Electrostatics Notes Charge The three fundamental facts about

Electrostatics Notes Charge!

The three fundamental facts about atoms: Every atom is composed of a positively charged nucleus, surrounded by negatively charged electrons. n The electrons of all atoms are identical. Each has the same quantity of charge and the same mass. n Protons and neutrons compose the nucleus. Protons are about 1800 times more massive than electrons but carry an amount of charge equal to the negative charge of electrons. Neutrons have slightly more mass than the protons and have no charge. n

The “normal” configuration of the atom. n Most atoms are neutral. n Thus most atoms have an equal number of protons and electrons. n Remember that the number of protons determines the element.

What is an ion? n When an atom gains an extra electron, – it is negatively charged. It is then a negative ion. n When an atom loses an electron, – it is positively charged. It is then a positive ion.

An Atomic Model Objects are made up of atoms n Atoms have positively charged, massive protons and negatively charged, light electrons n The electrons can move more easily than the protons n n We model objects as sets of positive charges fixed in place and negative charges that can shift in response to other charges – Analogy: Desks as protons, students as electrons

What does it mean to say that an object is neutral? n. A neutral object has no net charge. n A neutral object has equal amounts of positive charge and negative charge.

What does it mean to say that an object is charged? n. A charged object has a net charge. n A positively charged object has a greater quantity of positive charge than negative charge. n A negatively charged object has a greater quantity of negative charge than positive charge.

Electrons move, Protons don’t! n Protons are very massive. They have too much inertia. They are in the center of the atom. n Electrons are outside the nucleus. It is easier to move particles that are on the perimeter. n Generally, the electrons in the outermost orbits are the ones that transfer and move first.


What do you have to do to make an object positively charged? n You need to take electrons away from the object.

What do you have to do to make an object negatively charged? n You need to transfer electrons to the object.

What does it mean for an object to be polarized? n When an object is polarized, its charges have shifted so that one side of the object has a net negative charge and the other side of the object has a net positive charge. n A polarized object might be neutral (no net charge) or have a net charge.

Can charge be created or destroyed? n No! Charge can not be created or destroyed. n Conservation of Charge.

Conductors vs. Insulators Loosely bound electrons. n Allow the flow of electric charge. n Examples include metals. n Tightly bound electrons. n Slow the flow of electric charge. n Examples include rubber, plastic and Styrofoam. n

Coulomb’s Law n Two charged objects exert a force on each other. n The magnitude of the force is directly proportional to the product of the quantities of charge. n The magnitude of the force is inversely proportional to the distance between the objects squared.

More Coulomb’s Law (We’ll come back to this later…)

Charged objects exert forces on each other. n Like charges repel. – Two positively charged objects repel each other. • Two protons repel each other. – Two negatively charged objects repel each other. • Two electrons repel each other. n Opposite charges attract. – A positively charged object and a negatively charged object attract each other. • A proton and an electron are attracted to each other. n Newton’s Third Law: Equal and Opposite Forces.

Interactions with Neutral Objects n Remember that a neutral object is comprised of innumerable positively and negatively charged particles. n A charged object (positive or negative) will be attracted to a neutral object. – We’ll discuss exactly why later…

Consider two identical charged particles near each other as shown. The force exerted by q 1 on q 2 points A. Left B. Right C. Up D. Down E. Nowhere; there is no force.
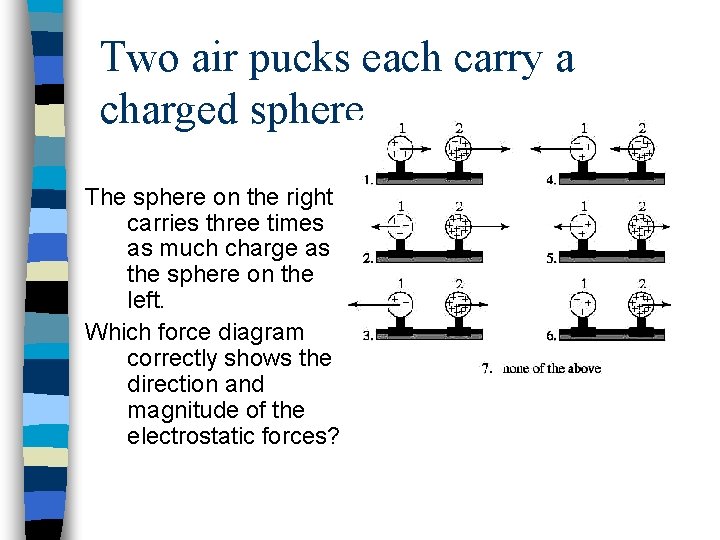
Two air pucks each carry a charged sphere. The sphere on the right carries three times as much charge as the sphere on the left. Which force diagram correctly shows the direction and magnitude of the electrostatic forces?

What if one object is neutral? Note that a neutral object and a charged object will attract each other. A charged object will polarize a neutral (or weakly charged) object. Animation of Polarization Illustrations of Polarization

Three pith balls hang from threads… Wait a minute! What’s a “pith ball”? A pith ball is a small, light sphere coated with conductive material. The coating allows it to be easily charged or grounded, and its small mass allows it to respond easily to small forces. May we go on?

