ELEMENTS AND COMPOUNDS BUTTONS Click here Clicking here

ELEMENTS AND COMPOUNDS BUTTONS Click here; Clicking here will allow you tocommon hear some information onhere the Clicking here will take you to ayou ion table. Clicking here will take to aback periodic table, clicking here Clicking here will reveal some information. Clicking here will move you to the next Clicking here will reveal an answer. Clicking here will bring you topage. this page. Clicking here will move you back a page. topic. Clicking here again willpage stop the sound. again will take you back toback the were on previously. again will take you to theyou previous page. TOPICS Jump to… …elements and compounds. Jump to… …chemical formula. Jump to… …working out the formula (two pages). Jump to… …practice questions (two pages). Jump to… …exam paper questions (four pages).

ELEMENTS AND COMPOUNDS ELEMENTS An element is a substance that cannot be broken down into anything simpler by chemical means. They are the building blocks of everything you see around you. COMPOUNDS Compounds are substances made of two or more different types of atoms. The atoms in a compound are chemically bonded to each other and are difficult to separate. Each individual element is made up of just one type of atom, e. g. • sulfur just contains sulfur atoms; S S S The elements iron and sulfur combine to make the compound iron sulfide. • whilst iron just contains iron atoms; Every compound has a chemical formula which shows the number of each element making it up, e. g. iron sulfide is Fe. S. Fe Fe Fe S Fe Fe S S Fe
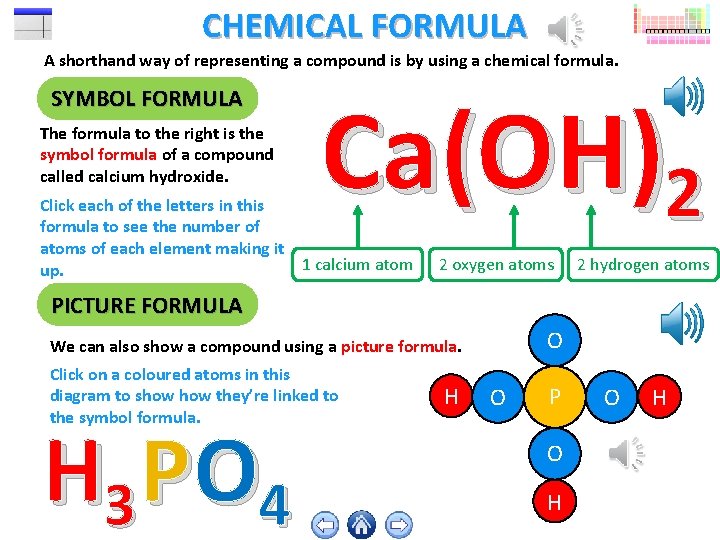
CHEMICAL FORMULA A shorthand way of representing a compound is by using a chemical formula. SYMBOL FORMULA The formula to the right is the symbol formula of a compound called calcium hydroxide. Ca(OH)2 Click each of the letters in this formula to see the number of atoms of each element making it 1 calcium atom up. 2 oxygen atoms 2 hydrogen atoms PICTURE FORMULA O We can also show a compound using a picture formula. Click on a coloured atoms in this diagram to show they’re linked to the symbol formula. H 3 P O 4 H O P O H

