Elements Compounds and Chemical Reactions 8 th Grade















































- Slides: 50

Elements, Compounds, and Chemical Reactions 8 th Grade Science

DO NOW Wednesday October Copy the following: th 25 Time NEW POLICY r A timer will be set on the DO NOW. When time is up, work will be checked. If DO NOW is not complete, you will get a mark on clip board. If you don’t know how to do the DO NOW, you should at least write the question. 3 marks on clip board, choice card signed for off-task behavior. 5 marks on clip board, parents will be called. 7 marks on clipboard, write-up sent to office. Marks on clip board will be given for ANY off-task behavior during class.

TODAY’S PLAN • Create a diagram of a Lewis Dot Structure • Differentiate between an element and a compound • Distinguish between a symbol and a formula TODAY’S DO • Quiz: Calculating protons, neutrons, and electrons, • • drawing electronic structures Draw Lewis Dot Structures for elements Differentiate between elements, compounds, and mixtures

Lewis Dot Structures Use dots to represent the number of valence electrons (outermost electrons) in an atom Look at the group number (column number) That is the number of dots that will be around the symbol for the element Maximum = 8 valence electrons = 8 dots Example: Nitrogen = Group 5 = 5 valence electrons = 5 dots

DO NOW Thursday October th 27 Time r How many valence electrons does Tin (Sn) have? Draw its Lewis Dot Structure.

TODAY’S PLAN • Differentiate between an element and a compound • Distinguish between a symbol and a formula • Describe and give examples of physical and chemical • properties. Compare and contrast physical and chemical changes. TODAY’S DO • Differentiate between elements, compounds, and • • mixtures Review physical and chemical properties. Differentiate between physical and chemical changes.

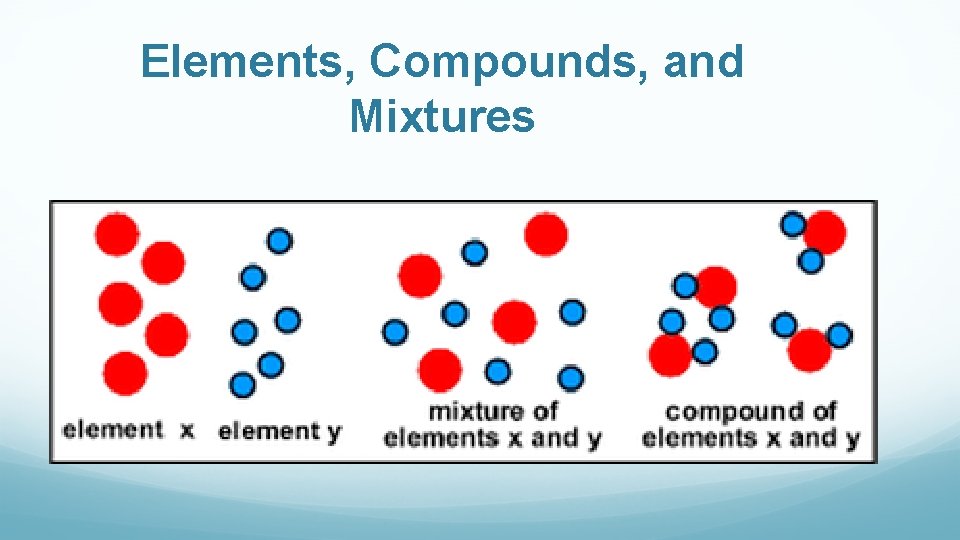
Element Made of only one type of atom Represented by a symbol 1 capital letter Ex. C 1 capital and one lower case letter Ex. Na

Compound Made of 2 or more elements chemically combined (chemical reaction) Consist of a new substance with different properties than the elements it was made of Represented by a chemical formula Will have at least 2 capital letters in the formula Ex. Na. Cl or Ca. CO 3

Mixture Made of 2 or more substances (elements or compounds) physically combined (NOT a chemical reaction) May have the word “and” in the name Two types: 1. Heterogeneous: Appears different, can see different things in it, not evenly mixed Example: cereal and milk, a hamburger 2. Homogeneous : also called a solution Appears the same throughout, evenly mixed Example: sweet tea

Elements, Compounds, and Mixtures

Which is it? ? Element, Compound, Homogeneous Mixture or Heterogeneous Mixture 1. 4. Table sugar (C 6 H 12 O 6) 2. 3. 5. Helium gas (He)

Which is it? ? Element, Compound, or Mixture

Physical and Chemical Properties and Changes

Properties Which type are easily observed or measured? PHYSICAL Give examples of physical properties. Size, shape, color, malleable, ductile, conductor, shiny Which type can only be observed when the identity of a substance is changed? CHEMICAL Give examples of chemical properties. Corrosive, reactive, flammable, explosive, combustible, able to rust

Physical Change A change in the appearance, without changing the identity of the material. It is a physical change if. . . • Can be reversible, or irreversible It changes or size but the way the atoms link • üSubstance mayshape seem different, up It is dissolves. the same. ü ü Or the substance changes phase.

