Electrostatics Electrostatics is electricity at rest on the















- Slides: 16

Electrostatics

• Electrostatics is electricity at rest on the surface of an object. • Charges move due to forces between objects and the behavior of the charged materials • Charges can be attractive or repulsive.

Chemistry Flashback… • A positively charged nucleus (protons and neutrons) is surrounded by negatively charged electrons • All electrons are identical in mass and charge • An atom usually has the same number of protons as electrons

Basic laws of attraction will apply!! • Likes repel • Opposites attract

Charging an object means you are adding or removing electrons …basically creating an ion However, all the electrons are accounted for creating “conservation of charge”

Example 1: Piece of silk + plastic rod → silk with extra electrons (- charge) + plastic rod with a lack of electrons (+charge) (with induction)

• By rubbing, electrons are exchanged • Electrons will “equalize” by a slow discharge (balloon sticking to a hair then falling off over time) OR quickly by a shock (rubbing socked feet on carpet then reaching for the light switch)

• Electrons can be controlled by using conductors to direct the direction of flow – Semiconductors or semimetals carry small currents – Superconductors carry large currents • Electrons can also be contained by insulators

Coulomb’s Laws of Attraction

• Coulomb’s Law is similar to Newton’s Laws of Gravity in that it compares 2 objects, the distance between them , the constant and the forces of attraction or repulsion
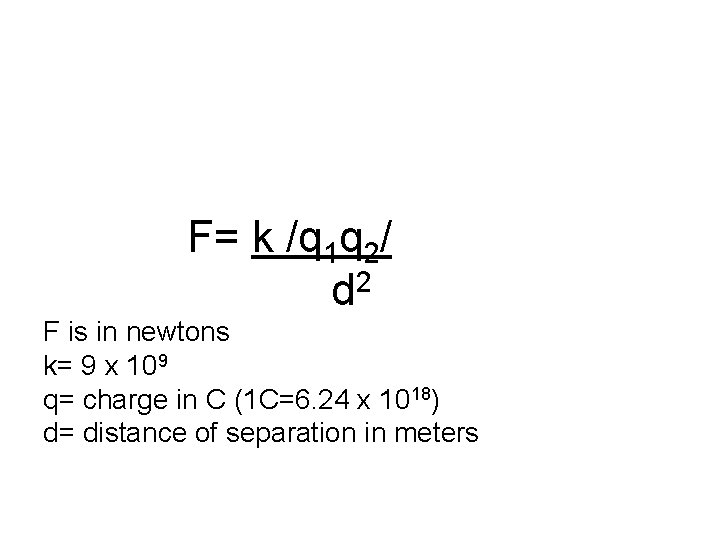
F= k /q 1 q 2/ 2 d F is in newtons k= 9 x 109 q= charge in C (1 C=6. 24 x 1018) d= distance of separation in meters

• Notice that the k constant in this formula shows that electrical forces are ENORMOUS compared to gravitation constants we have used before.

Example 1: • Hydrogen is composed of 1 proton, 1 neutron and 1 electron. If the average separation between charged particles is 5. 3 x 10 -11 m, what is the electrical force of attraction between them? (electrons have a -1. 6 x 10 -19 C charge and protons are +1. 6 x 10 -19 C charge)

• Many physicists have identified the similarities between Fe and Fg that Einstein spent years of his life searching for a “unified field theory”…linking gravity and electricity as the same principle.

Example 2: • Speaking of hydrogen again, the mass of a proton is 1. 7 x 10 -27 kg and the mass of an electron is 9. 1 x 10 -31 kg, calculate the Fg in a hydrogen atom. (use distance from example 1) F= g /me mp/ 2 d

Example 3: • Using your answers for examples 1 and 2, compare the forces of gravity and electricity in a hydrogen atom.