Elements Isotopes and More Atomic Number Atomic number

Elements, Isotopes and More

Atomic Number Atomic number is the number of protons in the atom’s nucleus. ¡ The elements are in order of their atomic number on the Periodic Table. ¡ l Example: Hydrogen has 1 proton; its atomic number is 1. ¡ Oxygen has 8 protons; its atomic number is 8. ¡
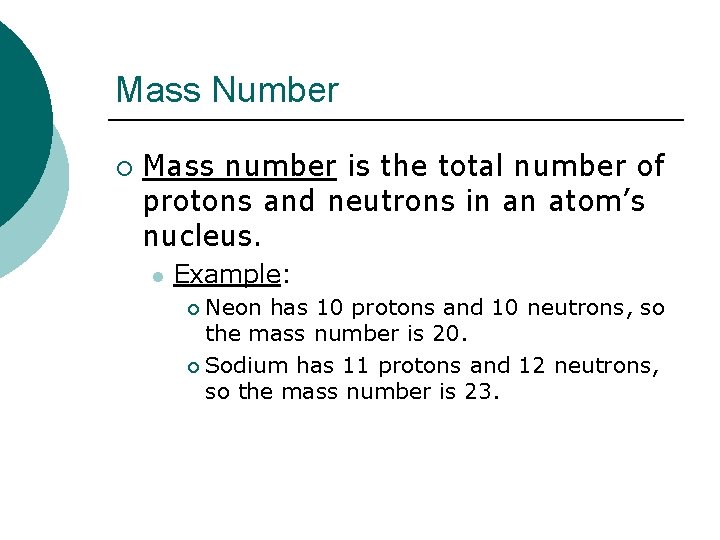
Mass Number ¡ Mass number is the total number of protons and neutrons in an atom’s nucleus. l Example: Neon has 10 protons and 10 neutrons, so the mass number is 20. ¡ Sodium has 11 protons and 12 neutrons, so the mass number is 23. ¡

Isotopes have differing numbers of neutrons, but the same number of protons. ¡ We can identify isotopes because the mass of the atom changes. ¡ l Neon has 3 isotopes: Ne-20, Ne-21, and Ne-22. All have 10 protons, but Ne-20 has 10 neutrons, Ne-21 has 11 neutrons and Ne-22 has 12 neutrons.

Ions have differing numbers of electrons, which gives the atom an electrical charge. ¡ Fewer electrons gives a positive charge, more electrons gives a negative charge. ¡ l Example: Potassium usually loses one electron, and it’s symbol becomes K+1. ¡ Chlorine usually gains one electron, and it’s symbol becomes Cl-1. ¡

Average Atomic Mass ¡ Atomic mass on the Periodic Table is actually an average of the masses of all the naturally-occurring isotopes and how abundant they are in nature. l Example: Element X has two natural isotopes. The isotope with mass 10. 012 amu has an abundance of 19. 91%. The isotope with mass of 11. 009 amu has a relative abundance of 80. 09%. What is the average atomic mass? 10 X: 10. 012 amu X 0. 1991 = 1. 993 amu 11 X: 11. 009 amu X 0. 8009 = 8. 817 amu 10. 810 amu

Complete Symbols ¡ Contain the symbol of the element, the mass number and the atomic number. Mass Superscript → number Subscript → Atomic number X

Symbols n Find each of these: a) number of protons b) number of neutrons c) number of electrons d) Atomic number e) Mass Number 80 35 Br

Symbols n If an element has an atomic number of 34 and a mass number of 78, what is the: a) number of protons b) number of neutrons c) number of electrons d) complete symbol

Atomic Masses Atomic mass is the average of all the naturally occurring isotopes of that element. Isotope Symbol Carbon-12 12 C Carbon-13 13 C Carbon-14 14 C Composition of the nucleus 6 protons 6 neutrons 6 protons 7 neutrons 6 protons 8 neutrons Carbon = 12. 011 % in nature 98. 89% 1. 11% <0. 01%

To calculate the average: ¡Multiply the atomic mass of each isotope by it’s abundance (expressed as a decimal), then add the results. ¡ If not told otherwise, the mass of the isotope is expressed in atomic mass units (amu)
- Slides: 11