Elements and Compounds elements combine together to make

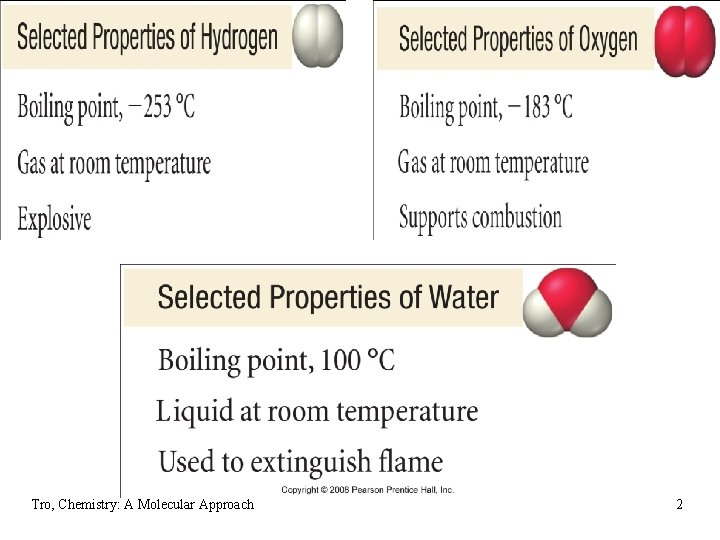
Elements and Compounds • elements combine together to make an almost • limitless number of compounds the properties of the compound are totally different from the constituent elements • H 2(g) + O 2(g) H 2 O(l) Tro, Chemistry: A Molecular Approach 1
Tro, Chemistry: A Molecular Approach 2
Tro, Chemistry: A Molecular Approach 3

Formation of Water from Its Elements Tro, Chemistry: A Molecular Approach 4

Allotropes: one of two or more forms of an element that differ in their basic structure diamond Tro, Chemistry: A Molecular Approach graphite 5

