ELECTROPLATING Electroplating It is the process by which

ELECTROPLATING Electroplating: It is the process by which the coating metal is deposited on the base metal by passing a direct current through an electrolytic solution containing the soluble salt of the coating metal.

Objectives of Electroplating � � � To increase the corrosion resistance To increase resistance to chemical attack and wear resistance To improve physical appearance and hardness To improve surface properties To increase decorative and commercial values of metal.

Electrodes and Reactions Anode: Coating Metal(For ex. Cu)/Graphite � Cathode: Base Metal(For ex. Cup) � Electrolyte Used: Cu. SO 4 solution. � Reaction at Cathode: Cu+2 + 2 e- –-----> Cu Reaction at anode: Cu + SO 4 -2 –----> Cu. SO 4 + 2 e�

Procedure: � � � The article to be electroplated is first treated with organic solvent like tetra chloro ethylene to remove oil, grease etc. then it is treated with dil Hcl or H 2 SO 4 to remove surface scales, oxides etc. The cleaned article is then made as cathode of an electrolytic cell. The anode is either the coating metal itself or an inert material of good electrical conductivity like graphite.

� � The electrolyte is a solution of a soluble salt of the coating metal. When direct current is passed coating metal ions migrate to the cathode and get deposited there. Thus a thin layer of coating metal is obtained on the article made as cathode. For brighter and smooth deposits, favourable conditions such as low temperature, medium current density and low metal ion concentrations are used.
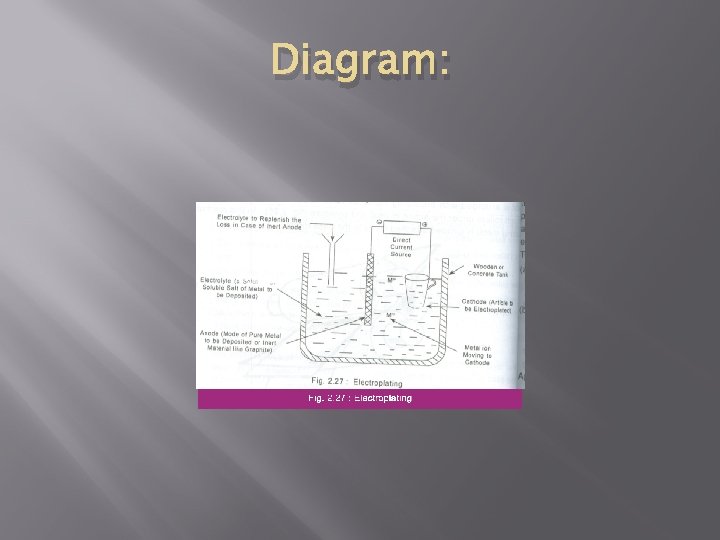
Diagram:

Factors Affecting Electroplating: � � � � � p. H Temperature Current density p. H: For good electrodeposit, the p. H of the bath must be properly maintained. For most plating baths, p. H ranges from 4 -8. The desired p. H of the plating bath is usually maintained by using buffers. p. H lower than the optimum value means stronger hydrogen evolution at cathode occurs, thereby resulting in a burnt deposit. p. H higher than the optimum value means precipitation of hydroxides of the metal on electrode surface. In a neutral bath, if the current efficiency is greater at the anode than at the cathode, the bath becomes more alkaline. If the electrode efficiencies are similar, the p. H of the bath remains unchanged. Hence change in p. H of a plating bath is a good indication of electrode efficiencies.

� � Temperature: In general, the electroplating bath solutions should be used at room temperatures. Warm baths are also used which cause an increase in bath temperature causes an increase in the crystal size. Increase in bath temperature increases solubility and thereby the transport number, which in turn leads to increased conductivity of the solution. It also decreases the viscosity of the solution, thereby replenishing the double layer relatively faster. At higher bath temperature hydrogen evolution at cathode takes place thereby leading to corrosion of equipment and decomposition of organic additives.

� � � Current Density: In the electro deposition process the uniform coating of specimen is depend upon the current distribution parameter. It is usually expressed in Am-2 of the electrode surface. At low current density, the surface diffusion process is faster than electron transfer. Consequently, adatoms can find the most favourable points thereby resulting in uniform deposit. On increasing the current density the process of surface diffusion retards in comparison to electron transfer. Consequently, the adatoms my not be in a position to reach the favourable points. Higher voltage is required. The no. of nuclei formed are likely to increase thereby leading to a less ordered deposit with micro scale features.

� To summarize, the optimum current density range for given plating bath is depending on composition of salt, operating conditions and the type of the plating sought. Anode current densities are also an important plating parameter and should be controlled properly.

Applications Used for Industrial applications Superior conductivity of metals Decorative purposes
- Slides: 11