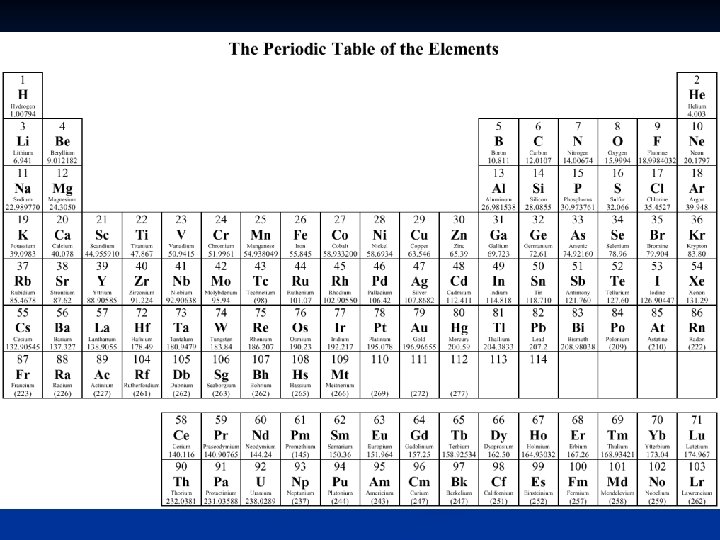
element chemical symbol atomic nucleus protons atomic number

element chemical symbol atomic nucleus protons atomic number neutrons electrons isotopes mass number atomic mass period group or family periodic trend Define these common chemistry vocabulary terms

Element Any material that is made up of only one type of atom Use chemical symbol to abbreviate 1 st letter uppercase 2 nd letter lowercase

Atomic Nucleus Contains protons and neutrons Contains most of atoms mass Protons n n n Determines the element Positively charged Mass = 1 amu (atomic mass unit) 6 x 1023 amu = 1 gram Atomic number tells the number of protons in the atom Neutrons n n Neutral charge Mass about the same as proton – 1 amu

Electrons Negatively charged n Located outside nucleus in electron cloud n Mass = 1/1836 amu n

Ions Charged atoms n In neutral atoms, number of protons and electrons are equal so they have a 0 net charge n In a 2+ ion, the atom has two less electrons than protons to give a positive 2 net charge n
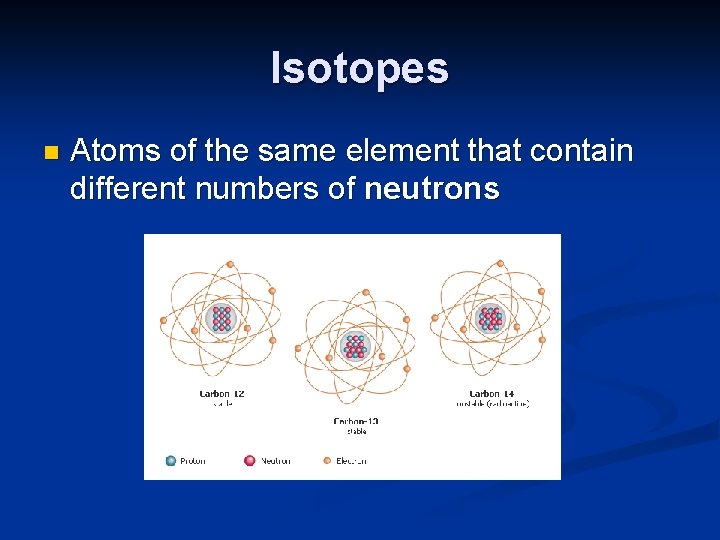
Isotopes n Atoms of the same element that contain different numbers of neutrons

Mass number n Total number of protons and neutrons in an atom Atomic mass Average mass of all isotopes of that element n Found on periodic table n # neutrons = Mass number – atomic number
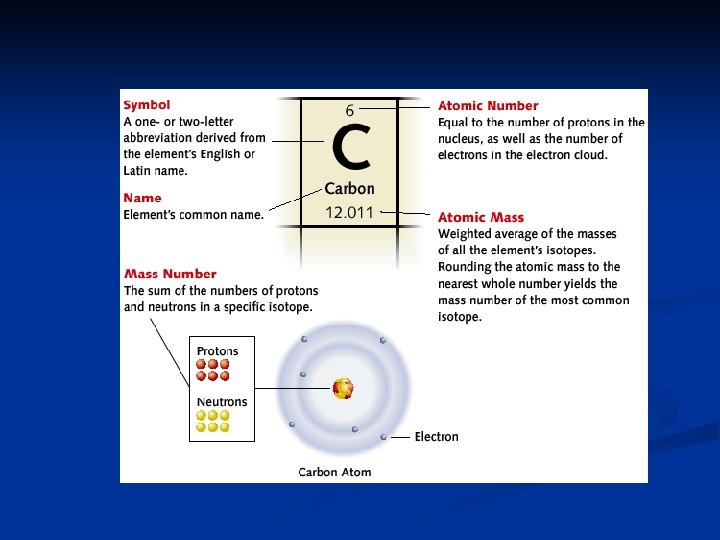

