ELEMENTS OF MECHANICAL ENGINEERING UNIT1 THERMODYNAMICS Unit I
































- Slides: 36

ELEMENTS OF MECHANICAL ENGINEERING UNIT-1 THERMODYNAMICS

Unit I - Introduction Energy Sources and Conversion The various sources of energy are 1. 2. 3. 4. 5. 6. 7. 8. Fuel Fossil Fuels Nuclear energy Geothermal Energy Solar energy Tidal power Flowing Streams of water(Rivers) Wind Energy 2

FUELS: A fuel is a substance which gives heat energy on combustion. The main combustible elements of a fuel are carbon and hydrogen. The presence of sulphur is undesirable though it is also a combustible. Fuels Primary fuel Prepared fuel Primary fuels occur directly in the nature. Prepared fuels are also called as derived fuels and are prepared artificially. Classification of fuels: 1) Solids e. g: wood, coal, charcoal, anthracite, lignite, peat. 2) liquids e. g: crude oil, petroleum, kerosene, alcohol, etc 3) gaseous e. g: natural gas, producer gas, coal gas, etc. 3

NUCLEAR ENERGY The energy released during nuclear fission or fusion, especially when used to generate electricity. � One of the out standing facts about nuclear power is the large amount of energy that can be released from a small mass of nuclear active material. � Complete fission of one kg of uranium contains the energy equivalent of 4500 tonnes of coal or 2000 tonnes of oil. � The nuclear power is not only available in abundance but it is cheaper than the power generated by conventional sources. � 4

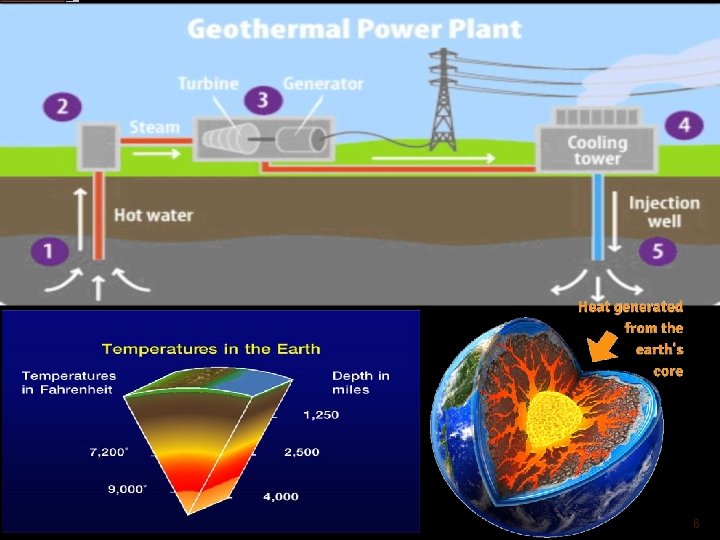
GEOTHERMAL ENERGY Earth is a molten core. This statement is supported by volcanos at many places on the earth. The steam that comes out of the natural steam well is used for power generation. This energy is termed as geothermal energy. There are two ways in power production from geothermal energy 1) direct method 2) indirect method. In Direct system the hot geothermal water/steam is used to operate the turbine directly. In this method a separator is used to remove the moisture and foreign particles. The Indirect method is used when temperature of geothermal source is not sufficient to drive the turbine. In this method the hot steam/water is used to heat the secondary fluid with help of heat exchanger. The secondary fluid like Freon, iso-butane circulated in the closed cycle. The main advantage of this method is low temperature water/steam can be used effectively. 5

6

SOLAR ENERGY The major advantages are 1) Operating cost is eliminated 2) Conversion of energy is easy 3) Not harmful to environment The major disadvantages are 1) It cannot be used on cloudy days or at nights 2) It is uneconomical 3) It requires large area even for production of small power. Applications of solar energy 1) solar water pumps 2) solar water heater 3) solar power plants. 7

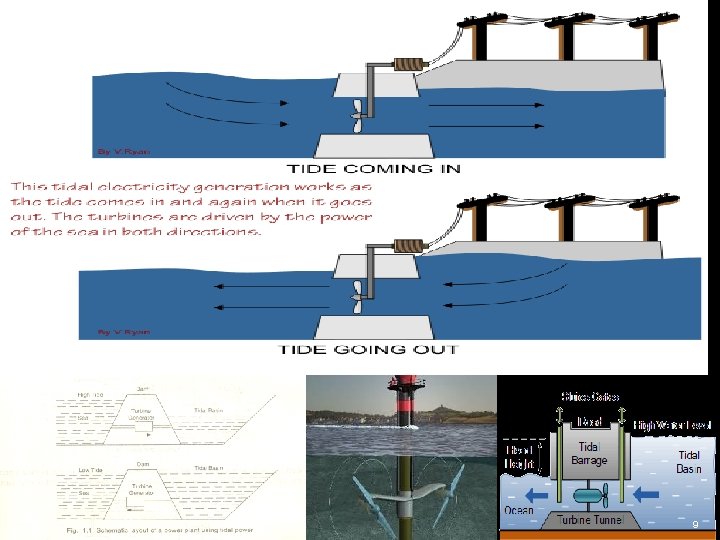
TIDAL ENERGY Tides contain large amount of energy. Rise and fall of tides create water head which helps in driving the turbine. Water is stored during tide rise and water is discharged during fall. The available head is low. So to increase the power generation the catchment area should be increased. The head developed is just about few meters. During high tide the water level on sea tide (high tide) side is above the tidal basin and exactly opposite in low tide case. During low tide the height of the tide is lower than tidal basin. During the period water tends to flow out driving the turbine unit. The turbine unit does not operate if the tide seal level is equal to basin level. ADVANTAGES: 1) power generation is rain independent. 2) no uncertainty in power development. 3) power generation is free from pollution. 4) undesirable wastes (like ash, gases) are not produced. 8