Allotropes of carbon diamond Tro, Chemistry: A Molecular Approach graphite 6

Allotrope of carbon 7
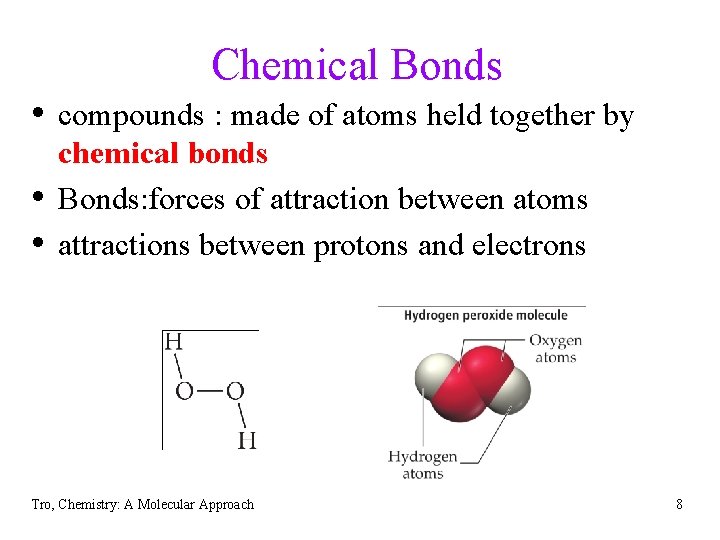
Chemical Bonds • compounds : made of atoms held together by • • chemical bonds Bonds: forces of attraction between atoms attractions between protons and electrons Tro, Chemistry: A Molecular Approach 8
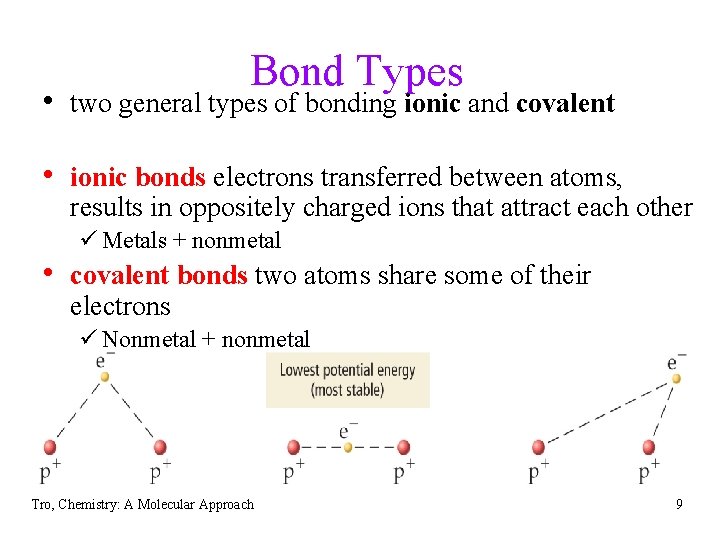
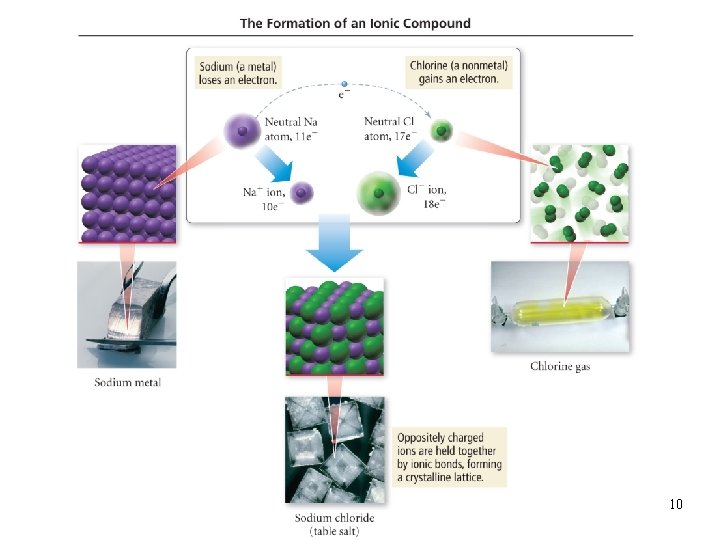
Bond Types • two general types of bonding ionic and covalent • ionic bonds electrons transferred between atoms, results in oppositely charged ions that attract each other ü Metals + nonmetal • covalent bonds two atoms share some of their electrons ü Nonmetal + nonmetal Tro, Chemistry: A Molecular Approach 9
10
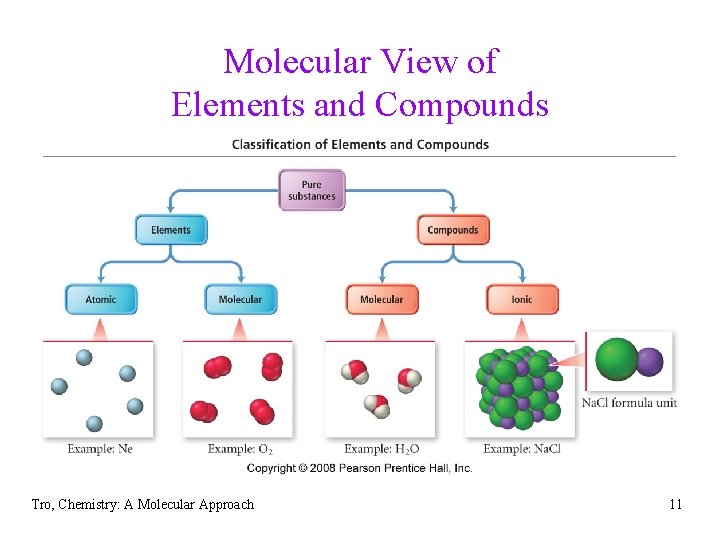
Molecular View of Elements and Compounds Tro, Chemistry: A Molecular Approach 11
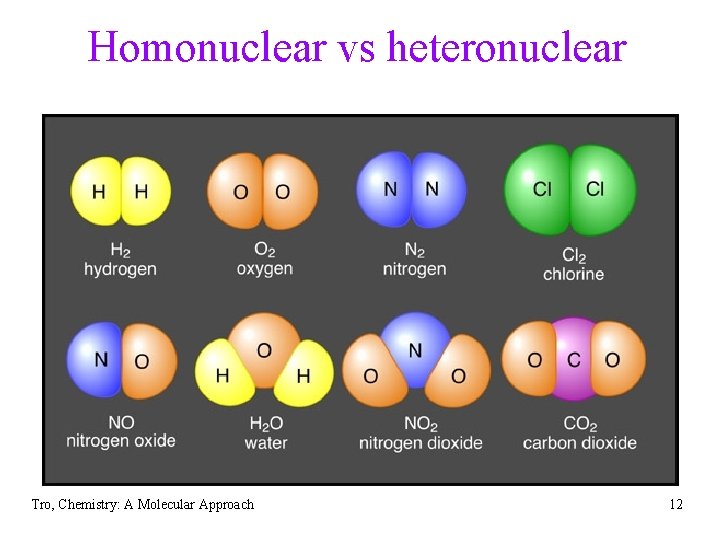
Homonuclear vs heteronuclear Tro, Chemistry: A Molecular Approach 12
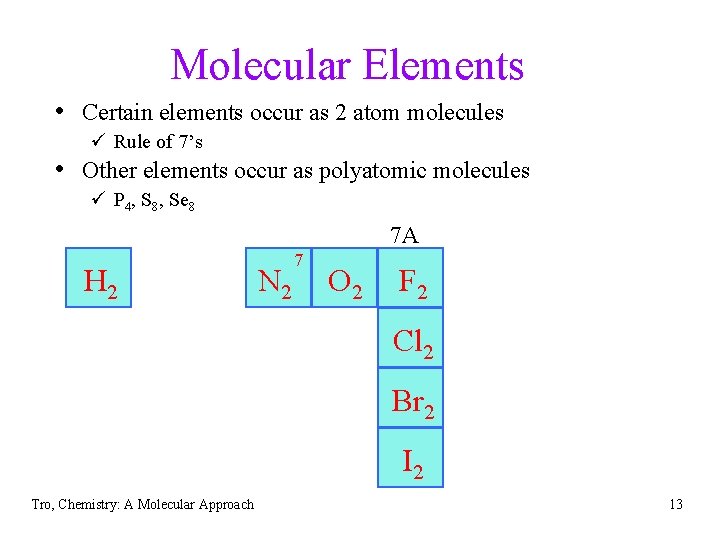
Molecular Elements • Certain elements occur as 2 atom molecules ü Rule of 7’s • Other elements occur as polyatomic molecules ü P 4, S 8, Se 8 7 A H 2 7 N 2 O 2 F 2 Cl 2 Br 2 I 2 Tro, Chemistry: A Molecular Approach 13

