Electrons in Atoms Electron Configuration Orbital Notation LewisElectron

Electrons in Atoms Electron Configuration Orbital Notation Lewis-Electron Dot Diagram

Electron Configuration A form of notation which shows how the electrons are distributed among the various atomic orbital and energy levels ◦ Example of e- configuration for Hydrogen: 1 s 1 Principle Quantum number which stands for the energy level Total # of electrons in that sublevel. 1 st column of s-block S-Block
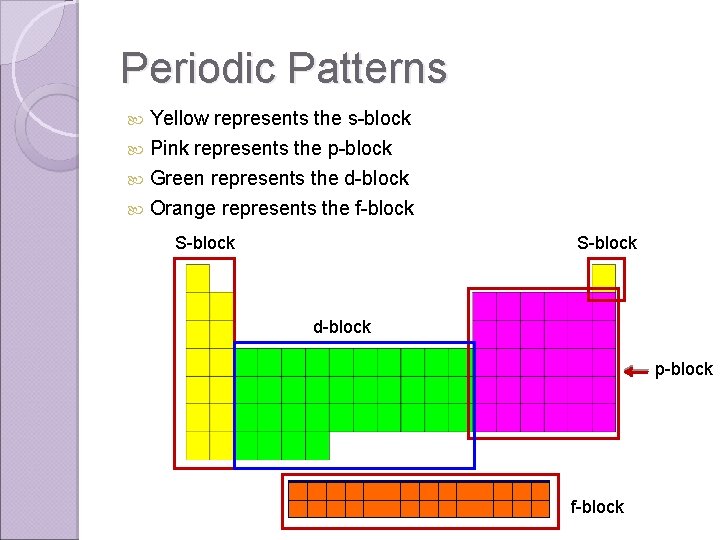
Periodic Patterns Yellow represents the s-block Pink represents the p-block Green represents the d-block Orange represents the f-block S-block d-block p-block f-block

Order of filling sublevels 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 Sublevel (block) # of orbitals Maximum # of e- S 1 2 p 3 6 d 5 10 f 7 14 You must fill in each sublevel with its maximum number of electrons before you move to the next energy level

Examples e- configuration for Oxygen ◦ Oxygen has 8 electrons and is in period 2 ◦ 1 s 2 2 p 4 2 energy level and located in the p-block 6 valence electrons

Practice Write the e- configuration for the following elements: ◦ Helium ◦ Neon ◦ Sulfur ◦ Calcium
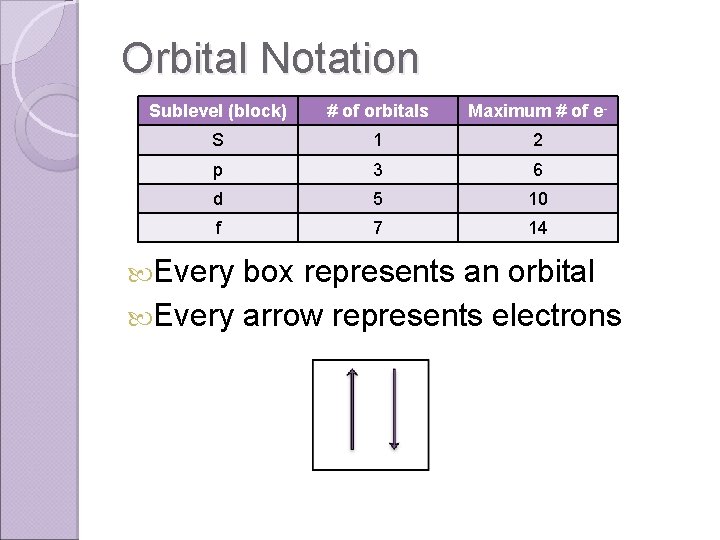
Orbital Notation Sublevel (block) # of orbitals Maximum # of e- S 1 2 p 3 6 d 5 10 f 7 14 Every box represents an orbital Every arrow represents electrons

General Rules Pauli Exclusion principle ◦ Every orbital can only hold 2 electrons with opposite spins Hund’s rule ◦ Within a sublevel, place one e- per orbital before pairing them. WRONG Right

Example: 1 s 2 Orbital 2 s 2 2 p 4 Notation for Oxygen: ◦ You know the e- configuration for oxygen is 1 s 22 p 4 ◦ Remember: Every orbital is represented by a box Every orbital holds a maximum of 2 electrons s-block represented by 1 orbital p-block represented by 3 orbitals

Practice Show orbital notation for the following elements: ◦ ◦ Helium Neon Sulfur Calcium

Lewis Electron Dot Structure An atom’s Lewis electron dot structure consists of : ◦ element’s symbol-which represents the atomic nucleus and inner-level electrons, surrounded by dots representing the atom’s valence electrons. Source: Glencoe Chemistry Matter and Change pg. 140
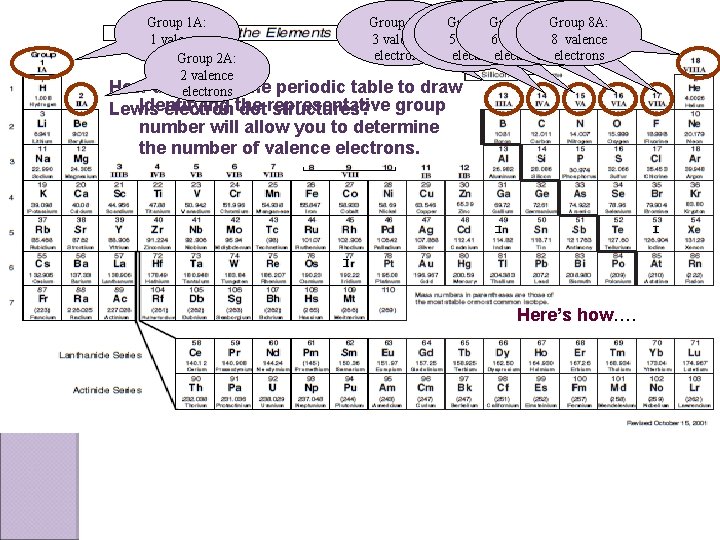
Group 1 A: 1 valence electron Group 2 A: 2 valence How do we use electrons Group 3 A: Group 5 A: Group 6 A: Group 7 A: 8 A: 4 A: 3 valence 4 5 valence 6 valence 7 valence 8 valence electrons electrons the periodic table to draw Identifying Lewis electron the dotrepresentative structures? group number will allow you to determine the number of valence electrons. Here’s how….

Writing out Lewis Electron Dot Structure Group # = number of valence electrons Example: Phosphorus is on group 5 A, so it has 5 valence electrons. Step 3: Continue placing one Step 1: write the element symbol P Step 2: Start at the top of the symbol and place one electron on each of the four sides of the symbol & then pair up the electrons until you have placed all valence electrons around the symbol

Practice Write the Lewis Electron Dot Structure for the following elements: ◦ Helium ◦ Neon ◦ Sulfur ◦ Calcium
Questions? ?
- Slides: 15