Elements The Periodic Table The groups go up

Elements & The Periodic Table
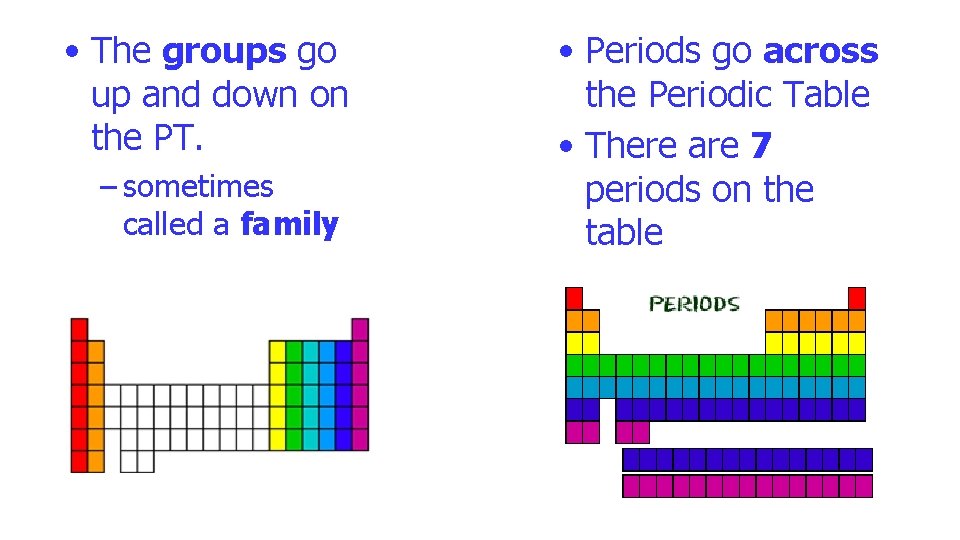
• The groups go up and down on the PT. – sometimes called a family • Periods go across the Periodic Table • There are 7 periods on the table

BATTLESHIP
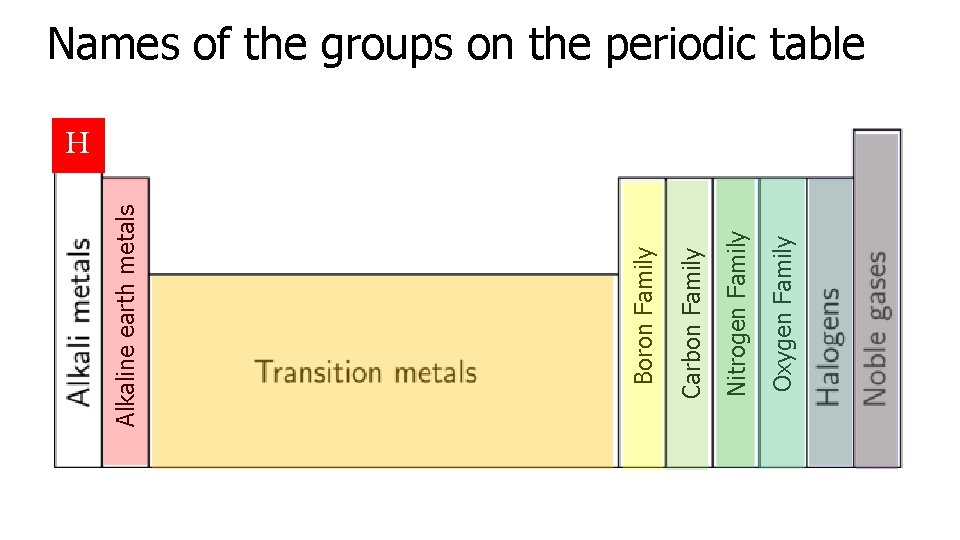
Oxygen Family Nitrogen Family Carbon Family Boron Family Alkaline earth metals Names of the groups on the periodic table H

Metal characteristics • Shiny / lustrous • Good conductors of heat (thermal conductors) and electricity because the electrons can move throughout the metal. • Malleable – able to be hammered into shapes • Ductile – able to be made into wires

Non-metals 1. Brittle 2. Poor conductors of heat and electricity 3. Dull i. e. lack brightness and sheen

