Electrons in Atoms Unit Bohr Model Lewis Dot

Electrons in Atoms Unit Bohr Model, Lewis Dot for Elements, Valence Electrons
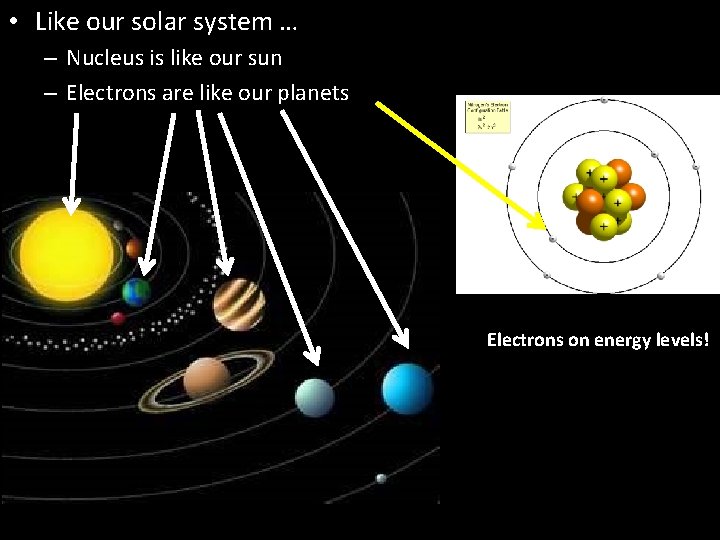
• Like our solar system … – Nucleus is like our sun – Electrons are like our planets Electrons on energy levels!

• Model Only Really works for Hydrogen How do we draw a Bohr Model? 1. ) Draw a nucleus 2. ) Determine how many electrons you need 3. ) Draw energy levels around nucleus Each energy level can only hold so many electrons 1 st = 2 e 2 nd = 8 e 3 rd = 8 e 4 th and up = 2 e 4. ) Place electrons on each energy level until you reach the amount of electrons you need (refer to step #2) 20 Ca 40. 078 How many e- on 1 st = ____ 2 nd = ____ 3 rd = ____ 4 th = ____ Total = ____

1. ) Draw a nucleus 2. ) Determine how many electrons you need 3. ) Draw energy levels around nucleus Each energy level can only hold so many electrons 1 st = 2 e 2 nd = 8 e 3 rd = 8 e 4 th and up = 2 e- 4. ) Place electrons on each energy level until you reach the amount of electrons you need (#2) • Draw the Bohr Model for: Sodium Oxygen

Identify the element from its Bohr Model Proton Neutron Electron How many e- on 1 st = ____ 2 nd = ____ 3 rd = ____ 4 th = ____ Total = ____

Identify the element from its Bohr Model Proton Neutron Electron How many e- on 1 st = ____ 2 nd = ____ 3 rd = ____ 4 th = ____ Total = ____

Valence Electrons • The outermost electrons on the highest occupied energy level of an element

• How do we find the number of valence electrons? – Bohr diagram How many valence electrons are present in the last energy level of Calcium? -- - - ++ ++++ + - + + +++++ + - - +++- - - -- – Periodic table • BEST CHEAT SHEET EVER!
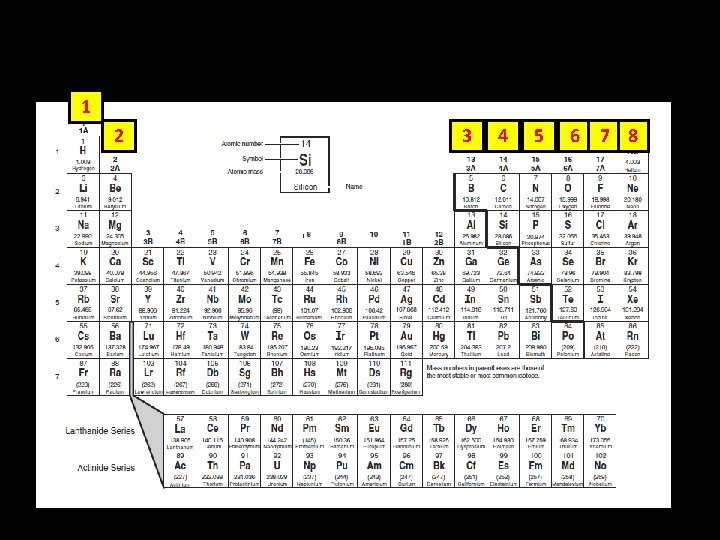
1 2 3 4 5 6 7 8

