Elements are organized into the Periodic Table of
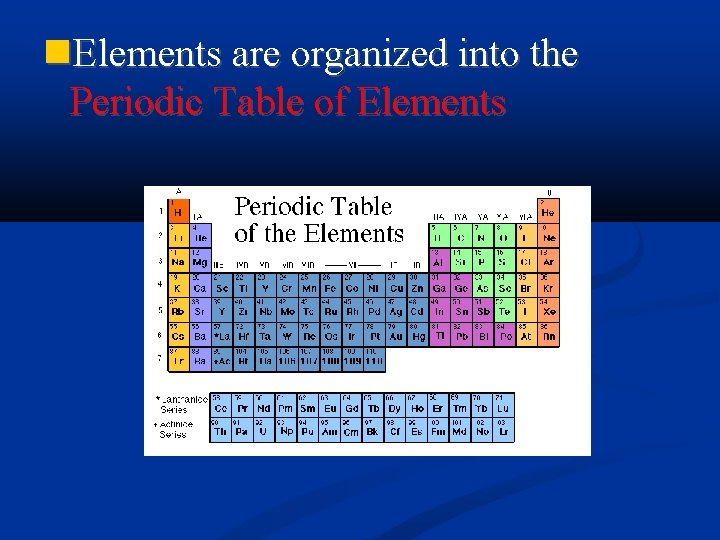
Elements are organized into the Periodic Table of Elements

Organize my grocery list so that I can be more efficient: Peanut butter Milk Ground beef Cucumber Frozen corn Fish sticks strawberries Bread Frozen waffles Cheese Cheerios Sliced ham Ice cream

What are some of the ways you organized the grocery list? Alphabetical Aisle Frozen foods Food groups Animal/plant products Processed/natural foods

• The father of the periodic table • In the 19 th century, a lot of elements were discovered • 1869 - Mendelev organized these elements according to their atomic masses • He left blank spaces for elements that had not yet been discovered • He used rows and columns

PUZZLE
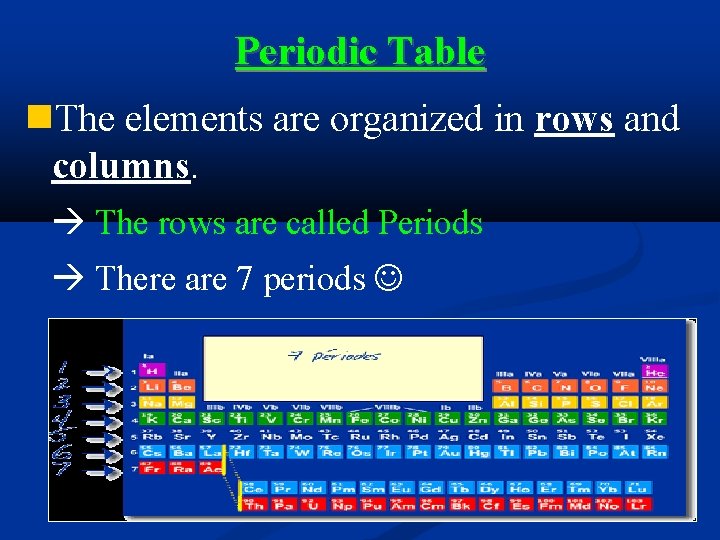
Periodic Table The elements are organized in rows and columns. The rows are called Periods There are 7 periods

Groups/Families A column is called a group or a family There are 18 groups/families

The periodic table has a lot of information organized in different patterns: Groups – vertical columns - elements in the same group have similar chemical properties Periods – horizontal rows - elements have the same number of electron shells Staircase – separates metals and nonmetals

Elements in the same family are similar and have the same chemical properties.