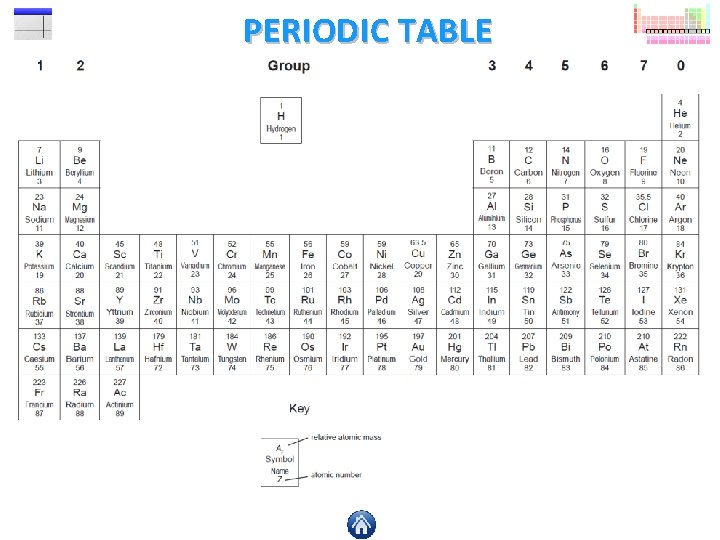
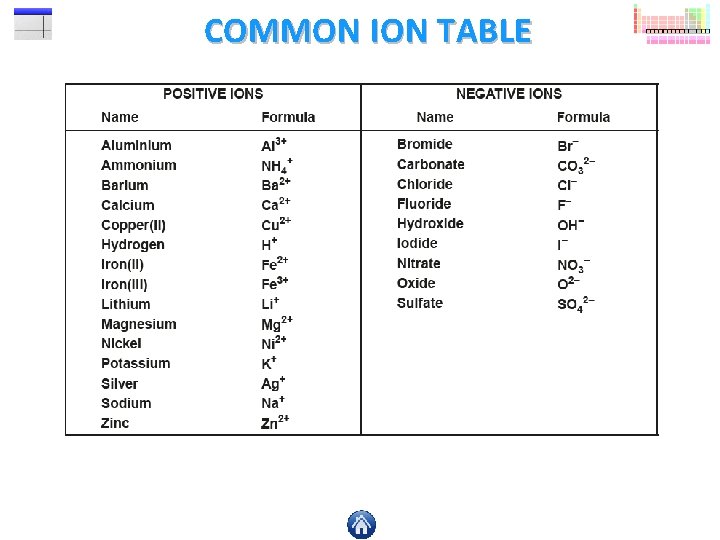
WORKING OUT THE FORMULA 1 What is the formula of iron(III) oxide? STEP 1 Use a common ion table to find the ions for the elements in the formula, so here we have iron(III) (Fe 3+) and oxide (O 2 -). STEP 2 Rub out the + and – charges, leaving just the numbers above each symbol. STEP 3 Now move the numbers diagonally downwards so that they are sitting just below their opposite symbol. STEP 4 You should now have the symbol formula for the compound iron(III) oxide. 3+ 2 - Fe O