Chemical Changes Chemical changes occur when a substance reacts and forms one or more new substances. You know a chemical change has occurred when there is. . . ü A change in color or odor. ü Production of a gas. (bubbling and fizzing) ü Formation of a precipitate. (solid) ü Absorb or release energy (gets hot or cold).

Examples of Chemical Changes: Burning Molding: Or Combustion : Color change, Color Odorchange, change odor change, Produces a gas, gets hot Decaying : Corroding: Color change, change Odor change Digesting: Color. Rusting change, odor : Change, produces a Color change Gas, releases energy

What kind of change is it? A physical Its still a ribbon, didn’t change its identity.

What kind of change is it? B chemical Its changing wood to ash, color change, odor change, gas produced, gets hot

What kind of change is it? C physical Snow is frozen water, when it melts it’s still water, didn’t change identity.

What kind of change is it? D physical Ice is frozen water, when it melts it’s still water, didn’t change identity.

What kind of change is it? E chemical It’s producing a gas because its bubbling.

What kind of change is it? F physical Even when you paint it, the wall is still a wall made of wood and drywall, didn’t change identity.

DO NOW Friday October th 28 Time r 1. Tell if each property is physical or chemical. A. Color C. Reactivity B. Conductivity D. Solubility 2. What are the four indicators that a chemical change is happening? 3. Tell if each change is physical or chemical. A. Rusting C. Dissolving B. Melting D. Reacting with acid

TODAY’S PLAN • Describe and give examples of physical and chemical • properties. Compare and contrast physical and chemical changes. TODAY’S DO • Conduct Physical and Chemical Changes Lab

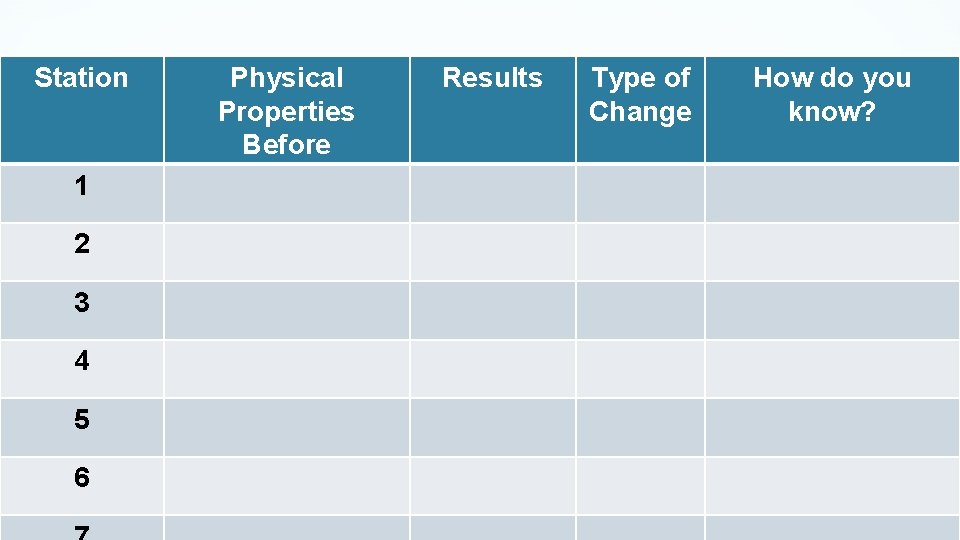
Station 1 2 3 4 5 6 Results Type of Copy this table. Physical Properties Before Change How do you know?

Lab Expectations Stay with your group at your table. Stay on task. Behave appropriately.

DO NOW Tuesday November Time r st 1 Classify each as an element, compound, homogeneous mixture, or heterogeneous mixture: a. b. c. d. Vinegar (H 2 C 2 H 3 O 2) Pancake syrup Chili A copper bracelet (Cu)

TODAY’S PLAN • Conclude the number of atoms of each element in a compound. • Explain the Law of Conservation of Matter. TODAY’S DO • Practice counting the number of atoms of each element in a compound using the compound’s formula. • Discuss chemical equations and the Law of Conservation of Matter.

Counting Atoms The chemical formula for a compound tells us the types of elements in the compound. Subscripts in the formula also tell us the number of atoms of each element in the compound.