9

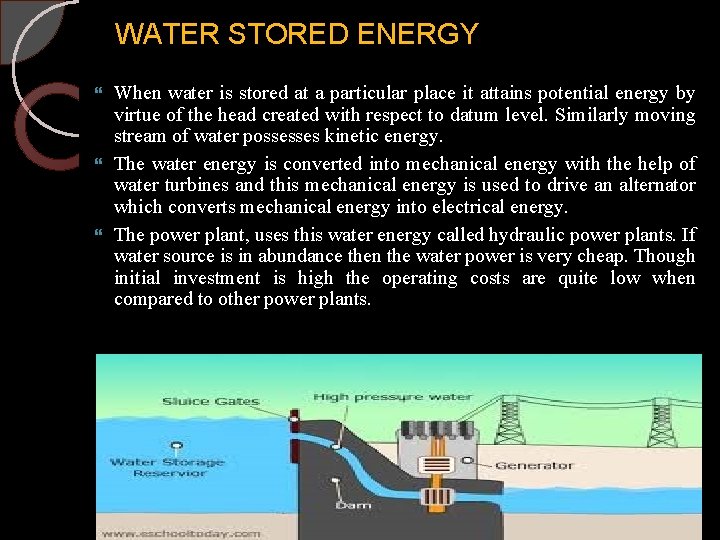
WATER STORED ENERGY When water is stored at a particular place it attains potential energy by virtue of the head created with respect to datum level. Similarly moving stream of water possesses kinetic energy. The water energy is converted into mechanical energy with the help of water turbines and this mechanical energy is used to drive an alternator which converts mechanical energy into electrical energy. The power plant, uses this water energy called hydraulic power plants. If water source is in abundance then the water power is very cheap. Though initial investment is high the operating costs are quite low when compared to other power plants. 10

WIND ENERGY Wind energy is the cheapest source of power. Wind energy is used to rotate wind turbine/wind mill to develop mechanical energy The limitations of this wind energy are 1) variable output 2) uncertainty in wind 3) power generated is very low. The advantages are 1) no operator is needed. 2) very low maintenance and almost nil repairs. The main application of wind energy is in pumping water from deep wells. The equipment altogether used to produce power from wind energy is called Wind Mill 11

Basic Concepts of Thermodynamics �The term Thermodynamics is derived from the Greek words ‘Thermic’ which means Heat and ‘Dynamics’ which means Force. �Thermodynamics is the science that deals with the relationship of heat and mechanical energy and conversion of one into the other. �TD began in 19 th Century to exploit the motive power of heat – the capacity of hot bodies to provide work.

�The study of TD is based on two general laws of nature, the First law and the second law of TD. �First Law : Heat and work are two mutually convertible forms of energy. �Heat never flows on its own from an object at low temperature to an object at higher temperature, this statement is basis for the second law. �Second Law : Heat energy of the source cannot be converted continuously to work but part of it has to be rejected to sink at lower temperature.

MICROSCOPIC & MACROSCOPIC ANALYSIS Behavior of matter can be studied in two viewpoints: Microscopic : Behavior of individual atoms or molecules. Behavior of gas is described by summing up behavior of each molecule (Statistical Thermodynamics). Macroscopic : Behavior of more number of molecules is taken into account. Eg : Force on a given area can be measured by Pressure gauge.

Classification of Heat Engines

�Phase: A quantity of matter, homogenous in chemical composition and physical structure, is called a Phase. q. Homogeneous: A system which consists of a single Phase. Eg: Air and watervapour. q. Heterogeneous: A system which consists of two or more phases. Eg: Ice and water ; water and oil.

�Pure Substance: A pure substance is the one that has a homogeneous and invariable chemical composition even though there is change in phase. Ø System: A system is a finite quantity of matter or prescribed region of space. Boundary: It is a real or imaginary envelope enclosing a system.

Surroundings: Everything external to the system. Universe: System and its surroundings together comprise a system. There are three types of System: • Closed System • Open System • Isolated System

Property and State �Property is any observable or measurable characteristic of a substance. Ex: Pressure, Volume and temperature i) Intensive and Extensive Properties ii) Specific Properties State is the condition of the system as described or measured by the properties.