Periodic Table 7 PERIODS 18 FAMILIES

Inferring Symbols for Elements Activity See Page 193 Complete Entire Activity BUT NOT Extensions # 2 -3 See Periodic Table of the Elements on pg. 562 and Appendix D on pg. 564

Rules for Element Symbols 1. First letter of the element name. 2. First letter and another from its name (to differentiate it from other elements with the same first letter). • Examples: Chlorine (Cl), Calcium (Ca), Chromium (Cr), Cobalt (Co), Cesium (Cs). 3. Element initials from its name in language of origin. • Examples: Iron – [Latin, Ferrum] (Fe), Gold – [Latin, Aurum] (Au), Tungsten – [German/Slavik, Wolfram] (W)

Naming Elements were named in the first language of the sciencentist who discovered it. Ex: Iron uses the symbol Fe because it is Ferrous in Latin Ex: Mercury uses the symbol Hg because it is Hydrargyrum in Greek

Naming Elements Find the symbol for the following elements: Chromium Calcium Sodium Oxygen Chlorine Hydrogen

Naming Elements Find the names for the following elements: Cu Ne N Mg U Au

The Atomic Number The number of protons found in the nucleus of the atom Represented by the letter Z Atomic Number is 26 Atomic Number is 1 26 1 H Fe

Atomic Numbers Elements are organized by their atomic numbers Ex: Hydrogen contains 1 proton; therefore, its atomic number is Each elements has its own atomic number

Determine the atomic numbers for the following elements: • • • Hydrogen Beryllium Neon * Lithium * Californium * Lead

Atomic Numbers Find the atomic numbers for the following elements: Tungsten Lead Xenon Fluorine

Atomic Numbers Find the elements for the corresponding atomic number: 16 33 98 102
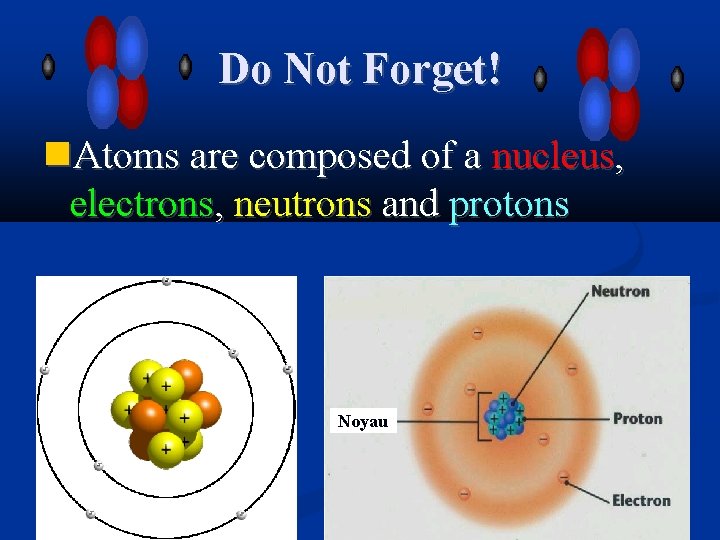
Do Not Forget! Atoms are composed of a nucleus, electrons, neutrons and protons Noyau

Atomic Mass The mass of the protons and neutrons of an atom Measured in amu (atomic mass unit) (u) Proton: mass of Neutron: mass of Electron: mass of
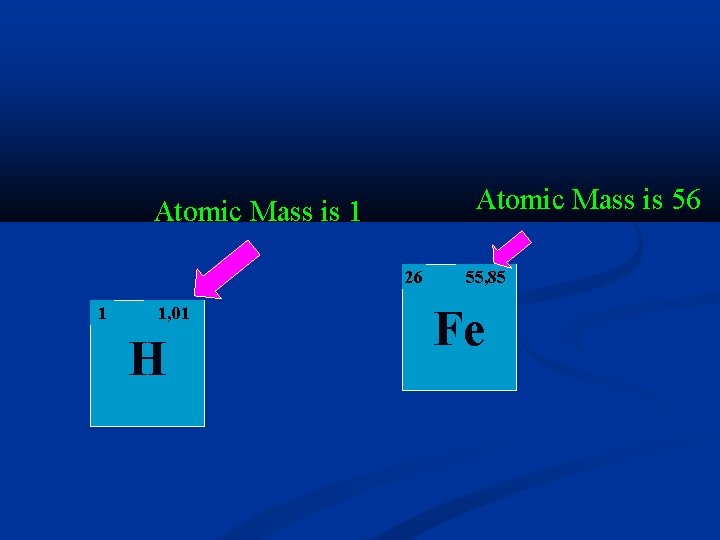
Atomic Mass is 56 Atomic Mass is 1 26 1 1, 01 H 55, 85 Fe