How many valence electrons are in. . . • • • H _____ N _____ Cl _____ Ba _____ Ne _____ He _____

Lewis Dot Structures – System of arranging dots to represent the valence electrons – G. N. Lewis developed this system – Dots represent the valence electrons • Max of 8 around the symbol • First two together • Rest go “counter-clockwise” • • • • X

Example: • How many valence electrons does phosphorous have? • Write the Lewis dot structure for phosphorous
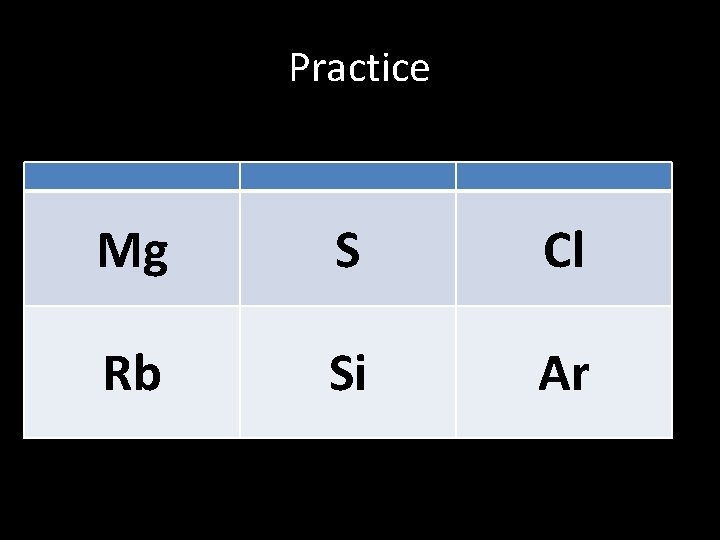
Practice Mg S Cl Rb Si Ar

IONS – charged atom • Elements will gain or lose electrons to achieve an OCTET. – This means they want to have 8 valence electrons – Known as the OCTET RULE

• Cation – – Positive ion – Loses electrons to become more positive • Why? • Easier to lose 1, 2 or 3 e- than it is to gain 5, 6 or 7 e- K becomes K+ B becomes B 3+

• Anion – Negative ion – Gains electrons to become more negative • Why? • Easier to gain 1, 2 or 3 electrons than to lose 5, 6, or 7 e- Cl becomes Cl. O becomes O 2 -


Draw the Lewis Dot Structure for these ions: + Na 2 O 2+ Mg
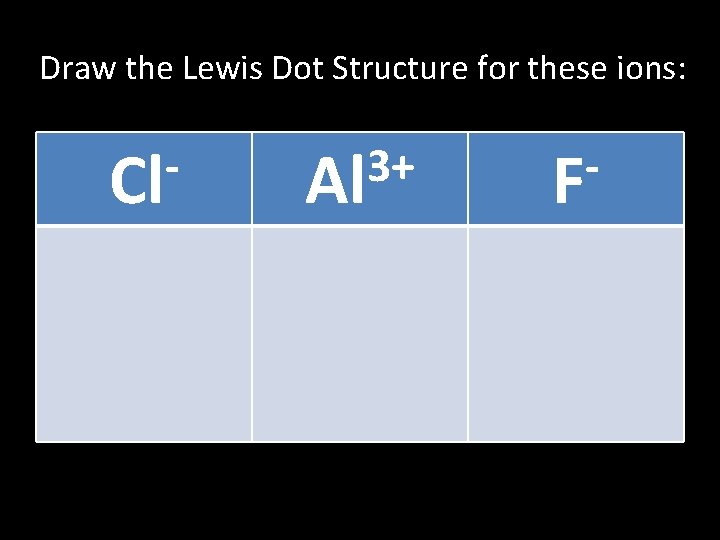
Draw the Lewis Dot Structure for these ions: Cl 3+ Al F
- Slides: 19