Elements Compounds and Mixtures How does the composition







- Slides: 8

Elements, Compounds and Mixtures How does the composition of matter help us explain its diversity and predict its properties?

Learning Targets • I can classify matter as a mixture or a pure substance. • I know the difference between and element and a compound. • I can identify physical properties and give examples of physical changes • I can identify chemical properties and give examples of chemical changes.


Physical properties and physical change • Properties that you can measure or see through direct observation are called physical properties. • Ex: phase- Water is a colorless liquid at room temperature • Other examples • • Malleability-can be hammered into thin sheets Brittle- will break if material is hammered or bent Density- mass per unit of volume Mass- amount of matter • Physical changes- a change in physical properties, such as shape, phase; examples: grinding, melting, boiling, dissolving, heating or cooling.

Chemical properties and chemical change • Properties that can only be observed when one substances changes into a different substance are called chemical properties. • Ex: A chemical property of iron is that it will rust when it reacts with oxygen in the air. • Any change that transforms one substance into a different substance is called a chemical change. • Evidence of a chemical change: • • Bubbling- gas is forming Turns cloudy- a new solid is forming Temperature change- chemical bonds are forming or breaking Color change- a new substance is forming.

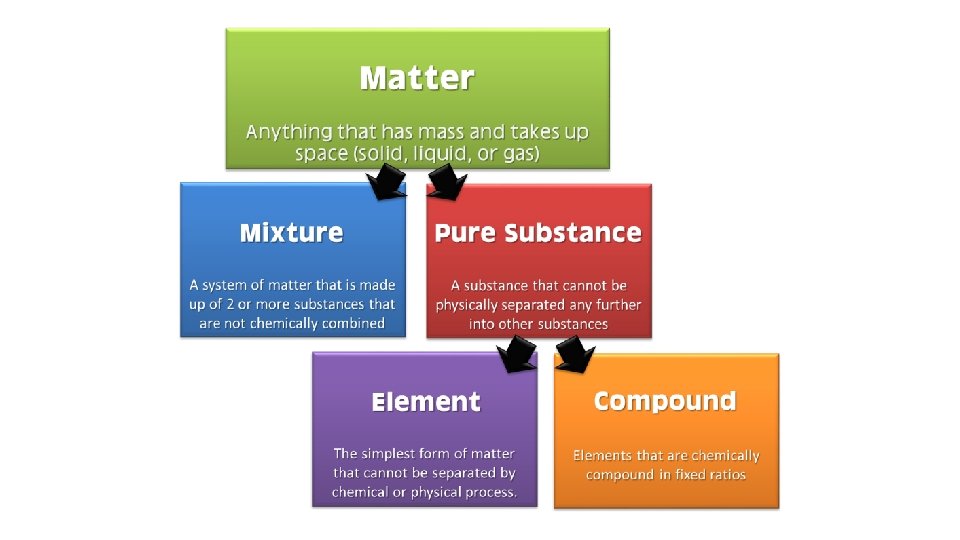
Elements and Compounds • Elements are the most fundamental “pure substances” from which all other matter is made. (Ex: oxygen, hydrogen, carbon) • The smallest unit of any element is a unique atom • Elements can not be separated into simpler substances by chemical means. • The periodic table organizes the elements according to how they combine with other elements. • There are 88 naturally occurring elements. • Compounds are substances that contain more than one element in which atoms of different elements are chemically bonded together. (Ex: water, carbon dioxide, sodium chloride) • The smallest unit of a compound that retains the identity of that compound is called a molecule.

Mixtures • A mixture is a physical blend of two or more substances that are not chemically combined. • Homogeneous mixture- a mixture that contains more than one type of matter and is the same throughout. • Ex: lemonade, air, alloys (mixture of metals-like brass) • A solution is a mixture that is homogenous on the molecular level (no clumps bigger than a molecule) • A solution always has a solvent and a solute. • Solvent- substance that makes up the biggest percentage of the mixture and is usually a liquid • Solute- the other substance in the solution (sometimes a dissolved solid, like sugar or salt) • Heterogeneous mixture- a mixture that contains more than one type of matter and is not the same throughout. • Ex: chicken soup, soil, concrete

Quiz Time!