Classifying Materials • atomic elements = Fe • molecular elements = N 2 • molecular compounds = H 2 O • ionic compounds = Sr. Cl 2 Mg. CO 3 Tro, Chemistry: A Molecular Approach 14
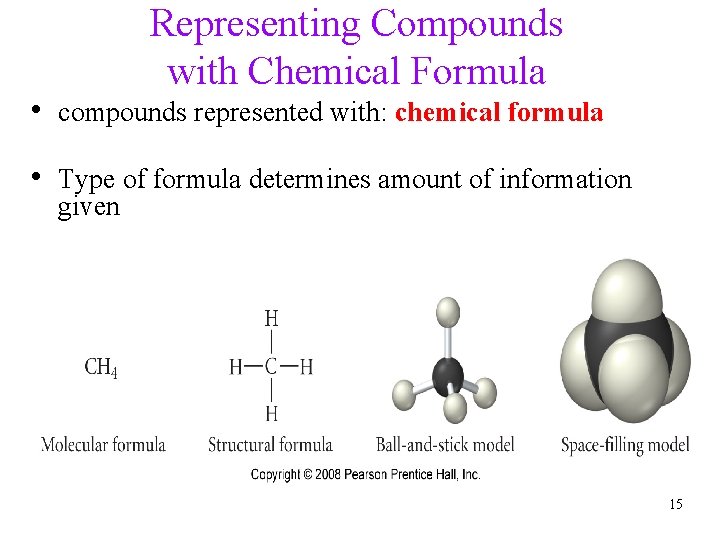
Representing Compounds with Chemical Formula • compounds represented with: chemical formula • Type of formula determines amount of information given 15

Types of Formula Empirical Formula • Empirical Formula • Tells atoms and their ratio üdo not describe how many atoms, the order of attachment, or the shape üthe formulas for ionic compounds are empirical 16
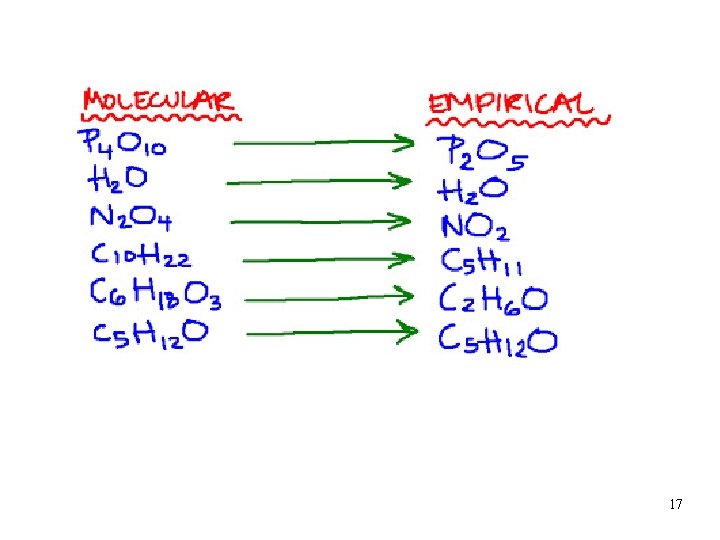
17

