Electrons WarmUp n On blank sheet of paper

Electrons

Warm-Up n On blank sheet of paper, write the following question and answer it: n I want you to reflect on how this class has gone for you over the past 2 six weeks. What have you done well, and what areas where you need to improve? n If you take this home and bring it back signed by your parent, I will give you a 100 Lab Grade!

Essential Question? ? ? n What are the properties of electrons as both waves and particles?

Electrons Tiny, very small mass, move in ecloud in energy levels Electrons have “split personality” (dual nature) • Behave like particles • Behave like light energy
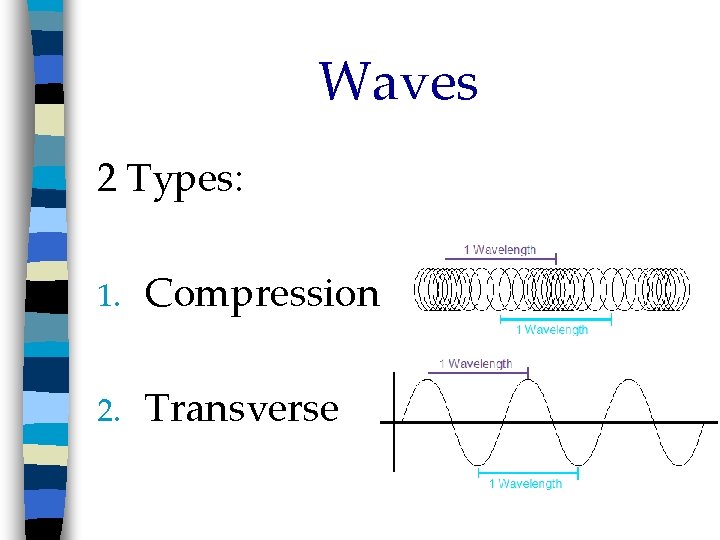
Waves 2 Types: 1. Compression 2. Transverse

Waves Compression (longitudinal) n Direction of wave movement is parallel to axis n Sound and some earthquake waves n Increase amplitude by increasing # of particles compressed

Waves Transverse (S waves) n Direction of wave movement is perpendicular to axis n Light, radio, x-rays n Wave = Crest + Trough

Waves Wavelength ( λ = lambda); complete crest and trough n High energy = short λ n Low energy = long λ

Waves Amplitude – distance from top of crest to axis n High n Low amplitude = bright amplitude = dull

Questions? n Notice, there are 2 pictures on the left side of the page. n Take 2 minutes and write a question that relates to figures you see.

Waves Frequency (f); # of λ passing point in a given time n High energy = high f n Low energy = low f freq. = __λ___ time Units: 1__ = Hz (Hertz) sec
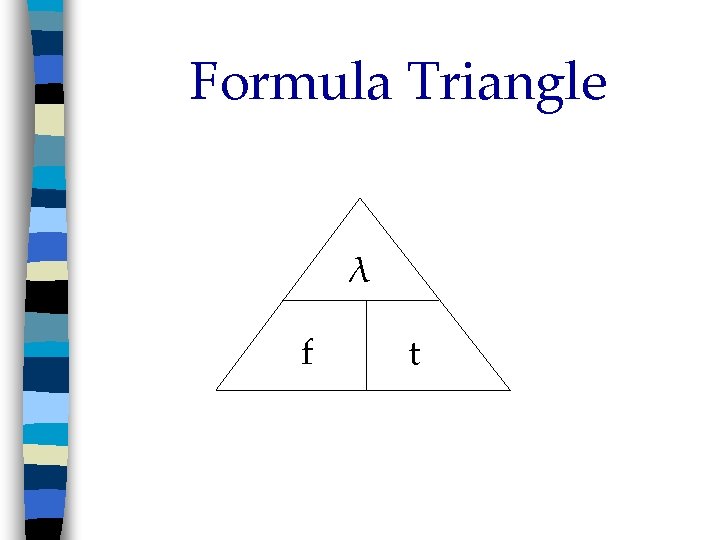
Formula Triangle λ f t

