF-36) • Single level + PLD- gr III or IV (exc. V) (Coric J Neurosurg: Spine, 2012)

Investigational Biologic Approaches Intradiscal JC- Cells- ISTO • Results: • 14 subjects: • BL- NRS/ODI was 5. 7 / 53. 3% • 12 mon - NRS / ODI was 3. 07 (p<0. 0019) / 20. 2% (p<0. 0001) (Coric J Neurosurg: Spine, 2012)
Investigational Biologic Approaches Intradiscal JC- Cells- ISTO

Investigational Biologic Approaches Intradiscal JC- Cells- ISTO • Results: • 87% (95%CI: 78 -96) 30% improvement in ODI • 3 subjs underwent TDR • No SAE’s (Coric J Neurosurg: Spine, 2012)
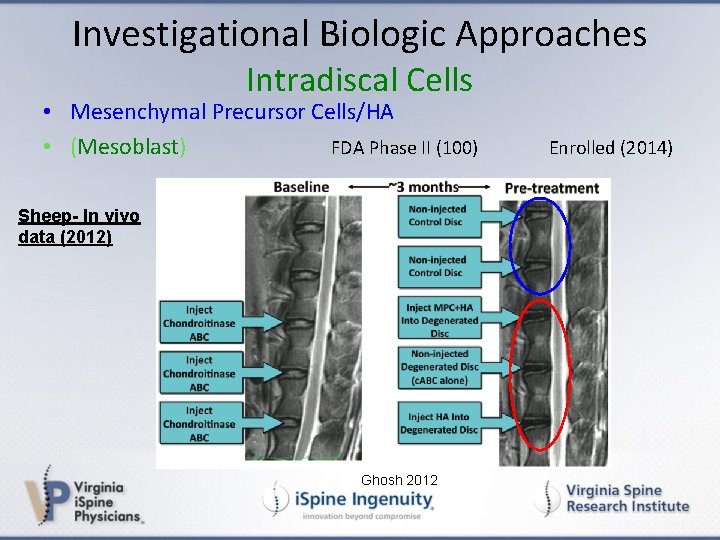
Investigational Biologic Approaches Intradiscal Cells • Mesenchymal Precursor Cells/HA • (Mesoblast) FDA Phase II (100) Sheep- In vivo data (2012) Ghosh 2012 Enrolled (2014)
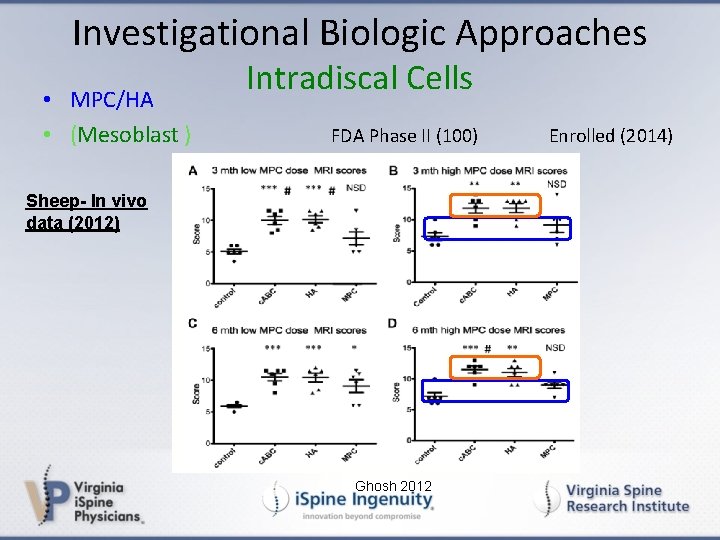
Investigational Biologic Approaches • MPC/HA • (Mesoblast ) Intradiscal Cells FDA Phase II (100) Sheep- In vivo data (2012) Ghosh 2012 Enrolled (2014)
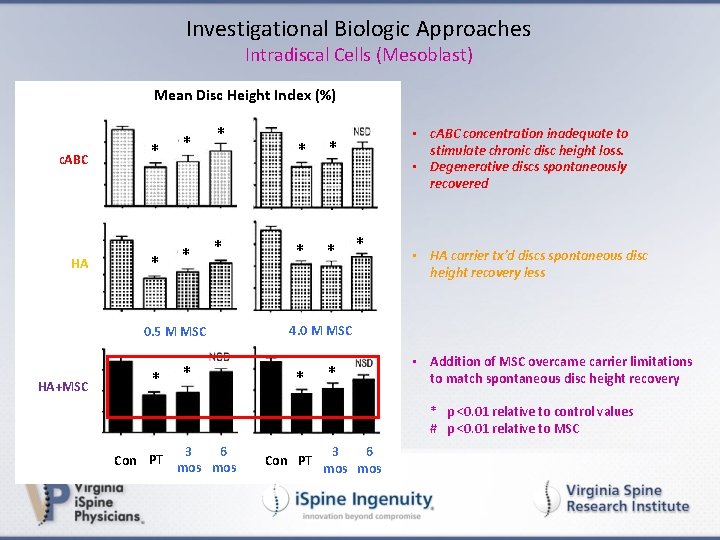
Investigational Biologic Approaches Intradiscal Cells (Mesoblast) Mean Disc Height Index (%) c. ABC HA * * * 0. 5 M MSC HA+MSC * * * • c. ABC concentration inadequate to stimulate chronic disc height loss. • Degenerative discs spontaneously recovered * • HA carrier tx’d discs spontaneous disc height recovery less 4. 0 M MSC * * • Addition of MSC overcame carrier limitations to match spontaneous disc height recovery * p <0. 01 relative to control values # p <0. 01 relative to MSC Con PT 3 6 mos Con PT 6 3 mos

