Electrons in Atoms Light and the Periodic Table

Electrons in Atoms, Light and the Periodic Table © 2012 Pearson Education, Inc.

9. 2 A packet of light energy or a particle of light is called a(n) a. b. c. d. e. proton. photon. electron. frequency. gamma ray. © 2012 Pearson Education, Inc.

9. 4 a. b. c. d. e. How are frequency and wavelength of light related? The lower the frequency, the smaller the wavelength. The wavelength and frequency are equal. The larger the wavelength, the higher the frequency. The higher the frequency, the smaller the wavelength. None of the above © 2012 Pearson Education, Inc.
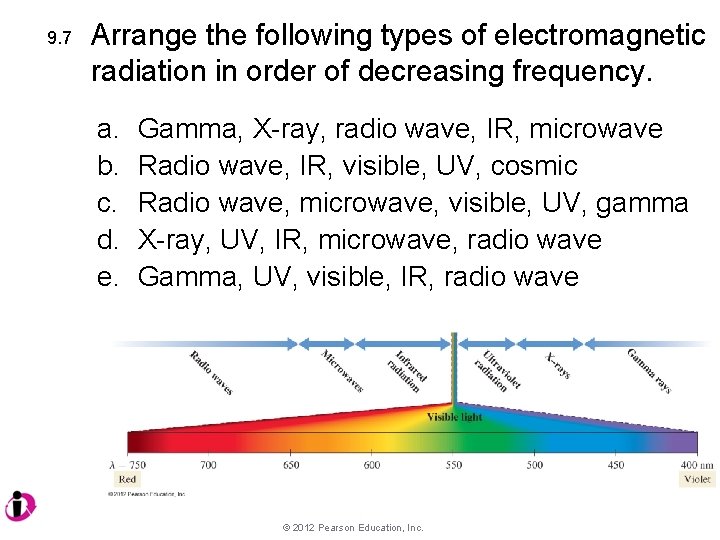
9. 7 Arrange the following types of electromagnetic radiation in order of decreasing frequency. a. b. c. d. e. Gamma, X-ray, radio wave, IR, microwave Radio wave, IR, visible, UV, cosmic Radio wave, microwave, visible, UV, gamma X-ray, UV, IR, microwave, radio wave Gamma, UV, visible, IR, radio wave © 2012 Pearson Education, Inc.
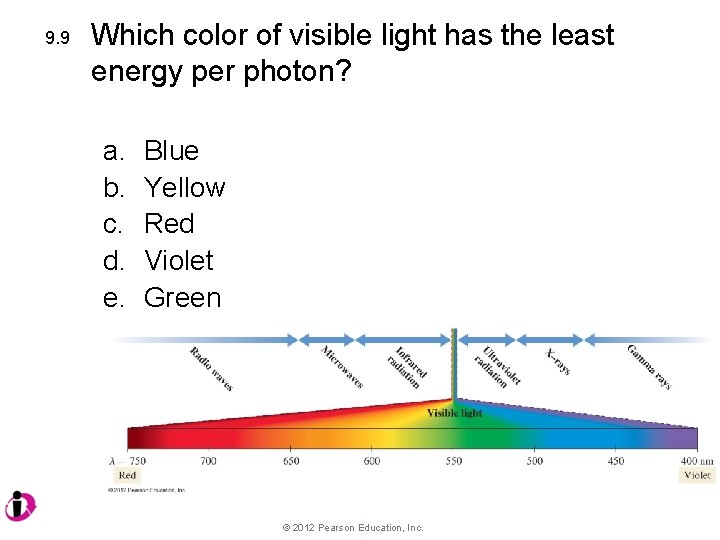
9. 9 Which color of visible light has the least energy per photon? a. b. c. d. e. Blue Yellow Red Violet Green © 2012 Pearson Education, Inc.

9. 10 The Bohr model correctly predicts the emission spectrum of a. b. c. d. e. hydrogen. helium. oxygen. neon. all atoms. © 2012 Pearson Education, Inc.

9. 11 What is the shape of a p orbital? a. b. c. d. e. Spherical Donut Pyramidal Dumbbell Square © 2012 Pearson Education, Inc.

9. 12 How many orbitals are associated with a given set of d orbitals with the same principal quantum number? a. b. c. d. e. 1 7 3 9 5 © 2012 Pearson Education, Inc.

9. 13 What is the maximum number of electrons allowed in each f orbital? a. b. c. d. e. 2 6 8 10 14 © 2012 Pearson Education, Inc.

9. 14 What is the maximum number of electrons in the n = 4 principal energy level? a. b. c. d. e. 6 8 18 32 64 © 2012 Pearson Education, Inc.

9. 15 Which of the following electron configurations is NOT possible? a. b. c. d. e. 1 s 22 p 3 [Ar]4 s 24 d 104 p 4 [Ne]3 s 2 [He]2 s 22 p 63 s 2 [Kr]5 s 24 d 105 p 2 © 2012 Pearson Education, Inc.

9. 16 Which of the following elements has the following electron configuration 1 s 22 p 63 s 23 p 5? a. b. c. d. e. O Cl F Ar S © 2012 Pearson Education, Inc.

9. 18 What is the electron configuration for sulfur? a. b. c. d. e. [Ar]4 s 24 p 5 [Ne]3 s 23 p 4 1 s 22 s 22 p 63 s 23 p 5 Two of the above © 2012 Pearson Education, Inc.

9. 20 What is the electron configuration for the copper atom? a. b. c. d. e. 1 s 22 p 63 s 23 p 64 s 23 d 7 [Ar]4 s 24 d 9 [Ar]4 s 13 d 10 1 s 22 p 2 [Ar]4 s 23 d 9 © 2012 Pearson Education, Inc.

9. 23 Arrange the following elements in order of increasing ionization energy: Si, F, K, Mg, S a. b. c. d. e. K, Mg, S, Si, F Si, S, K, F, Mg F, Si, S, Mg, K K, Mg, Si, S, F F, Mg, Si, K, S © 2012 Pearson Education, Inc.

9. 29 Light is emitted from an atom when a. electrons move farther from the nucleus in energized atoms. b. excited electrons return to lower energy paths around the nucleus becomes excited (or energized). c. d. photons move farther from the nucleus in energized atoms. e. Two of these statements are correct. © 2012 Pearson Education, Inc.
- Slides: 16