Empirical formula Molecular formula • HO • H 2 O 2 • _____ • C 8 H 8 • _____ • C 6 H 12 O 6 • H 2 O • __________ • Hg 2 Cl 2 18
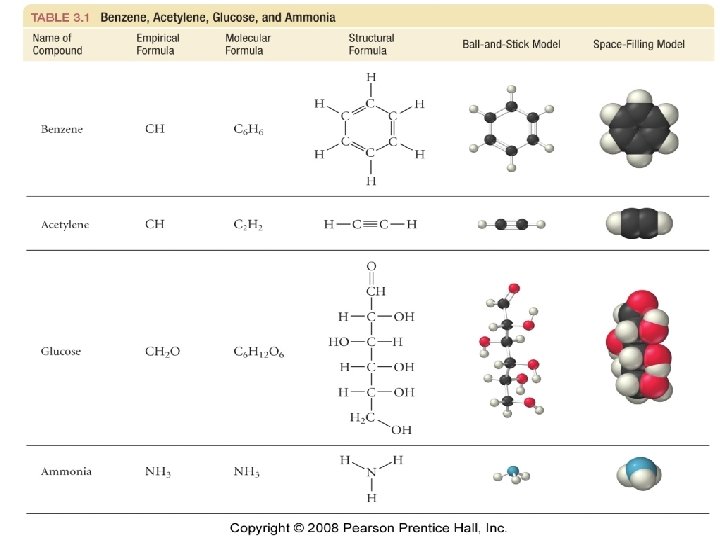
Types of Formula 19
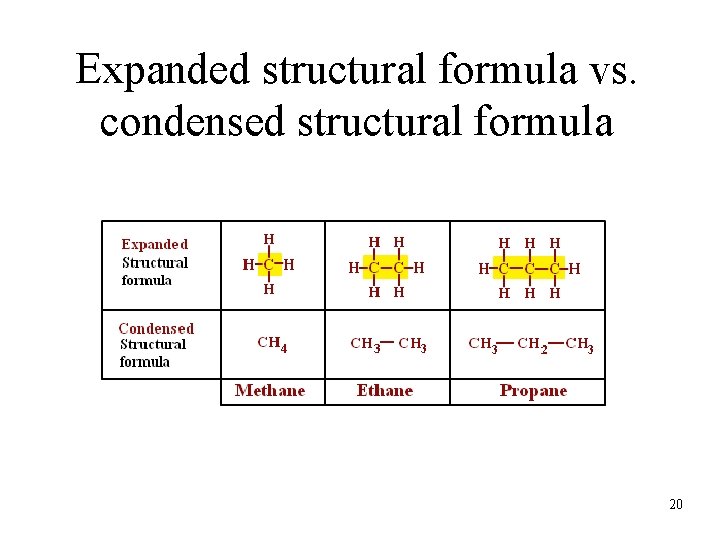
Expanded structural formula vs. condensed structural formula 20

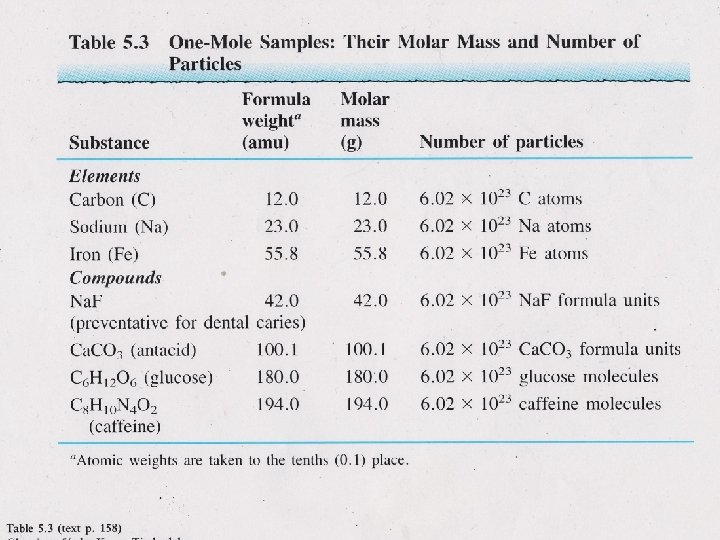
Formula Mass • the mass of an individual molecule or formula unit (FW) amu (D for Dalton) • molecular mass or molecular weight (MW or μ) g/mol • sum of the masses of the atoms in a single molecule or formula unit mass of 1 molecule of H 2 O = 2(1. 01 amu H) + 16. 00 amu O = 18. 02 amu Tro, Chemistry: A Molecular Approach 21