Electromagnetic Spectrum n Electromagnetic radiation is a form of energy that exhibits wavelike behavior as it travels through space n Together, all the forms of electromagnetic radiation form the electromagnetic spectrum
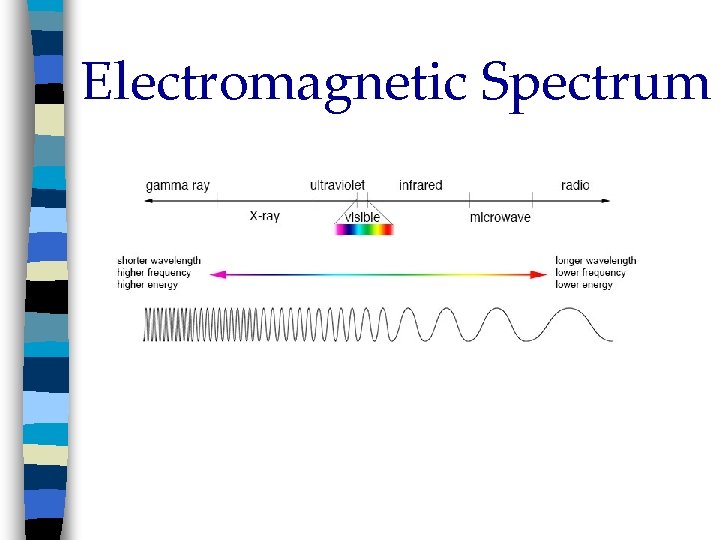
Electromagnetic Spectrum

Wrap-Up n Write 2 questions addressing the material we just covered on the back of the page. n Write a summary. Make sure you answer the Essential Question (What are the properties of electrons as both waves and particles? ).

Poster n In groups (or individually) create a poster that shows your assigned area of the electromagnetic spectrum n Follow the directions on the page given to you n This will be counted as LAB GRADE! n You will have 30 min today and time next class to finish

Practice n Do all questions 1 -10 (we will do 11 -15 later) n Use your notes and ask your table for help, if needed n Make sure you have gotten all of your make-up work and test corrections turned in!

Warm-Up n Pick up half-sheet of paper and answer the questions n Turn time. in the “Reflection” you did from last

Essential Question? ? n What is quantum mechanics and what are the calculations that can be performed?

Quantum Mechanics Study of the placement, movement, and number of e- in atoms

Quantum Mechanics Electrons & Atomic Emission Spectra 1. e- occupy different energy levels within e- cloud 2. e- in lowest energy level = ground state = no extra energy

Quantum Mechanics 3. Atoms absorb energy from heat/ electricity (absorbed energy = quantum energy) n e- get excited and “jump” to higher energy level (quantum leap)

Quantum Mechanics 4. Eventually lose extra energy and fall back to ground state while releasing extra energy n Only e- in positive ions will make quantum leaps 5. Released energy = photons = packets of emitted energy

Quantum Mechanics 6. When photon’s energy = energy of visible light: n Pass photons through prism to make atomic emission spectra

Atomic Emission Spectra n Every element has the ability to give off visible light when heated or when electricity passes through gas state n Why? Because of excited e-

Atomic Emission Spectra n Pass emitted light through prism = bands of color n Pattern of bands unique to each element n How we study stars and the universe!

In order to calculate the wavelength or frequency of an electromagnetic wave, use this formula: Speed of light = wavelength x frequency C= λ x f C = 3. 00 x 108 m sec C = 3. 00 x 1010 cm sec C = 3. 00 x 1017 nm sec


What is the wavelength of yellow light with a frequency of 5. 09 X 1014 Hz? What is the frequency of violet light with a wavelength of 408 nm?

Planck calculated the energy of a photon with this formula: Energy = Planck’s constant x frequency E =h x f h = Planck’s constant = 6. 63 x 10 -34 J·s

What is the energy of a photon that has the same frequency as radio waves, 1. 25 x 106 Hz ? What is the wavelength of yellow light with an energy of 4. 0 x 1011 J?

Notes Wrap-Up n Write 3 questions that cover the notes for today. n Write a summary for today’s notes on quantum theory and atomic spectra. Make sure it addresses the essential question.

After Project n Finish the “Waves Worksheet” (Problems 1 -15)

Warm-Up n Get out the page from last class (there are some at the door if you have lost yours) n Read the top section and highlight or circle the equations. Copy the formula triangles from board. n Today, we are working problems n Test review n Test next class! Thursday!!!

Warm-Up

Warm-Up 1. A 2. B 3. C 4. B 5. C

Warm-Up 6. A 7. D 8. C 9. B 10. A

Essential Question? ? ? n How do we classify elements based on their position in the periodic table and their electron configuration?

