Element Compound Mixture Models Formulas 100 Bonding Periodic
Element, Compound Mixture Models & Formulas 100 Bonding Periodic Table Equations 100 100 200 200 200 300 300 300 400 400 400 500 500 500

Two or more substances in the same place but not chemically combined are called this.

mixture 1 pt

The Earth’s atmosphere is composed of a bunch of different gasses and is an example of this.

mixture 2 pt

H 2 O is the formula for water and is an example of this.

compound 3 pt

Chemical formulas are used to represent this.

compound 4 pt

Pure substances are considered pure because they are made from only one type of …….

Particle/Element 5 pt

The number of sulfur atoms in the formula H 2 SO 4.

1 1 pt

A molecule of silver bromate is composed of 3 oxygen atoms (O), 1 bromine atom (Br), and 1 silver atom (Ag). This is the chemical formula for silver bromate.
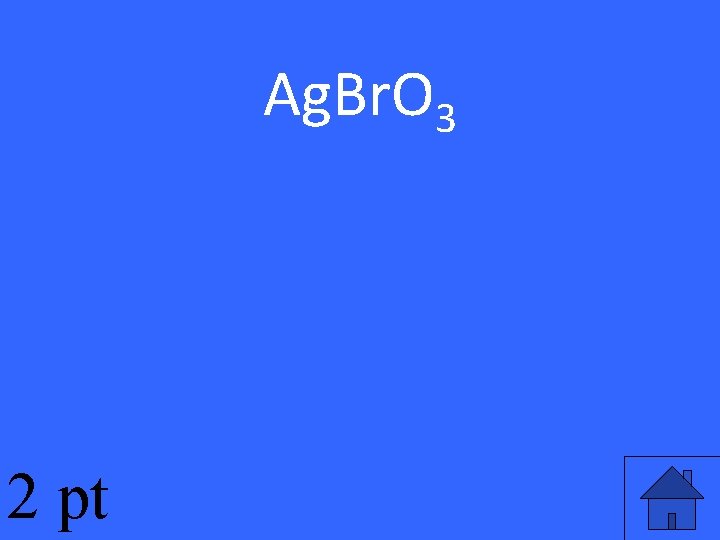
Ag. Br. O 3 2 pt

Which model below shows a compound? A B C

A 3 pt

Why does model C below show an element? A B C

It’s made from only 1 type of atom. 4 pt
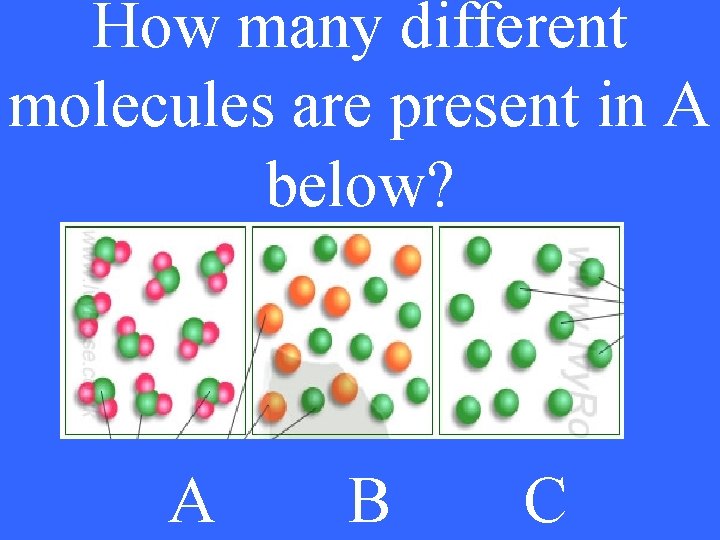
How many different molecules are present in A below? A B C

1 5 pt

Which of the following compounds is ionic? Li. F or C 2 H 4

Li. F 1 pt

This type of bond occurs when electrons are shared between atoms.

covalent 2 pt

When atoms gain or lose electrons and become charged particles, they are called this.

ion 3 pt

The joining of atoms to form new bonds is a result of this.