What Is a Mole? Tro, Chemistry: A Molecular Approach 22


How Big is a Mole? • N=6. 022 x 1023 • One mole of • Marshmallows would cover the entire earth 12 miles high One mole of marshmallows would fill the Grand Canyon and still be enough left over to displace all the water from Lake Michigan and more 24

Mole = 6. 022 x 1023 items Avogadro’s number N 25

Molar Mass of Compounds • the relative masses of molecules can be calculated • from atomic masses Formula Mass = 1 molecule of H 2 O = 2(1. 01 amu H) + 16. 00 amu O = 18. 02 amu since 1 mole of H 2 O contains 2 moles of H and 1 mole of O Molar Mass = 1 mole H 2 O = 2(1. 01 g H) + 16. 00 g O = 18. 02 g so the Molar Mass of H 2 O is 18. 02 g/mole Tro, Chemistry: A Molecular Approach 26

Moles as Conversion factors • Grams to moles • Use Molar Mass • Ex: How many • Moles to molecules or • moles are in 9. 01 g • of water? atoms Use Avogadro’s number Ex: How many molecules of water are in 9. 01 g of water? 27

Moles • How many molecules of Chlorine are in 19. 5 grams of chlorine? • How many moles of sodium atoms is 29. 0 grams of sodium? Tro, Chemistry: A Molecular Approach 28
Tro, Chemistry: A Molecular Approach 29

Molecular formula • In 5. 0 moles of the compound Mg(H 2 PO 4)2 • How many moles of Mg are there? ____ • How many moles of phosphorus are there? ___ • How many moles of oxygen are there? ______ Tro, Chemistry: A Molecular Approach 30

Molar mass • What is the formula weight (FW) of Mg(H 2 PO 4)2 including units? ____ • What is the molar mass (MW or µ) of Mg(H 2 PO 4)2 including units? _____ Tro, Chemistry: A Molecular Approach 31

Practice - Converting Grams to Molecules How many molecules are in 50. 0 g of Pb. O 2? (Pb. O 2 = 239. 2 g/mol) Tro, Chemistry: A Molecular Approach 32

Percent Composition • Percentage of each element in a compound ü Mass of element/mass of compound • The percentages may not always total to 100% due to rounding Tro, Chemistry: A Molecular Approach 33
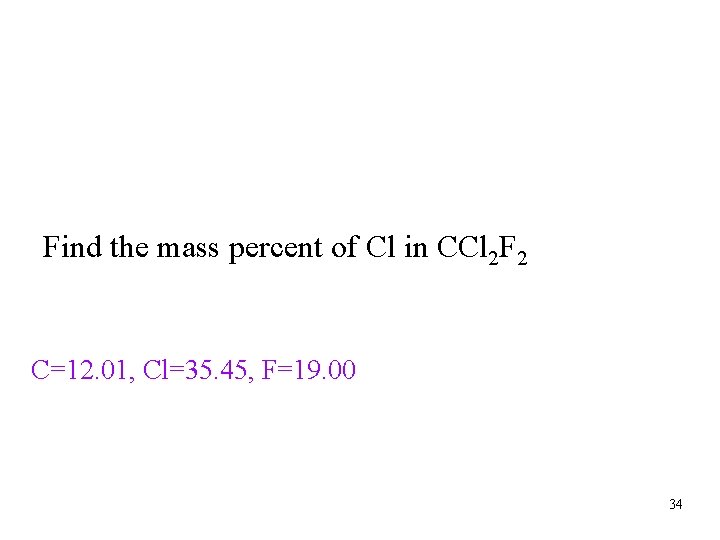
Find the mass percent of Cl in CCl 2 F 2 C=12. 01, Cl=35. 45, F=19. 00 34