Investigational Biologic Approaches Intradiscal Stem Cells- Mesoblast • • 100 patient RCT- single ID inj 3 year outcome: results @12 mon Primary endpoint= safety Secondary endpoint= effectiveness (VAS, ODI, MRI) • Single level mod Pfirrmann 3 -6; type I or II MC • Incomplete annular tear (Gr V exc) (NASS Ann Mtg, Nov 13, 2014; De. Palma SIS ASM, 2014)

Investigational Biologic Approaches Intradiscal Stem Cells- Mesoblast • Results @ 12 months: • 30 subj: 6 M cells; 40 subj: control • % who had > 50% reduction of VAS: • 69% (95%CI: 53, 86) tx arm • 33% (95%CI: 19, 48) ctrl arm • % with residual < 2/10 VAS LBP: • 52% (95%CI: 34, 70) • 18% (95%CI: 6. 1, 30) (NASS Ann Mtg, Nov 13, 2014, De. Palma SIS ASM, 2014)
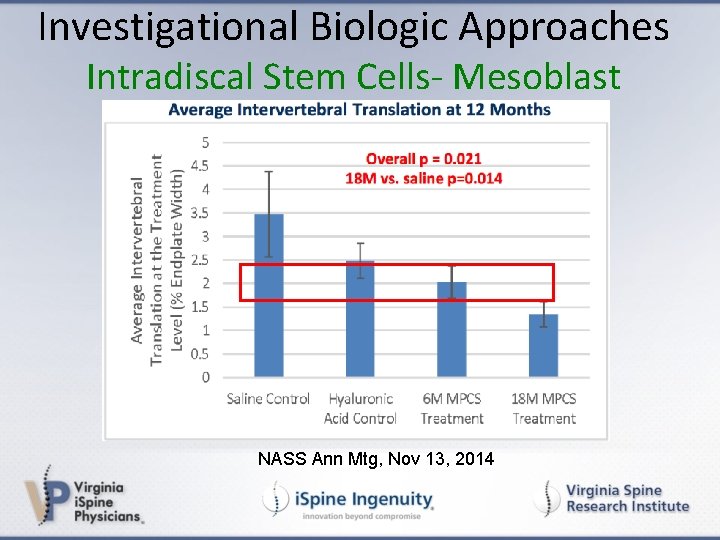
Investigational Biologic Approaches Intradiscal Stem Cells- Mesoblast NASS Ann Mtg, Nov 13, 2014