Ex: Lithium (Li) Has 3 protons and 4 neutrons Atomic Mass = # protons + # neutrons Atomic Mass of Lithium: 3 + 4 = 7 Remember: electrons do not have a mass

Review!! The atomic number and the atomic mass are completely different. Atomic # = # of protons (determines the placement on the periodic table) Atomic Mass = # protons + # neutrons Both are given on the period table

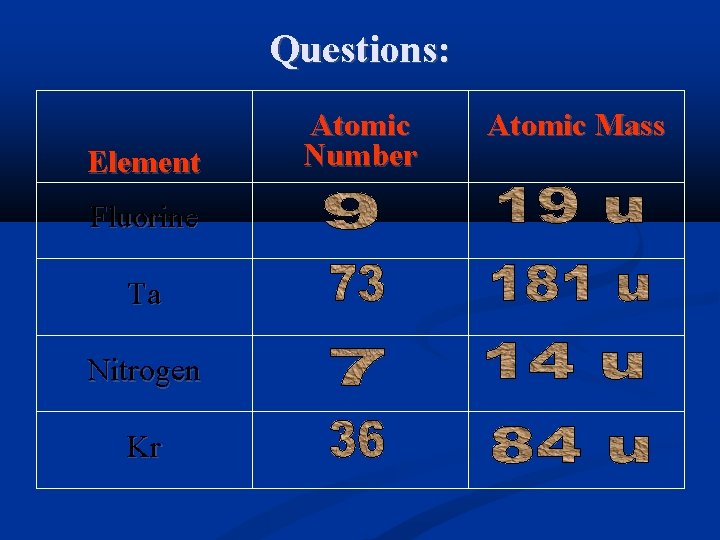
Questions: Element Fluorine Ta Nitrogen Kr Atomic Number Atomic Mass

Standard Atomic Notation

Standard Atomic Notation Aluminium Calcium Chlorine Fluorine

Standard Atomic Notation Helium Magnesium Nitrogen Oxygen
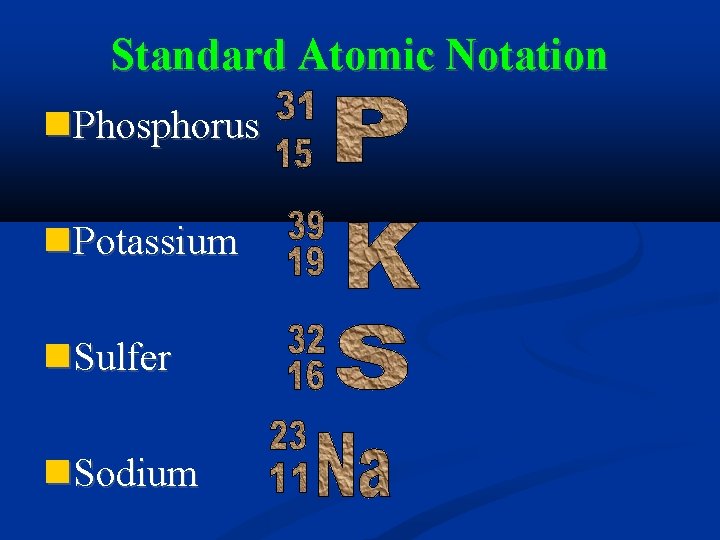
Standard Atomic Notation Phosphorus Potassium Sulfer Sodium

Protons and Electrons Atoms have the same number of protons and electrons Number of Protons = Atomic Number of Electons = Number of Protons # of Neutrons = Atomic Mass - Atomic #

The atomic number of Krypton (Kr) is 36. Kr - 36 protons (therefore) – 36 electrons # of Neutrons = 84 – 36 # of Neutrons = 48 neutrons

23 Vanadium Protons? Electrons? Neutrons? (51 – 23) V Vanadium 50, 94

44 Ruthenium Ru Ruthenium 101, 07 Protons? Electrons? Neutrons? (101 – 44)

Lead Protons? Electrons? Neutrons? (207 – 82)
- Slides: 37