Em ONC Toolkit Appendix 3 Ensuring Data Quality
















- Slides: 17

Em. ONC Toolkit Appendix 3 Ensuring Data Quality for Monitoring and Evaluation

Objectives • To define high-quality data • To discuss the characteristics of goodquality data • To describe the ethical considerations in collecting data for M&E

What Are Data? • Facts or information, especially when examined and used to find out things or to make decisions • The plural of datum Oxford Advanced Learner's Dictionary

What Are High-Quality Data? Data are of high quality if they are fit for their intended use in operations, decisionmaking, and planning. Source: Juran, J. M. (1964). Managerial breakthrough. New York, N. Y. , USA: Mc. Graw -Hill.

What Are High-Quality Data? Data are of high quality if they are fit for their intended use in operations, decisionmaking, and planning. Source: Juran, J. M. (1964). Managerial breakthrough. New York, N. Y. , USA: Mc. Graw -Hill.

Importance of High-Quality Data • High-quality information is an important resource for the health sector in planning, managing, delivering, and monitoring high quality, safe, and reliable health care • The Ministry of Health recommends that data quality review fora (meetings, workshops, conferences) be held periodically at each appropriate level (national, county / subcounty, facility, and community).

Dimensions of High-Quality Data 1. Accuracy 2. Precision 3. Completeness 4. Reliability 5. Timeliness 6. Relevance 7. Ethical integrity

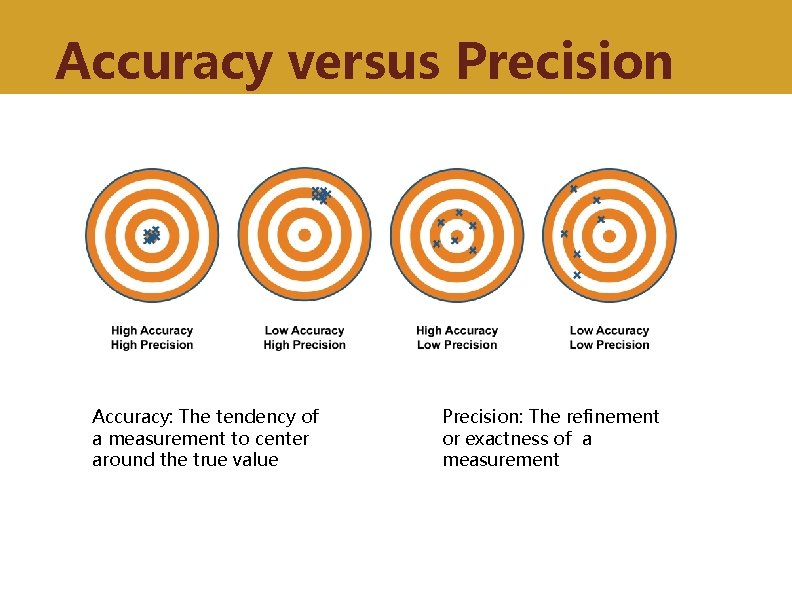
Accuracy • Data should provide a clear representation of the activity/interaction • Data should be in sufficient detail • Data should be captured once only, as close to the point of activity as possible • Data sources: Direct visual inspection, interview, review of medical records • Accuracy can be ascertained by supervision and verification

Precision • The number of observations (e. g. , health facilities or medical records) sampled should be sufficient to estimate measures to an acceptable level of refinement • The fewer the number of observations, the lower the precision • Need to balance precision with the logistical challenge of increasing the number of observations sampled

Accuracy versus Precision Accuracy: The tendency of a measurement to center around the true value Precision: The refinement or exactness of a measurement

Completeness • Missing data are impossible to interpret • Leaving a field blank does not equate to absence of the indicator being accessed or “not applicable” Y N Does not equate to Y N

Reliability • Data collection processes must be clearly defined and stable to ensure consistency over time • Standard operating procedures enhance consistency in process of data collection across locations and over time

Timeliness • Data should be collected and recorded as quickly as possible after the event or activity • Data should remain available for the intended use within a reasonable or agreed period • Delayed transmission of data reduces data’s utility for decisionmaking

Relevance • Data should be relevant for the purposes for which they are used • Data requirements should be clearly specified and regularly reviewed to reflect any change in needs • The amount of data collected should be proportional to the value gained from them

Ethical integrity • Appropriate authorization is essential • Data from medical records must be handled with confidentiality and extracted data must be made anonymous • Data storage must be secure and only accessible to authorized people • Highest standards of integrity are required among those collecting data. Falsification of data may result in serious consequences. [Group to discuss possible consequences of data falsification. ]

References • Government of Kenya. (2014). Kenya health sector data quality assurance protocol. Nairobi, Kenya: Ministry of Health, Afya. Info Project. • Government of Kenya. (2015). Addendum to Kenya health sector data quality assurance protocol (2014) data quality review: Guidelines for conducting data quality reviews at all levels. Nairobi, Kenya: Ministry of Health, Afya. Info Project.

MEASURE Evaluation PIMA is funded by the U. S. Agency for International Development (USAID) through associate award AID-623 -LA-12 -00001 and is implemented by the Carolina Population Center at the University of North Carolina at Chapel Hill, in partnership with ICF International; Management Sciences for Health; Palladium; and Tulane University. The views expressed in this presentation do not necessarily reflect the views of USAID or the United States government. www. measureevaluation. org/pima