Substances �Pure Substance: A substance whose chemical composition is both homogeneous and constant is known as a pure substance. Eg: Mixture of steam and water. • Homogeneous Substance: Substances existing in single phase are called as Homogeneous Substances. • Heterogeneous Substance: Substances existing in more than one phase are Heterogeneous substances.

Thermodynamic Equilibrium �A system is said to be in thermodynamic equilibrium when there is no change in the macroscopic property, when the system is isolated from the surroundings. �A system will be in thermodynamic equilibrium when the conditions for following equilibrium are satisfied: • Mechanical Equilibrium • Chemical Equilibrium • Thermal Equilibrium

Process � When a system changes from one equilibrium state to another equilibrium state it is said to have undergone a process. � Continuous series of equilibrium states through which the system passes for reaching from initial state to final state is known as path. When path is completely specified it is known as process. �Different Types of Process § Reversible & Irreversible § Thermodynamic cycle § Point and Path function

Heat & Work Ø Similarities: • Boundary Phenomenon • Both are Path Functions • Both are interchangable • Both are inexact differentials and hence are not thermodynamic properties. Ø Differences: • There cannot be reversible work transfer but there is no restriction for transfer of heat. • For transfer of heat, temperature difference is needed. • Sign Convection.

Ø Displacement work Ø Temperature: It is an intensive thermodynamic property related to the “hotness” or “coldness” of a body measured on a definite scale. Ø Zeroth Law of Thermodynamics: When a body ‘A’ is in thermal equilibrium with body ‘B’ and also separately with body ‘C’ then B and C will be in thermal equilibrium with each other.

Ø “If two systems are in thermal equilibrium with a third system, they must be in thermal equilibrium with each other. ” Ø Conversion Scales Ø Properties of Ideal Gases: • A gas is a substance which cannot be liquified by application of pressure at constant temperature. • A vapour is a gaseous substance which can be liquified by applying pressure at constant temperature. • The laws of perfect gas does not apply to vapour. Substance like air, dry or superheated steam are treated as gas.

Ø Boyle’s Law: Boyle’s Law states that when any gas is heated at constant temperature, the pressure and volume of the gas are inversely propotional. Ø Charle’s Law: Charle’s law states that when any gas is heated at constant pressure, its change in volume varies directly with the absolute temperature change.

First Law of Thermodynamics Ø Law of conservation of energy. Ø First law of TD: “During any cycle that a closed system undergoes, the net work transfer is equal to the net heat transfer. ” Ø Specific Heat Ø Joule’s Law: Joules law states that “ The internal energy of a perfect gas is a function of the absolute temperature. ”

�Enthalpy: Sum of internal energy and pressure volume product (pv) is called Enthalpy (h). h=u+pv • Ratio of specific heats

Application of First Law of TD to Closed System or Non-Flow System i) Reversible constant volume or Isochoric process. ii) Reversible constant Pressure or Isobaric Process. iii) Reversible constant Temperature or Isothermal Process. iv) Reversible Adiabatic Process or Isentropic Process.

JOULE’S EXPERIMENT

Perpetual Motion Machine-1 Ø There can be no machine which would continuously supply mechanical work without some form of energy disappearing simultaneously. Such a fictitious machine is called PMM-1. Hence it is impossible. Ø Application of first law to steady flow process: Assumptions are made in the system analysis: The mass flow through the system remains constant. • Fluid is uniform in composition. • The only interaction between the system and surroundings are work and heat. •

The state of fluid at any point remains constant with time. • In the analysis only potential, kinetic and flow energies are considered. • Ø Limitations of the First Law of TD: It has been observed that energy can flow from system in the form of heat or work. • The law doesn't indicate whether conversion of energy from one form to another are performed perfectly or not. • It doesn’t speak about the conditions under which the transformations of energy are possible. •

The first law doesn’t answer whether complete conversion of heat is possible. • The complete conversion of work into heat is possible in cases like compression of gas and work spent in overcoming friction. • Complete conversion of heat into work is not possible. • PERPETUAL MOTION MACHINE-2 (PMM-2): “It is a fictitious machine which would absorb heat from single thermal reservoir and would convert this heat completely into work. ”

Second Law Of Thermodynamics Ø CLAUSIUS STATEMENT: The Clausius statement of the second law states that, “it is impossible for a self starting machine working in a cyclic process unaided by any external agency, to convey heat from a body at lower-temperature body to a body at highertemperature”. Ø KELVIN-PLANCK STATEMENT: The Kelvin-Planck statement of the second law of thermodynamics states that “It is impossible to construct an engine, which while operating in a cycle produce no other effect except to extract heat from a single reservoir and produce work”.

Refrigerator and Heat Pump �A Refrigerator is a device which, operating in a cycle, maintains a body at a temperature lower than the temperature of the surroundings. �A Heat Pump is a device, operating in a cycle, maintains a body , at a temperature higher that the temperature of the surroundings. ENERGY RESERVOIRS: A thermal energy reservoir (TER) is defined as a large body of infinite heat capacity, which is capable of absorbing or rejecting an unlimited quantity of heat without suffering appreciable change. •