n Row on the periodic table is called n n Period Column on the periodic table is called n Group or family
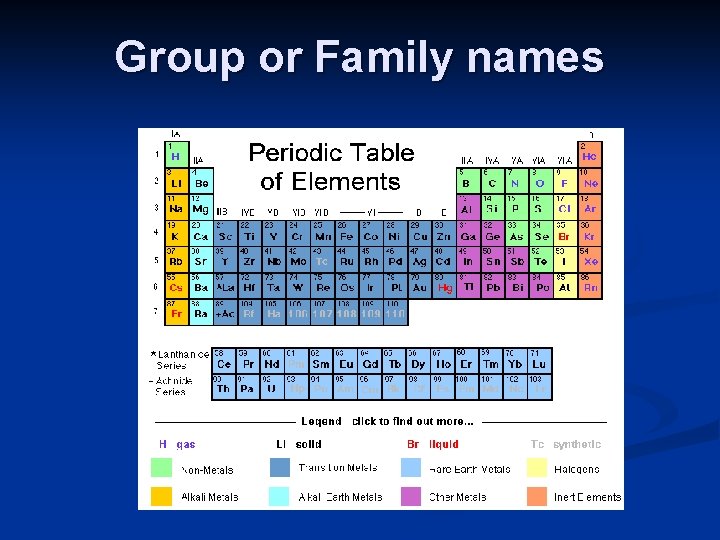
Group or Family names

Diagram of atom Atoms are mostly empty space n Nucleus is 1/10000 of the size of the whole atom n
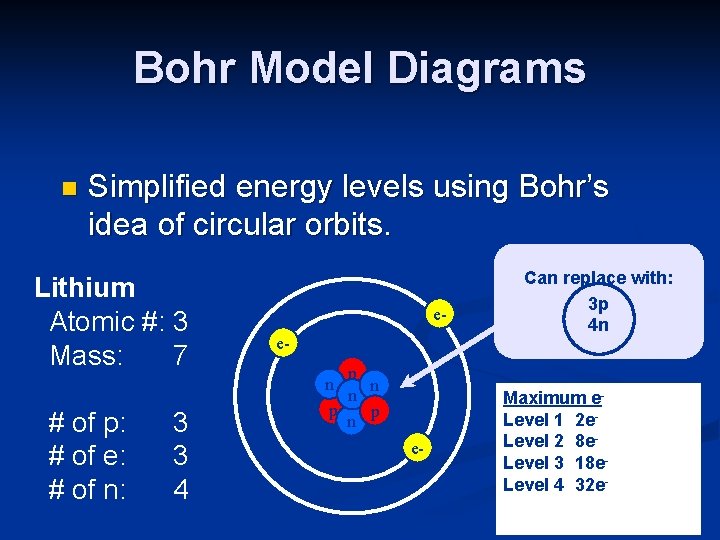
Bohr Model Diagrams n Simplified energy levels using Bohr’s idea of circular orbits. Lithium Atomic #: 3 Mass: 7 # of p: # of e: # of n: 3 3 4 eep n n n p p n e- Can replace with: 3 p 4 n Maximum e. Level 1 2 e. Level 2 8 e. Level 3 18 e. Level 4 32 e-

Bohr Models

Periodic Trend Things that occur in a repeating pattern on the periodic table n Atomic number, # of energy levels, valence electrons, oxidation #, atomic size, metallic properties n

Physical properties of metals Malleable – can be hammered into a thin sheet n Ductile – can be pulled into thin wire n High density n Solid at room temperature n Good conductors of heat and electricity n Luster - shiny n

Physical properties of nonmetals Brittle n Low density n Low melting point n Solid, liquid, or gas at room temperature n Poor conductors of heat and electricity n dull n

n Oops! Those "harmless" germanium capsules you just swallowed may have an extra proton in each nucleus. Is this good news or bad news? Why?
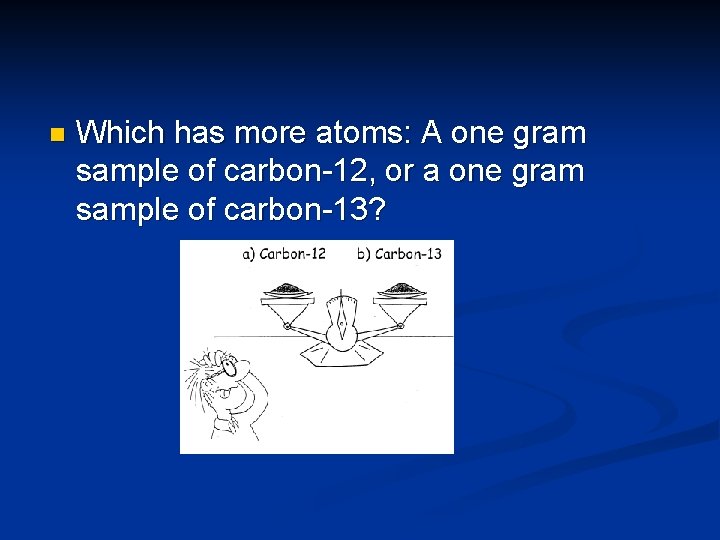
n Which has more atoms: A one gram sample of carbon-12, or a one gram sample of carbon-13?

n Suppose you could add or subtract protons from oxygen nuclei. To turn oxygen into a gas that would glow red when an electric current flows through it, would you add or subtract protons? How many?

n Compared to hydrogen, the element helium has a) more mass and is larger in size b) more mass and is about the same in size c) more mass and is smaller in size d) none of the above
- Slides: 22