Elements Section 3 Objectives Use the periodic table

Elements Section 3
Objectives • Use the periodic table to name elements, given their symbols • Use a periodic table to write the symbols of elements, given their names • Describe the arrangement of the periodic table • List the characteristics that distinguish metals, nonmetals, and metalloids
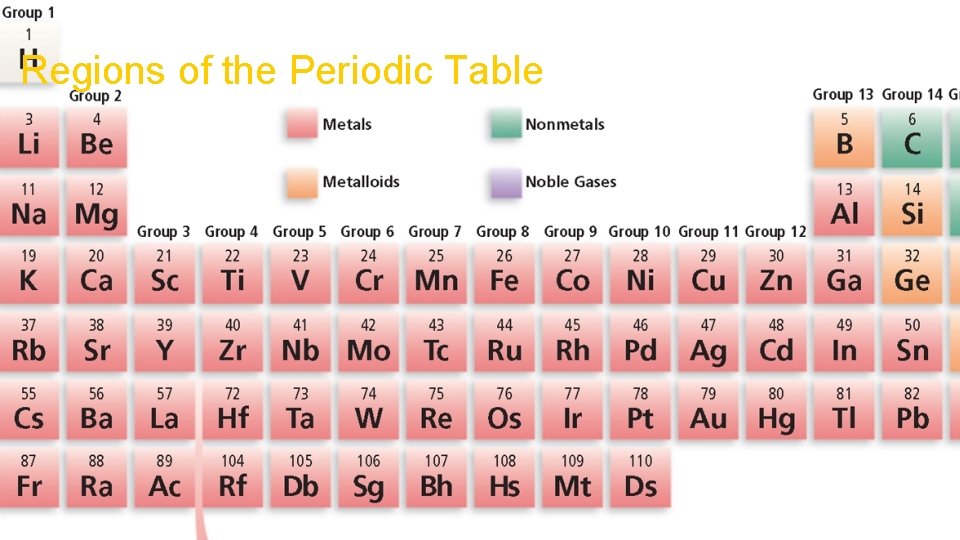
Chapter 1 Section 3 Elements Regions of the Periodic Table

Periodic Law • Modern Periodic Table developed my Mendeleev • He noticed patterns in elemental properties • Arranged them in columns based on similarities • Left holes where he thought a new (undiscovered element would fit) • Periods – similar sizes (# of layers) big on left, small on right • Next row, additional layers (so all are bigger than element above)

Chapter 1 Section 3 Elements Introduction to the Periodic Table • The vertical columns of the periodic table are called groups, or families. • Each group contains elements with similar chemical properties. • The horizontal rows of elements in the periodic table are called periods. • Physical and chemical properties change somewhat regularly across a period.

Types of Elements Metals • A metal is an element that is a good electrical conductor and a good heat conductor. • Properties of metals • most are solids at room temperature • malleable - they can be hammered or rolled into thin sheets • ductile - they can be drawn into a fine wire • conduct electricity and heat well

Types of Elements • Gold, copper, and aluminum are metals

Types of Elements Nonmetals • A nonmetal is an element that is a poor conductor of heat and electricity. • Properties of nonmetals • many are gases • solids are brittle • poor conductors of heat and electricity

Types of Elements • Various nonmetal elements (a) carbon, (b) sulfur, (c) phosphorus, and (d) iodine

Types of Elements Metalloids • A metalloid is an element that has some characteristics of metals and some characteristics of nonmetals. • Properties of metalloids • all metalloids are solids at room temperature • semiconductors of electricity

Types of Elements Noble Gases • elements in Group 18 of the periodic table • generally unreactive • gases at room temperature
Objectives • Use the periodic table to name elements, given their symbols • Use a periodic table to write the symbols of elements, given their names • Describe the arrangement of the periodic table • List the characteristics that distinguish metals, nonmetals, and metalloids

SHORT ANSWER Answer the following questions in the space provided. 1. A horizontal row of elements in the periodic table is called a(n) ________. 2. The symbol for the element in Period 2, Group 13, is ________. 3. Elements that are good conductors of heat and electricity are ________. 4. Elements that are poor conductors of heat and electricity are ________. 5. A vertical column of elements in the periodic table is called a(n) ________. 6. The ability of a substance to be hammered or rolled into thin sheets is called ________. 7. Is an element that is soft and easy to cut cleanly with a knife likely to be a metal or a nonmetal? ________. 8. The elements in Group 18, which are generally unreactive, are called ________. 9. At room temperature, most metals are ________. 10. Name three characteristics of most nonmetals. 11. Name three characteristics of metals. 12. Name three characteristics of most metalloids. 13. Name two characteristics of noble gases.
- Slides: 13