Three pith balls each hang from an insulating thread. PB 1 and PB 2 repel each other. n PB 2 and PB 3 repel each other. Which of these statements is true about the charges on the PBs? n A. B. C. D. E. 1 and 3 have charges of opposite sign. 1 and 3 have charges of the same sign, 2 has opposite charge. All three have charges of the same sign. One of the objects carries no charge. We need more data to determine the signs of the charges.

And now for a similar, yet different, problem…

Three pith balls each hang from an insulating thread. PB 1 and PB 2 attract each other. n PB 2 and PB 3 repel each other. Which of these statements is true about the charges on the PBs? n A. B. C. D. E. 1 and 3 have charges of opposite sign. 1 and 3 have charges of the same sign. All three have charges of the same sign. One of the objects carries no charge. We need more data to determine the signs of the charges.

Now to change the subject… You’ve been looking at the forces exerted on (or by) charged objects. Now, consider how objects become charged, or lose their charge.

Three Charging Methods n Triboelectricity n Charging by Conduction n Charging by Induction n Animations of Charging and Grounding

Conduction Example: Touching charged styrofoam to electroscope Works best from conductor to conductor n Requires the objects be in contact (or close enough for a spark) n The total charge remains constant n When two identical conductors come in contact, they share the total charge equally n

Induction Example: Holding styrofoam close to electroscope, briefly touching ‘scope with finger, then removing styrofoam A charged object charges a neutral conductor without contact n The conductor does make contact with a neutral object n The conductor ends up with a charge opposite that of the charged object brought near n

Triboelectric Examples: Tape; rubbing wool and styrofoam Two dissimilar materials brought into contact and separated will separate their charge n The contact can involve friction or bonding n Works best with insulators n

More on Triboelectricity n Charge separation when two insulators are rubbed together. n One of the insulators is more likely to grab electrons and one of the insulators is more likely to donate electrons. n CHARGE IS NOT CREATED, electrons are transferred.


Charging by Conduction Suppose you have two identical metal spheres, imaginatively named A and B. Each sphere is on an insulating stand. Originally, A has 10 units of charge, and B is neutral. If the two spheres are briefly touched together, then separated, what is the charge on each? A. A has 10 units, B has zero. B. B has 10 units, A has zero. C. A has 10 units, and B also has 10 units. D. A and B each have 5 units. E. A and B each have zero charge.
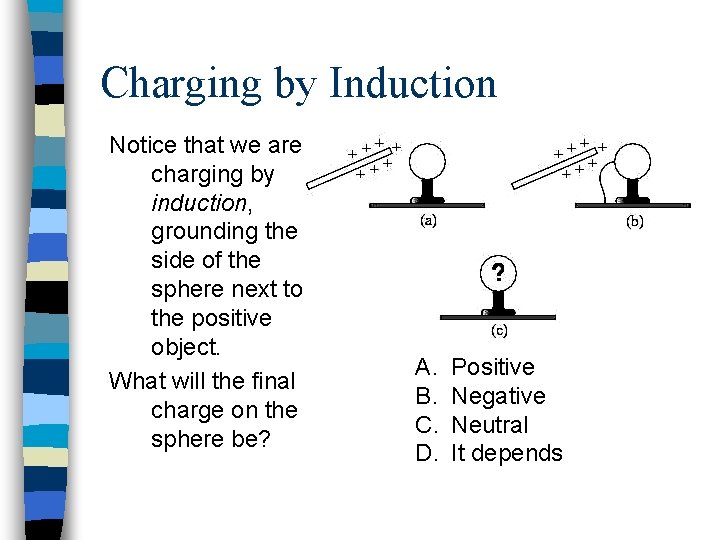
Charging by Induction Notice that we are charging by induction, grounding the side of the sphere next to the positive object. What will the final charge on the sphere be? A. B. C. D. Positive Negative Neutral It depends

Triboelectric Charging You rub a balloon against your hair, and the hair becomes positively charged. This means that A. Electrons moved from the balloon to your hair. B. Protons moved from the balloon to your hair. C. Protons moved from your hair to the balloon. D. Electrons moved from your hair to the balloon. E. The rubbing destroyed electrons in your hair, leaving it positive.

Triboelectric Series A Triboelectric Sequence ELECTRON GRABBERS Rubber Amber Cotton ELECTRON DONORS Silk Cat fur Wool Glass Rabbit fur If you rub cotton with amber, which becomes positive? A. Amber B. Neither C. Cotton Which of the following can make glass negative? A. Amber B. Cat fur C. Rabbit fur

Neutralizing/Grounding Objects When a charged object comes in contact with a very large, neutral conductor, the object becomes neutralized. Earth itself is a large, neutral conductor, so it neutralizes charged objects quite well.

Review Charged objects exert forces: Like repels like Opposites attract Charge and neutral attract How objects get charged or neutralized Conduction Induction Triboelectric Grounding

The End Hooray!

- Slides: 40