Chemical bonding 4 pt

When new substances form as a result of chemical bonding, most of their properties do this.

change 5 pt

Elements in the same group on the periodic table of elements have the same number of these.

Valence electrons 1 pt

Noble gasses don’t form bonds because of this.

They have full outer shells or 8 valence electrons and are stable. 2 pt

The number of valence electrons for an element can be found by using this on the periodic table of elements.

The group number. 3 pt

Why do elements in group 1 react so easily?

They only have 1 valence electron. 4 pt

Group 16 elements bond easily with this group.

Group 2 5 pt
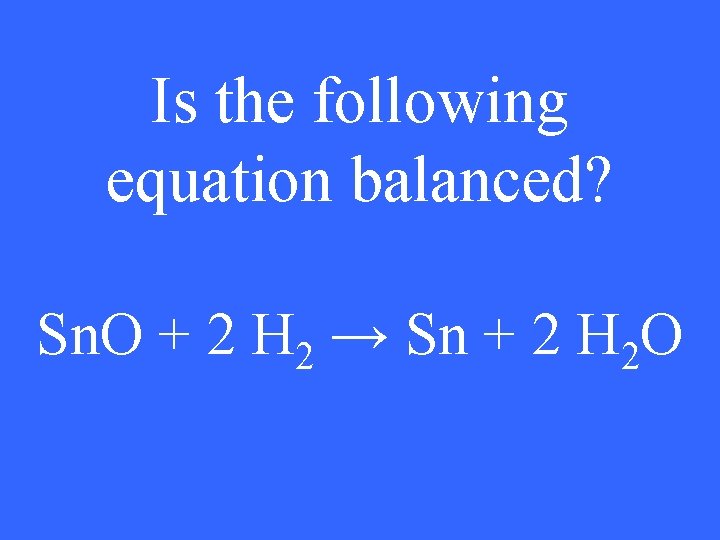
Is the following equation balanced? Sn. O + 2 H 2 → Sn + 2 H 2 O

No 1 pt
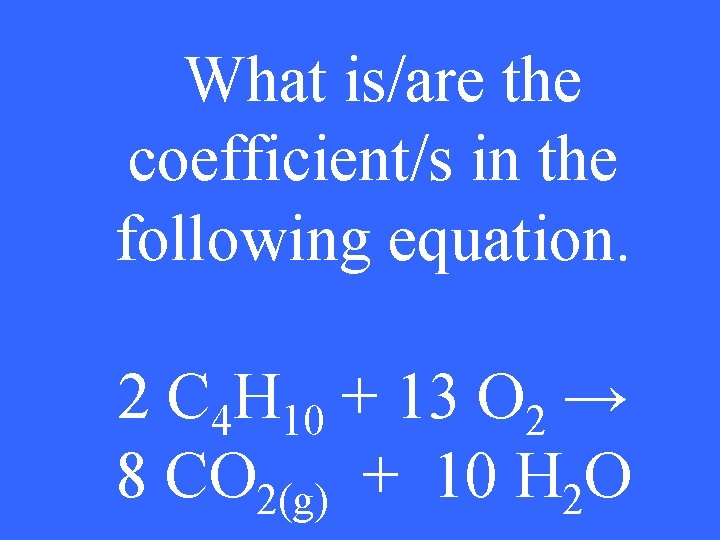
What is/are the coefficient/s in the following equation. 2 C 4 H 10 + 13 O 2 → 8 CO 2(g) + 10 H 2 O

