Elements and the Periodic Table There are 3
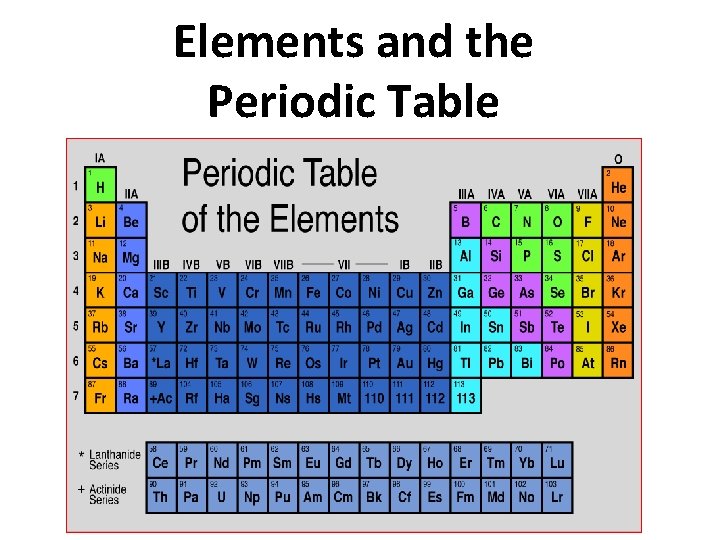
Elements and the Periodic Table

There are 3 categories for the elements 1. Metals 2. Metalloids 3. Nonmetals
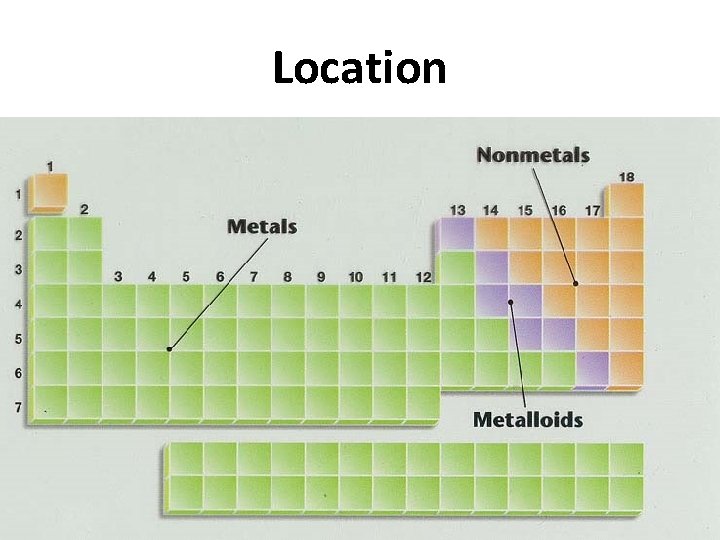
Location

Metals • Good conductors of heat and electricity • Solids at room temperature (except Hg) • Malleable: can be hammered or rolled into thin sheets • Ductile: can be drawn into a fine wire • Tensile strength is high: resists breaking when pulled
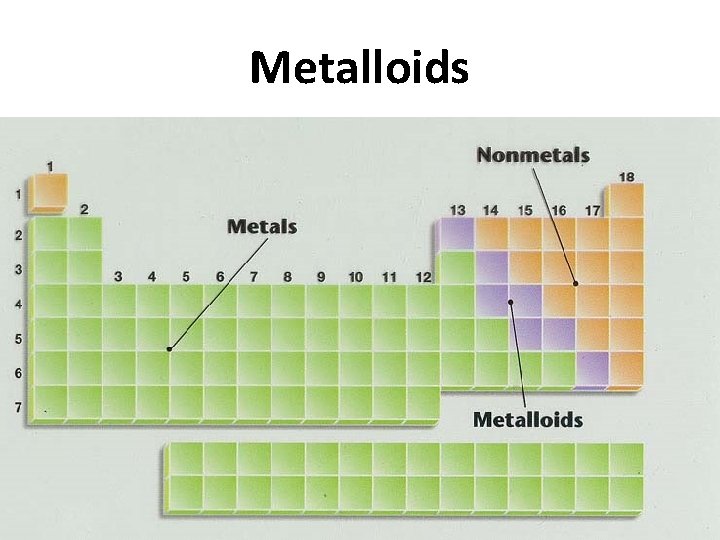
Metalloids