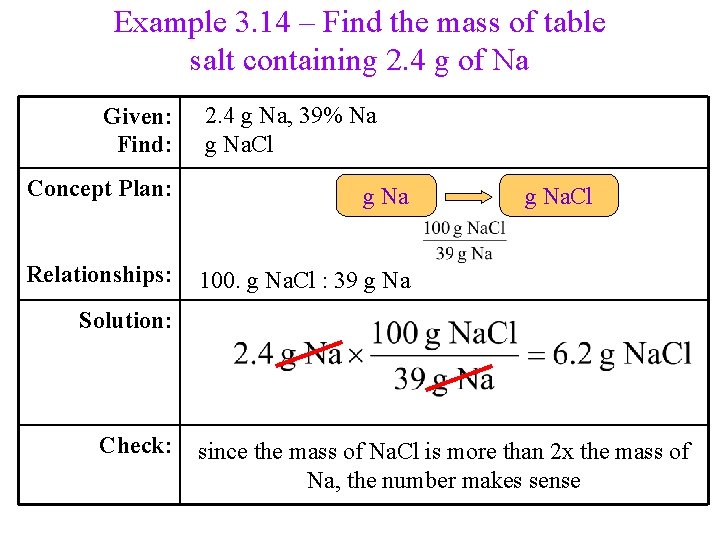
Mass Percent as a Conversion Factor • the mass percent tells you the mass of a constituent • element in 100 g of the compound üthe fact that CCl 2 F 2 is 58. 64% Cl by mass means that 100 g of CCl 2 F 2 contains 58. 64 g Cl this can be used as a conversion factor ü 100 g CCl 2 F 2 : 58. 64 g Cl
Example 3. 14 – Find the mass of table salt containing 2. 4 g of Na Given: Find: 2. 4 g Na, 39% Na g Na. Cl Concept Plan: g Na Relationships: 100. g Na. Cl : 39 g Na. Cl Solution: Check: since the mass of Na. Cl is more than 2 x the mass of Na, the number makes sense
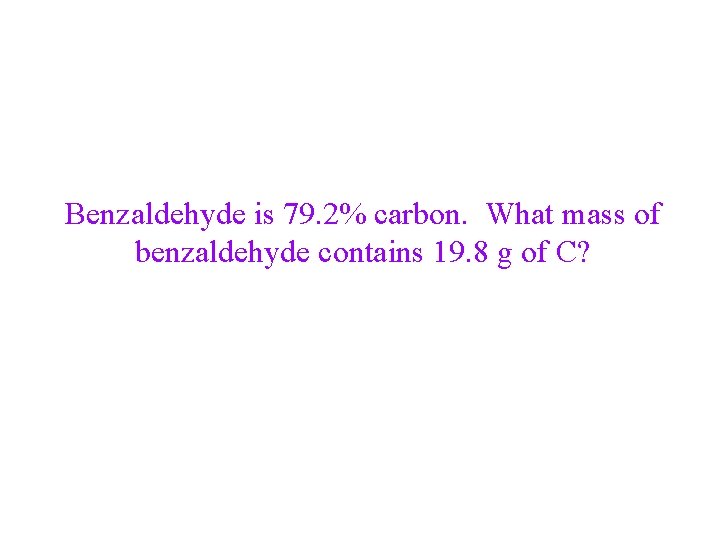
Benzaldehyde is 79. 2% carbon. What mass of benzaldehyde contains 19. 8 g of C?

Empirical Formula • simplest, whole-number ratio of the atoms of • elements in a compound can be determined from elemental analysis ümasses of elements formed when decompose or react compound Øcombustion analysis üpercent composition Tro, Chemistry: A Molecular Approach 39

Finding an Empirical Formula 1) convert the percentages to grams a) b) assume you start with 100 g of the compound skip if already in grams 2) convert grams to moles a) use molar mass of each element a) if result is within 0. 1 of whole number, round to whole number 3) write a pseudoformula using moles as subscripts 4) divide all by smallest number of moles 5) multiply all mole ratios by number to make all whole numbers a) if ratio ? . 5, multiply all by 2; if ratio ? . 33 or ? . 67, multiply all by 3; if ratio 0. 25 or 0. 75, multiply all by 4; etc. Tro, Chemistry: A Molecular Approach 40 b) skip if already whole numbers

Calculating Empirical formula • 1. A sample of an unknown compound with a mass of 2. 571 g was found to contain 1. 102 g of carbon and 1. 469 g of oxygen. What is its empirical formula? 41

Calculating empirical formula • An oxide of iron called black iron oxide, is analyzed. In a 2. 448 g sample the ore was found to contain 1. 771 g of Fe and 0. 6766 g of O. Calculate the empirical formula of this compound