Investigational Biologic Approaches Tissue Scaffolds • Conductive scaffolds to enhance cellular migration, proliferation and extracellular matrix synthesis in three dimensions – Fibrin sealant [Spinal Restoration] ADVANTAGES DISADVANTAGES • • • Viable cell concentration? • Scaffold longevity? • Biocompatibility? Anabolic (inductive, conductive) Anti-catabolic Proliferative Extensive In vitro, animal and human preclinical evidence • Inexpensive

Fibrin Sealant: Potential benefits for pathogenic IDD Vascular dependence Natural soft tissue processes Bleeding Clot formation Fibrin sealant Anabolic Tissue Formation Catabollic Tissue Resorption Cellular dependence Inflammation Proliferation Tissue Repair Tissue Remodeling Hours Days Weeks Time Months
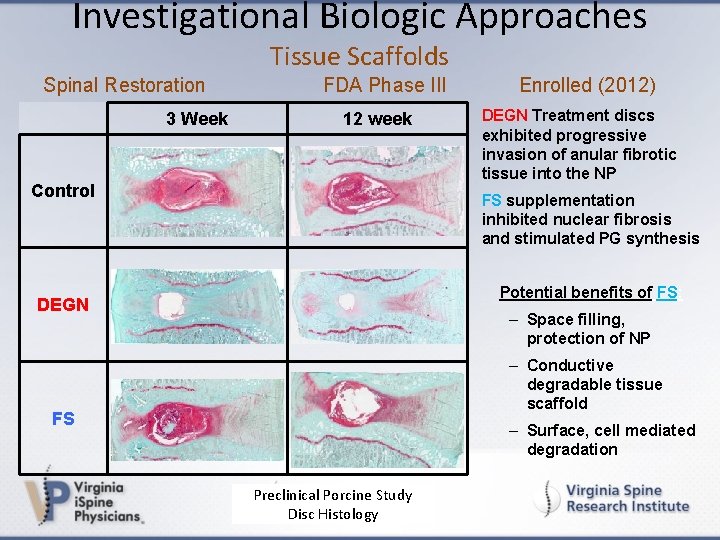
Investigational Biologic Approaches Tissue Scaffolds Spinal Restoration 3 Week FDA Phase III 12 week Control Enrolled (2012) DEGN Treatment discs exhibited progressive invasion of anular fibrotic tissue into the NP FS supplementation inhibited nuclear fibrosis and stimulated PG synthesis Potential benefits of FS: DEGN – Space filling, protection of NP – Conductive degradable tissue scaffold FS – Surface, cell mediated degradation Preclinical Porcine Study Disc Histology
![Investigational Biologic Approaches Tissue Scaffolds Denucleated discs (DEN) Time [wks] IL-1 b TNFa IL-6 Investigational Biologic Approaches Tissue Scaffolds Denucleated discs (DEN) Time [wks] IL-1 b TNFa IL-6](http://slidetodoc.com/presentation_image/a5c50bfb4db160791ab6ef9236b2e4bd/image-36.jpg)