Metalloids • Elements that have some characteristics of metals and some of nonmetals – B, Si, Ge, As, Sb, Te (sometimes At included) • Solids at room temperature • Less malleable than metals • Not as brittle as nonmetals • Semiconductors of electricity
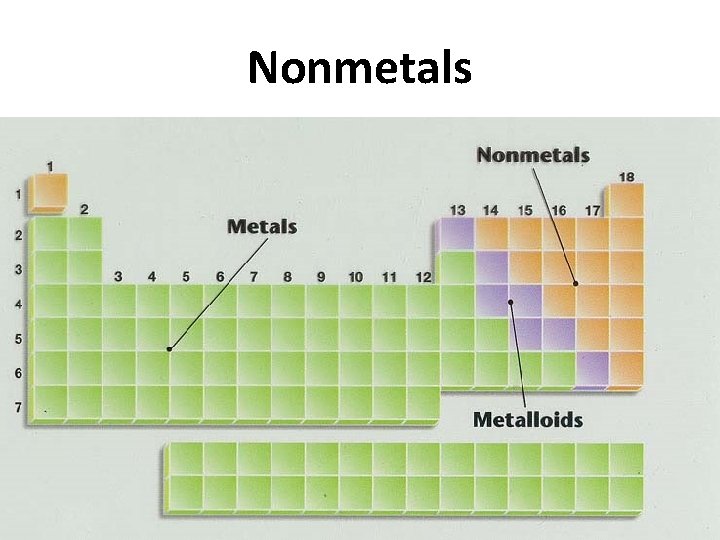
Nonmetals

Nonmetals • Poor conductors • Brittle (if solid) • 1 liquid, 11 gases, 6 solids at room temperature • Located at upper right of periodic table • Hydrogen is only nonmetal on the left side of the periodic table

Group 1 Alkali Metals • 1 electron in outer level • Very reactive (easier for them to bond to other elements creating compounds) • Soft, silver in color, shiny, low density *remember the video I showed you about how these metals react with water and air* *Li, Na, K, Rb, Cs, Fr

Group 2 Alkaline-Earth Metals • Contain 2 electrons in their outer level • Reactive, but not as reactive as Alkali Metals • Silver in color, higher density than alkali metals *Be, Mg, Ca, Sr, Ba, Ra Uses: Chalk, cement, bones, teeth all have Calcium in them. Magnesium can be combined with other low-density metals to build airplanes.

Groups 3 -12 Transition Metals • Can contain either 1 or 2 electrons in their outer level. • Less reactive than alkaline-earth metals • Shiny, good conductors of heat and electricity • Higher densities • Higher melting points than groups 1 and 2 except Mercury (Hg) • Uses: structural materials, jewelry, coins.
Transition Metals

Group 13 Boron Group Contains one metalloid and 5 metals 3 electrons in outer level Reactive Solid at room temperature Aluminum is the most common metal in Earth’s crust. *B, Al, Ga, In, Ti, • • •

Group 14 Carbon Group • One nonmetal, two metalloids, and three metals • 4 electrons in outer level • Reactivity varies among elements • Solid at room temperature *CARBON-so important, why? ? ? *C, Si, Ge, Sn, Pb,

Group 15 Nitrogen Group • Group contains two nonmetals, two metalloids, and two metals • 5 electrons in outer level • Reactivity varies among elements • Solid at room temperature except Nitrogen *Nitrogen is the most abundant gas in our atmosphere! *Phosphorus is extremely reactive for a nonmetal, it’s used to make matches! *N, P, As, Sb, Bi

Group 16 Oxygen Group Three nonmetals, two metalloids, and one metal 6 electrons in the outer level Reactive Solid at room temperature (except oxygen) 21% of our atmosphere is composed of oxygen! Oxygen is necessary for anything to burn, it is also important to most living things. • Sulfur is utilized greatly in the chemical industry in the form of sulfuric acid. *O, S, Se, Te, Po, Lv • • •

Group 17 Halogens Nonmetals Contain 7 electrons in outer level Very reactive Poor conductors of heat and electricity Violent reactions with alkali metals to form salts (never uncombined in nature) *uses: Chlorine and Iodine are used as disinfectants *F, Cl, Br, I, At • • •

Group 18 Noble Gases • Nonmetals • Contains 8 electrons in their outer level, except Helium which has 2. • Unreactive • Colorless, odorless gas at room temperature. • Argon is the most common noble gas on Earth, makes up 1% of our atmosphere. All other gases are called trace gases, because they are found in such small amounts. • *He, Ne, Ar, Kr, Xe, Rn

Hydrogen Nonmetal Has only one electron in outer level Very reactive Colorless, odorless gas at room temperature, explosive reaction with oxygen. • Low density *Most abundant element in our universe (stars)! • •
Lanthanides and Actinides

Lanthanides and Actinides • These metals are part of the transition metals. • They are located at the bottom of the periodic table. • Often mixed with other metals to make them stronger. • Most well-known actinide is uranium. • http: //chemistry. about. com/od/elementgrou ps/a/rareearths. htm (more facts about these two groups)

Synthetic (Man-made) Elements • Many of the very large elements are not found naturally on Earth. • Atomic numbers are greater than 92. • These elements are made by forcing nuclear particles together (fusion). • Ex. Uranium (92) and carbon (6) are fused together to create californium (98).

Trends in the Periodic Table Trend: a predictable change in a particular direction We will study 5 periodic trends
- Slides: 23