Example 3. 17 • Laboratory analysis of aspirin determined the following mass percent composition. Find the empirical formula. C = 60. 00% H = 4. 48% O = 35. 53%
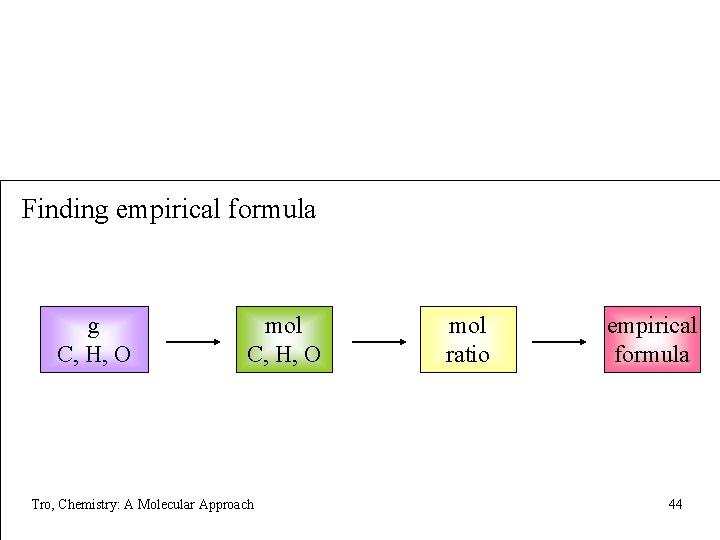
Finding empirical formula g C, H, O mol C, H, O Tro, Chemistry: A Molecular Approach mol ratio empirical formula 44

Determine the empirical formula of hematite, which contains 72. 4% Fe (55. 85) and the rest oxygen (16. 00) Tro, Chemistry: A Molecular Approach 46

Molecular Formulas • The molecular formula is a multiple of the • empirical formula To determine the molecular formula you need to know the empirical formula and the molar mass of the compound

Example 3. 18 – Find the molecular formula of butanedione empirical formula for butanedione = C 2 H 3 O; MW of molecular formula = 86. 03 g/mol

Benzopyrene has a molar mass of 252 g/mol and an empirical formula of C 5 H 3. What is its molecular formula? (C = 12. 01, H=1. 01)

hydrates

Hydrates • When the hydrate of Ru. Cl 3 is heated water is • • driven off to form the anhydrous salt as shown below: Ru. Cl 3. x. H 2 O Ru. Cl 3 + x. H 2 O If 1. 056 g of hydrated compound is heated and 0. 838 g of anhydrous salt remains what is the value of x? 51

Naming hydrates: name the ionic compound then the water part, use prefixes for number of water molecules Ionic formula • x H 2 O x = prefix (di, tri etc from covalent naming) H 2 O = hydrate Ex: Cu. SO 4 • 5 H 2 O copper(II) sulfate pentahydrate Ex. Ca. Cl 2 • 2 H 2 O calcium chloride dihydrate
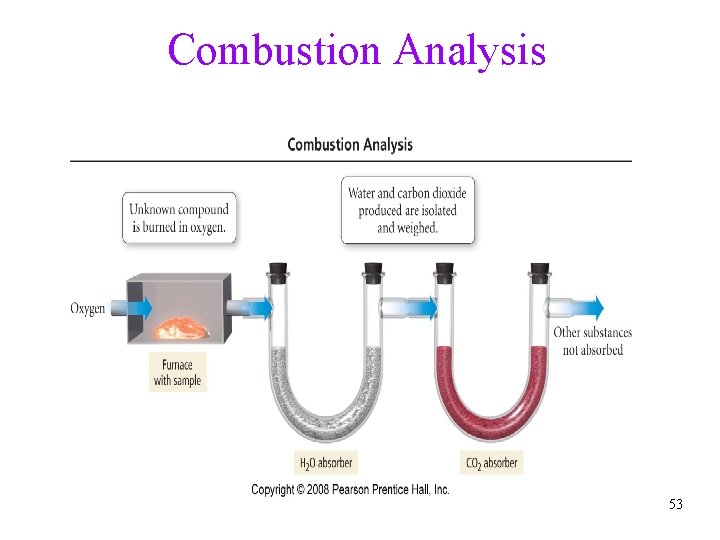
Combustion Analysis 53