Investigational Biologic Approaches Tissue Scaffolds Denucleated discs (DEN) Time [wks] IL-1 b TNFa IL-6 IL-8 IL-4 TGFb 2, 3 +129% +126% 160% +313% +33% +6% 6 +4% +114% -16% -32% +33% +70% 12 -20% +52% +33% +2995% +265% +49% IL-4 TGFb Fibrin supplemented treatment discs (FS) Time [wks] IL-1 b TNFa IL-6 IL-8 2, 3 -19%+ -34%+ +25%+ 130%+ 6 +100% +165% +167% -58% -88% +21% 12 -45% +56% +32% +260% 0% +120% +204%*+ +223%*+ Biochemical Compositional Analysis Relative cytokine synthesis [pg/ng DNA]
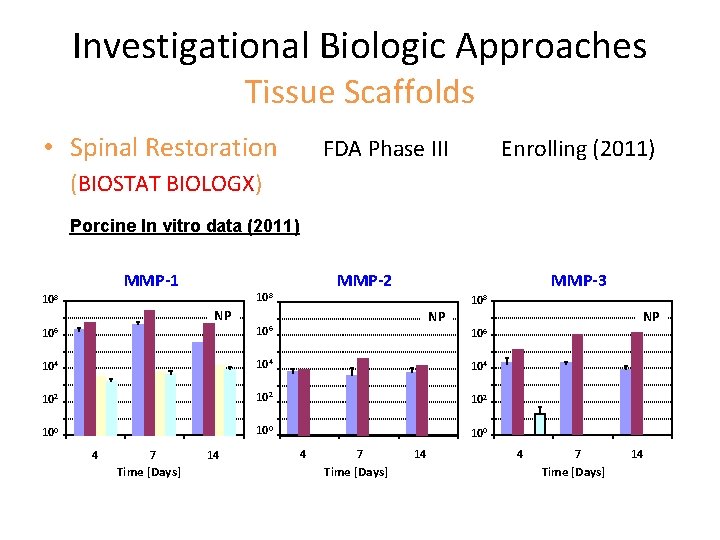
Investigational Biologic Approaches Tissue Scaffolds • Spinal Restoration FDA Phase III Enrolling (2011) (BIOSTAT BIOLOGX) Porcine In vitro data (2011) MMP-1 MMP-2 108 NP MMP-3 108 NP NP 106 104 104 102 102 100 100 4 7 Time [Days] 14 106 4 7 Time [Days] 14
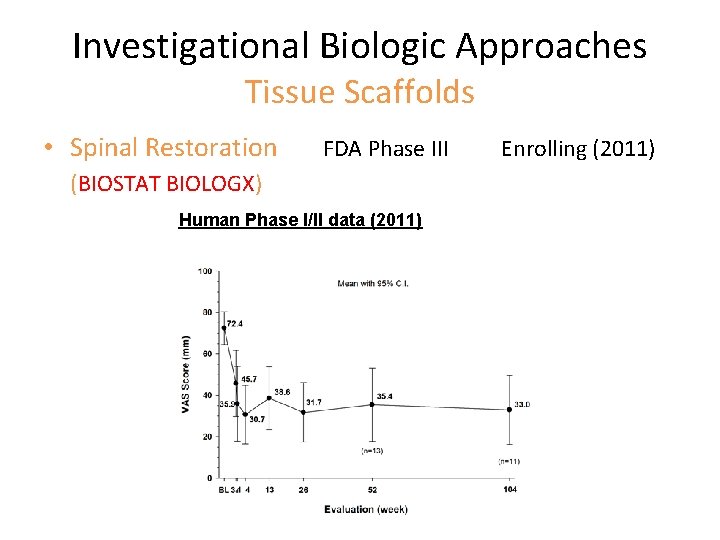
Investigational Biologic Approaches Tissue Scaffolds • Spinal Restoration FDA Phase III (BIOSTAT BIOLOGX) Human Phase I/II data (2011) Enrolling (2011)

Investigational Biologic Approaches Tissue Scaffolds • Spinal Restoration FDA Phase III Enrollment complete (2012) • (BIOSTAT BIOLOGX) RCT 220 subj. s - 26 wk primary endpoint: -33. 5% TX vs 39. 3% CTL achieved successful outcome -42. 1% TX vs 50. 0% CTL had clinically meaningful reduction in LBP -52. 4% TX vs 50. 5% CTL had clinically meaningful reduction in RMD Spinal Restoration Press Release 7/13

Conclusions • Painful discs cannot be explained solely by degeneration • Painful annular fissures flanked by innervated granulation tissue • Chronic LBP is related to non-healing of painful fissures

Conclusions • Growth factors, cell supplementation, tissue scaffolding • Combination of technologies • Ideal treatment hasn’t yet been identified – But must address regeneration as well as reconstitution of altered tissue mechanical properties

Thank you
- Slides: 42