Metalloids aka semi-metals have both metal and non -metal properties
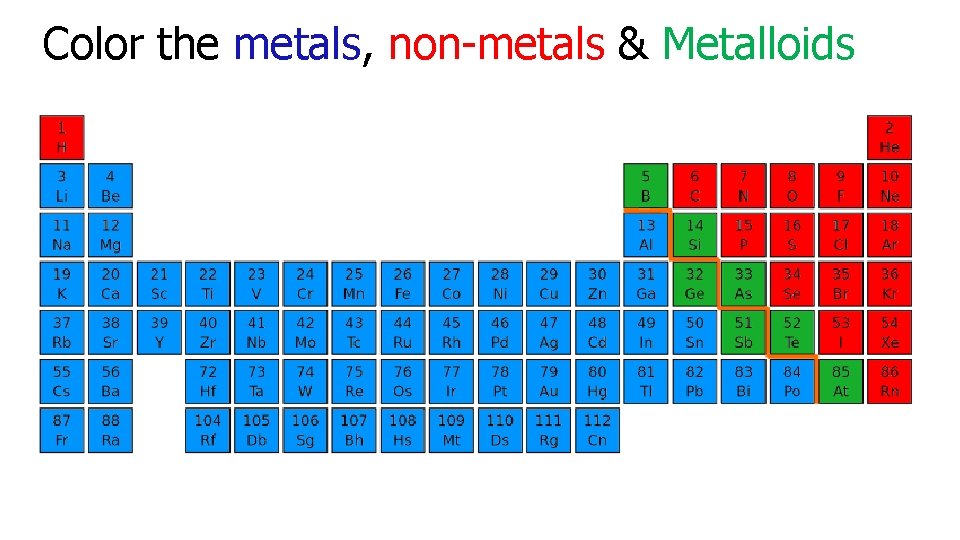
Color the metals, non-metals & Metalloids
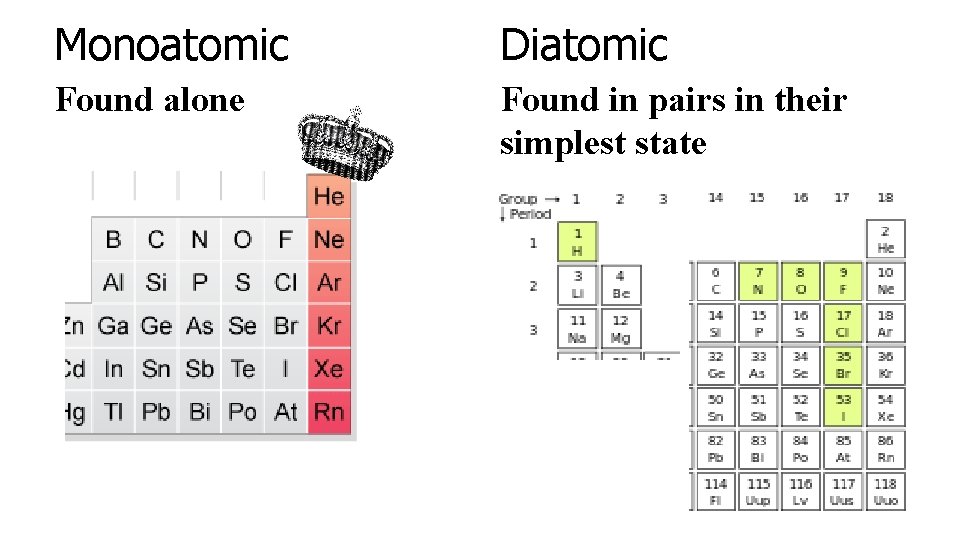
Monoatomic Diatomic Found alone Found in pairs in their simplest state

Solids, Liquids and Gases on The Periodic Table
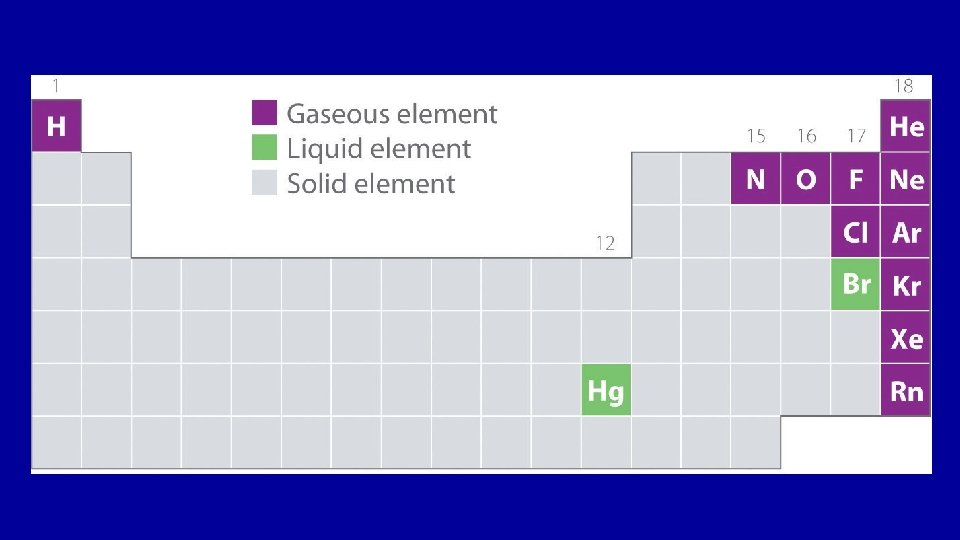

Solid(s) • strong intermolecular forces • keep definite shape & volume • particles packed tightly together – Incompressible (can’t be squeezed into a smaller volume) • particles vibrate in fixed positions

• Liquid(l) –intermolecular forces are not strong enough to keep a definite shape –particles move past each other in fluid motion –take the shape of their container –particles close together • incompressible

• Gas(g) –weakest intermolecular forces –particles take the shape of and completely fill any size container –Lots of space between the particles • Compressible

The End
- Slides: 15