Electrostatics Electricity Comes from Greek word elektron which












- Slides: 13

Electrostatics

Electricity Comes from Greek word elektron which means “amber” because it was noticed that when amber was rubbed with cloth it attracts dust and leaves This attraction is now called Static Electricity An object becomes charged due to rubbing and it posses a net electric charge Benjamin Franklin noticed two types of charge and called them positive and negative

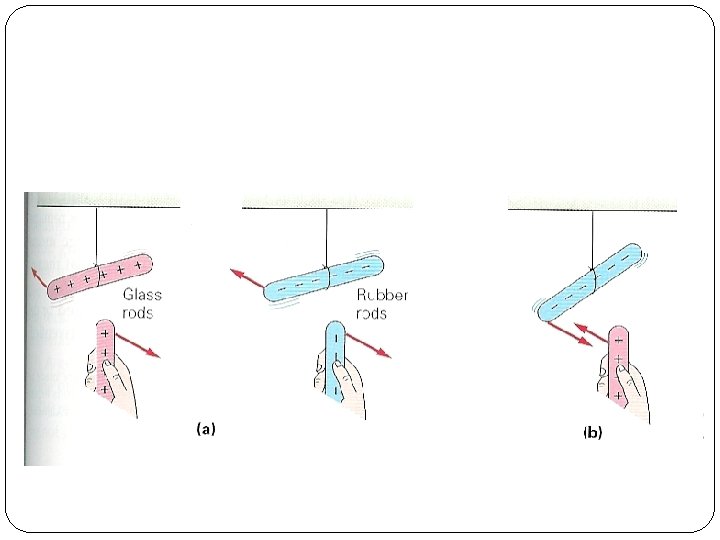
Fundamental Law of Charges Opposites attract Likes repel Any charged object attracts a neutral object


Law of Conservation of Charge Whenever a certain amount of charge is produced on one body, an equal amount of opposite charge is produced on another body Net amount of electric charge produced in any process is zero

Atoms and Electron Charge Charging of a solid results from the transfer of electrons from one material to another Negative charge results when an object acquires an excess of electrons Positive when has deficit

Electrostatic Series � � � gold sulfur brass copper rubber wax silk lead fur wool glass acetate hold e- tight lose e- easily

Examples When rub plastic or rubber object with fur electrons are transferred from the fur to the plastic: plastic becomes - and fur + What happens when glass is rubbed with silk?


Charged By Rubbing Objects charged by rubbing usually lose their charge quickly Charge may be neutralized by ions in the air Charge may “leak” off onto polar molecules like water in the air Why is there more static electricity on dry days?

Insulators and Conductors Solids fall into two main categories as far as electric properties Conductor – allows charge to flow electrons not tightly held so can move freely through the material Insulator – charge cannot flow electrons tightly bound and cannot move

Electric Fields Near Conductors • Because electrons have like charges, they repel each other. In a conductor they are free to move, so they spread far apart in a way that minimizes their potential energy. • The charges come to rest on the surface of the conductor. It does not matter if the conducting sphere is solid or hollow.

Electric Fields Near Conductors • The electric field is zero everywhere inside a closed, charged metal container. • The electric field at the surface depends on the shape of the conductor; free charges are closer together at the sharp points of a conductor.