Electrons n Principle Quantum Numbers (n) = ground states of e- n To find # of electrons in each energy level, use: # e- = 2 n 2 n=1=2 en=2=8 en=3=18 e- n=4=32 en=5=50 en=6=72 e-

Electrons n Theoretical n In numbers actuality, energy levels 5 – 7 only hold as many electrons as level 4

Electrons Orbitals – sublevels within each energy level; areas where probability of finding e- is very high 4 Types: s, p, d, f

Orbitals 1. S sphere e- have low energy only 1 “s” per energy level

Orbitals 2. P dumbell e- have more energy than S 3 “p” per energy level px py pz

Orbitals 3. d complex e- have high energy 5 “d” per energy level d 1 d 2 d 3 d 4 d 5

Orbitals 4. f = complex e- have highest energy 7 “f” per energy level f 1 f 2 f 3 f 4 f 5 f 6 f 7

Quantum Mechanics n Pauli Exclusion Principle – only 2 e- in any orbital # orbitals = n 2 n = energy level # n=1=1 orb. n=2=4 orb. n=3=9 orb. 1 s 1 s 3 p 5 d n=4=16 orb. 1 s 3 p 5 d 7 f

Electron Configurations n Mother Nature fills energy levels & orbitals according to amount of energy present; e- go in least energy first (Aufbau Principle) n When electrons occupy orbitals of equal energy (EX: 2 px, 2 py, 2 pz), one e- goes in each orbital before a second one goes into each orbital (Hund’s Rule)

Electron Configurations n Pattern on Periodic Table!!! - each box represents position of one e- order of filling L R across rows, follow increasing atomic #

Electron Configurations Elements with lots of electrons can have repetitive electron configurations so. . . here are some shortcuts to make the job easier: If you have a full s orbital – use s 2 If you have a full p orbital – use p 6 If you have a full d orbital – use d 10 If you have a full f orbital – use f 14


Electron Configurations Example electron configurations: H He C Ne Cu Pb

Electron Configurations Example electron configurations: H (Hydrogen): 1 s 1 He (Helium): 1 s 2 C (Carbon): 1 s 22 p 2 Ne (Neon): 1 s 22 p 6 Cu (Copper): 1 s 22 p 63 s 23 p 64 s 23 d 9 Pb (Lead): 1 s 22 p 63 s 23 p 63 d 104 s 24 p 64 d 105 s 25 p 64 f 145 d 106 s 26 p 2

Notes Wrap-Up n Write 2 questions over the material for today. n Write a summary over that answers the essential question (How do we classify elements based on their position in the periodic table and their electron configuration? ).

Worksheet n Do 1 -22 n We will work 23 & 25 together. Do 24 & 26. n Do 27 -28 n Hold on to this until next class

Exit Ticket n Scan the QR code and answer the questions OR pick up the page at the front with the questions printed out n On paper copy, make sure to add question 6: “What is one thing you are still confused about? ” n Make sure to turn it in before you leave today

Warm-Up n Pick up the page by the door. n There is a front and back, do both sides. Use your periodic table from last class to help you with the electron configurations.

Warm-Up n 1. D n 2. B n 3. A n 4. C n 5. C


Essential Question? ? ? n How do we classify elements based on their position in the periodic table and their electron configuration?

Electron Configurations You may backup to the nearest inert gas, put the symbol in a bracket to represent all the core electrons; then write out only the last row of the electron configuration. For noble gases you must write the entire last row not just the symbol in a bracket. Ex: For Sulfur: [Ne]3 s 23 p 4

Electron Configurations Excited e- configurations - have gaps in the e- sequence because ehas jumped to higher energy position - Example: 1 s 22 p 53 s 1 Impossible e- configurations - have more than the maximum number of electrons in an orbital - Example: 1 s 22 p 73 s 2

Electron Configurations Ions § Ions contain either more or fewer electrons than a neutral atom (has a charge, either + or -) § If positive, back up as many boxes as the charge on the periodic table § If negative, move forward as many boxes as the charge on the periodic table Ex: Li+1: 1 s 2 (not 1 s 22 s 1)

Electron Configurations Write using the bracket shortcut: Ca Ar Pb S-2 K+1
![Electron Configurations Write using the bracket shortcut: Ca: [Ar]4 s 2 Ar: [Ne]3 s Electron Configurations Write using the bracket shortcut: Ca: [Ar]4 s 2 Ar: [Ne]3 s](http://slidetodoc.com/presentation_image_h2/072d164eb124d9d38c62de731f339642/image-64.jpg)
Electron Configurations Write using the bracket shortcut: Ca: [Ar]4 s 2 Ar: [Ne]3 s 23 p 6 Pb: [Xe] 6 s 24 f 145 d 106 p 2 S-2: 2 extra electrons; same as argon; [Ne]3 s 23 p 6 K+1: 1 less electron; same as Argon; [Ne]3 s 23 p 6

Notes Wrap-Up n Write 2 questions over the material for today. n Write a summary over that answers the essential question (How do we classify elements based on their position in the periodic table and their electron configuration? ).

Exit Ticket n Think about what we’ve discussed with electron configurations… (last 2 classes) 1. Write 1 thing you could explain to a friend. 2. Write 1 thing that still confuses you.
- Slides: 66