Example 3. 20 • Combustion of a 0. 8233 g sample of a compound containing only carbon, hydrogen, and oxygen produced the following: CO 2 = 2. 445 g H 2 O = 0. 6003 g Determine the empirical formula of the compound Tro, Chemistry: A Molecular Approach 54
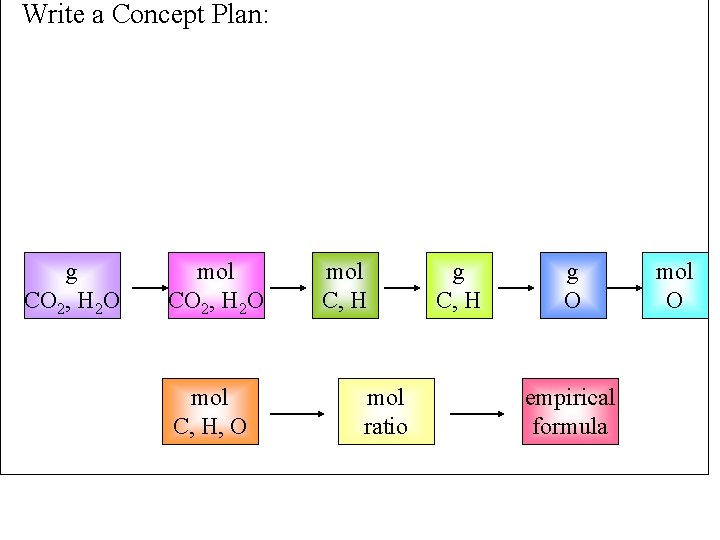
Write a Concept Plan: g CO 2, H 2 O mol C, H, O mol C, H mol ratio g C, H g O empirical formula mol O

Information Given: 0. 8233 g compound, 2. 445 g CO 2, 0. 6003 g H 2 O, Find: Empirical Formula, Cx. Hy. Oz 0. 05556 mol C, 0. 6673 g C, 0. 06662 mol H, 0. 06715 g H, 0. 0889 g O, 0. 00556 mol O Example 3. 20: Find the empirical formula of compound with the Plan: g CO 2 & H 2 O mol C & H given amounts of g C + H g O mol ratio emp. formula combustion products Tro, Chemistry: A Molecular Approach 56

The smell of dirty gym socks is caused by the compound caproic acid. Combustion of 0. 844 g of caproic acid produced 0. 784 g of H 2 O and 1. 92 g of CO 2. If the molar mass of caproic acid is 116. 2 g/mol, what is the molecular formula of caproic acid? (μ C = 12. 01, H = 1. 008, O = 16. 00)
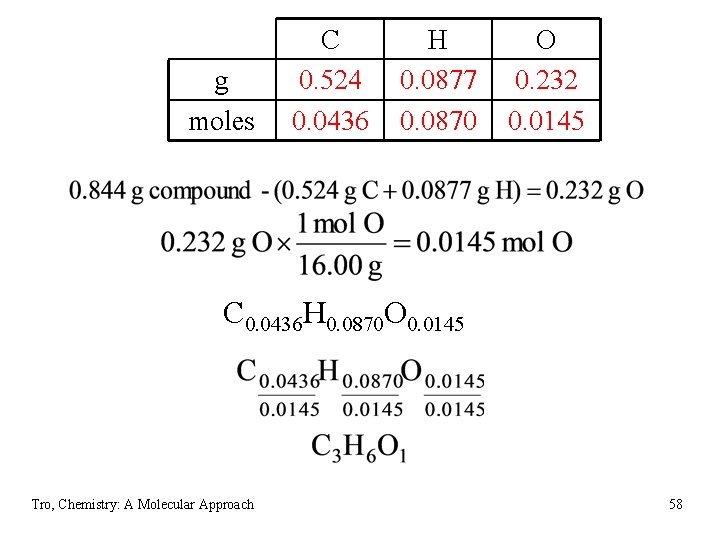
g moles C 0. 524 0. 0436 H 0. 0877 0. 0870 O 0. 232 0. 0145 C 0. 0436 H 0. 0870 O 0. 0145 Tro, Chemistry: A Molecular Approach 58

Molecular Formula = {C 3 H 6 O} x 2 = C 6 H 12 O 2 Tro, Chemistry: A Molecular Approach 59

1. Nicotine, a poisonous compound found in tobacco leaves, is 74. 0% C, 8. 65% H, and 17. 35% N. It has a molar mass of 162 g/mol. What is the molecular formula of nicotine? 2. The “average” cigarette in the US contains 9. 0 mg of nicotine but during smoking about 90% of the nicotine is burned off. How many molecules of nicotine are inhaled during smoking of one “average” cigarette?


Other fun things to do with moles Molarity

What is the molarity of a solution prepared by dissolving 15. 0 g of sodium hydroxide in enough water to make a total of 225 m. L of solution?

Practice Problems 1. What is the molarity of a solution that contains 1. 724 moles of H 2 SO 4 in 2. 50 L of solution? 2. What is the molarity of a solution prepared by dissolving 25. 0 g of HCl (g) in enough water to make 150. 0 m. L of solution?
How would you prepare 1 liter of a 0. 25 M (molar) solution of glucose (C 6 H 12 O 6) (molar mass 180 g/mol)?
- Slides: 63