WORKING OUT THE FORMULA 2 What is the formula of aluminium sulfate? STEP 1 STEP 2 3+ STEP 3 24 STEP 4 Al (SO ) Two or more of the following will require brackets in your formula; sulfate ion (SO 42 -) carbonate ion (CO 32 -) nitrate ion (NO 3 -) hydroxide ion (OH-) ammonium ion (NH 4+)
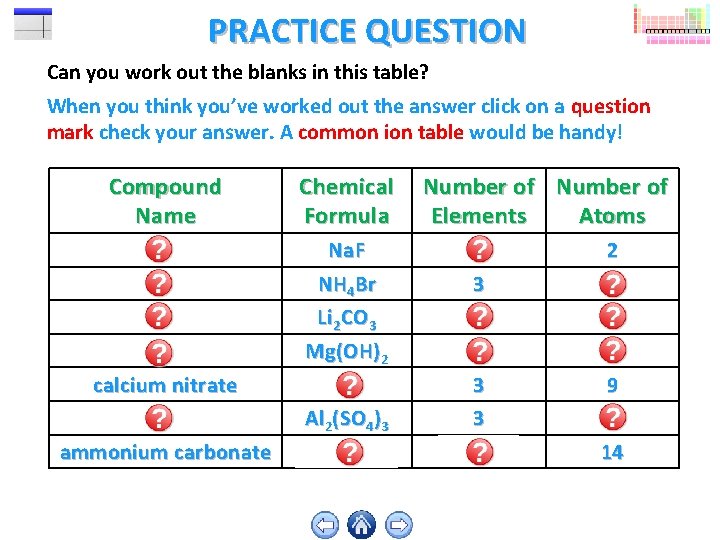
PRACTICE QUESTION Can you work out the blanks in this table? When you think you’ve worked out the answer click on a question mark check your answer. A common ion table would be handy! Compound Name Chemical Formula Number of Elements Atoms sodium fluoride ammonium bromide Na. F NH 4 Br 2 3 2 6 lithium carbonate magnesium hydroxide Li 2 CO 3 Mg(OH)2 3 3 6 5 calcium nitrate aluminium sulfate Ca(NO 3)2 Al 2(SO 4)3 3 3 9 17 ammonium carbonate (NH 4)2 CO 3 4 14
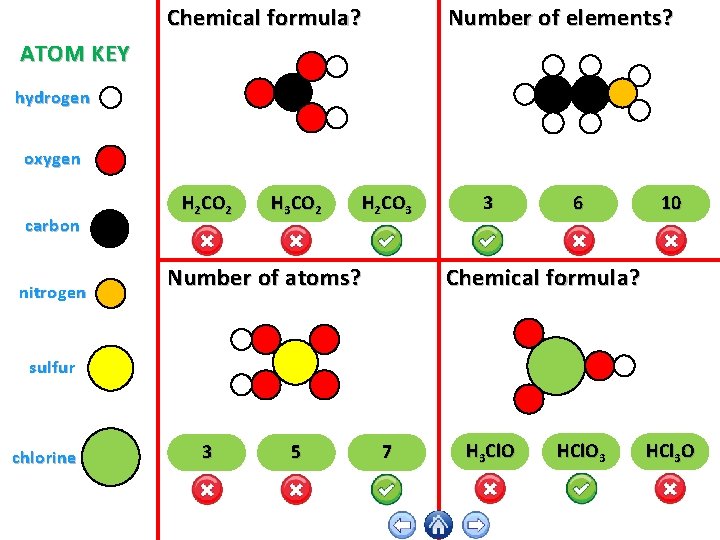
Chemical formula? Number of elements? ATOM KEY hydrogen oxygen carbon nitrogen H 2 CO 2 H 3 CO 2 H 2 CO 3 Number of atoms? 3 6 10 Chemical formula? sulfur chlorine 3 5 7 H 3 Cl. O HCl. O 3 HCl 3 O

EXAM QUESTION 1 Click the answers to see how many marks each response has been awarded by the examiner; 1 mark 0 marks The calcium is correct, but the other element is sulfur, not sulfide. 0 marks The ion for sodium is Na+, and oxide is O 2 -, so the correct formula is Na 2 O.
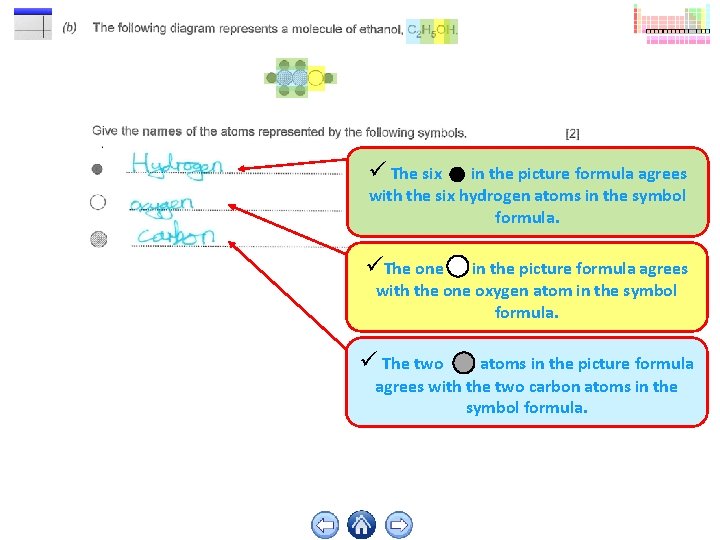
The six in the picture formula agrees with the six hydrogen atoms in the symbol formula. The one in the picture formula agrees with the one oxygen atom in the symbol formula. The two atoms in the picture formula agrees with the two carbon atoms in the symbol formula.

EXAM QUESTION 2 Try to answer these exam questions. Once you’ve had a go, click on the to see the answer the examiner was looking for; CH 4 or H 4 C button

but not 1 6
PERIODIC TABLE
COMMON ION TABLE
- Slides: 13