Counting Atoms Examples: 1. Cu. O (copper oxide) Cu = 1 atom O = 1 atom 2. Ca. Cl 2 (calcium chloride) Ca = 1 atom Cl = 2 atoms 3. H 2 C 2 H 3 O 2 (vinegar) H = 2 + 3 = 5 atoms C = 2 atoms O = 2 atoms

Counting Atoms Sometimes, the formula for a compound will have a coefficient in front of it. This coefficient should be multiplied by the subscript to determine the number of atoms of each element in the compound. Coefficient

Counting Atoms Examples: 1. 2 Ca. Cl 2 Ca = 2 x 1 = 2 atoms Cl = 2 x 2 = 4 atoms 2. 4 NH 4 OH N = 4 x 1 = 4 atoms H = 4 x (4 + 1) = 20 atoms O = 1 x 4 = 4 atoms

Let’s Practice: Tell the number of atoms of each element in these compounds 1. Sn. Cl 4 4. 2 KCl 2. Cu 2 Se. O 4 5. 3 H 2 C 2 H 3 O 2 3. As. Br 3

Chemical Equations https: //www. brainpop. com/science/matterandchemis try/chemicalequations/

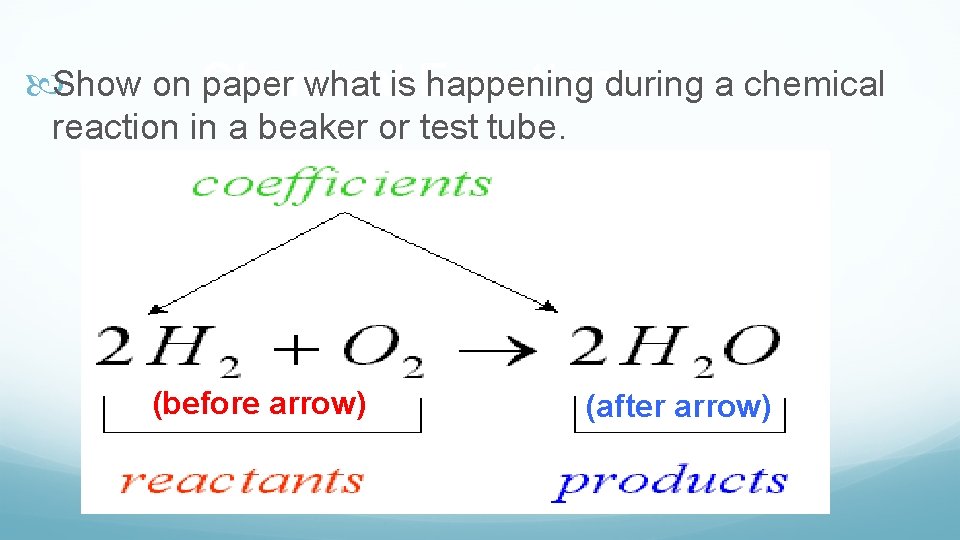
Show on Chemical paper what is Equations happening during a chemical reaction in a beaker or test tube. (before arrow) (after arrow)

Law of Conservation of Matter During a chemical reaction, matter cannot be created or destroyed, only transformed from one form to another. = same number and kinds of atoms/elements on each side of a chemical equation This why we MUST balance chemical equations.

DO NOW Wednesday November nd 2 How many atoms of each element are in the following compounds? 1. H 2 CO 3 2. 3 Na 2 SO 4 3. Why do we have to balance chemical equations?

TODAY’S PLAN • Explain and interpret the Law of Conservation of Matter. • Balance chemical equations. TODAY’S DO • Discuss chemical equations and the Law of Conservation of Matter. • Balance equations.

Balancing Chemical Equations We must put coefficients in front of the formulas for compounds in the equation to make the number of atoms on each side of the equation equal.

Balancing Equations Example _____ Na + _____Cl 2 _____ Na. Cl

Balancing Equations Example _____ Al + _____O 2 _____ Al 2 O 3

Balancing Equations Example

DO NOW Thursday November rd 3 1. Why do we have to balance chemical equations? 2. Balance the following equation. Show your work. ____Na. Br + ____Cl 2 ____Na. Cl + ____Br 2

TODAY’S PLAN • Explain and interpret the Law of Conservation of Matter. • Balance chemical equations. TODAY’S DO • Balance equations. • Exit Slip/Quiz

Balancing Equations Example ____S + ____O 2 ____SO 3

Balancing Equations Example ____N 2 + ____H 2 ____NH 3

Balancing Equations Example __HCl + __Ca. CO 3 __Ca. Cl 2 +__H 2 O + ___CO 2

DO NOW Wednesday November th 9 1. Why do we have to balance chemical equations? 2. Balance the following equation. Show your work. __HCl + __Ca. CO 3 __Ca. Cl 2 +__H 2 O + __CO 2

TODAY’S PLAN • Illustrate Mastery of Chemistry Learning Targets TODAY’S DO • Complete Chemistry Review Sheet