2, 13, 8, 10 2 pt
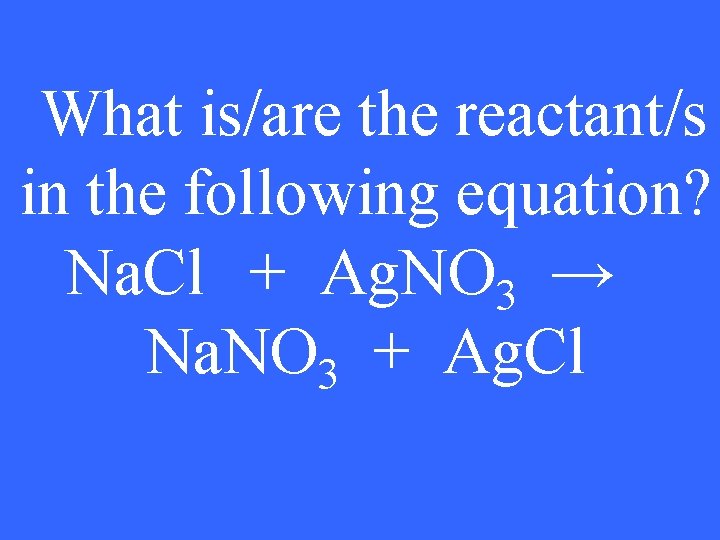
What is/are the reactant/s in the following equation? Na. Cl + Ag. NO 3 → Na. NO 3 + Ag. Cl
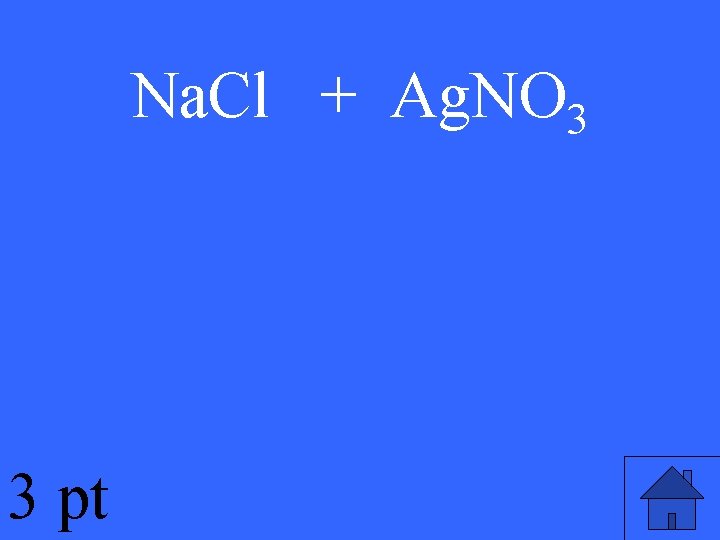
Na. Cl + Ag. NO 3 3 pt
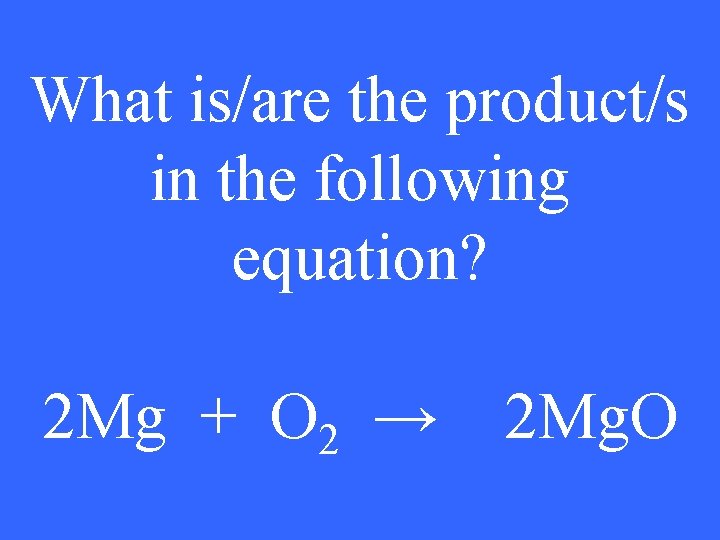
What is/are the product/s in the following equation? 2 Mg + O 2 → 2 Mg. O

2 Mg. O 4 pt

The Law of Conservation of Matter is reflected in balanced equations as the law states this.

Matter cannot be created or destroyed. 5 pt
- Slides: 51