Electrons in Atoms and The Periodic Table Chapters

Electrons in Atoms and The Periodic Table Chapters 4 and 5

Unanswered Questions How are the electrons placed around the nucleus? Why are the electrons NOT pulled into the nucleus? Opposites attract To answer these questions, scientists needed to have a new understanding of LIGHT. Starting with Electomagnetic Radiation: A form of energy that moves like waves through space end
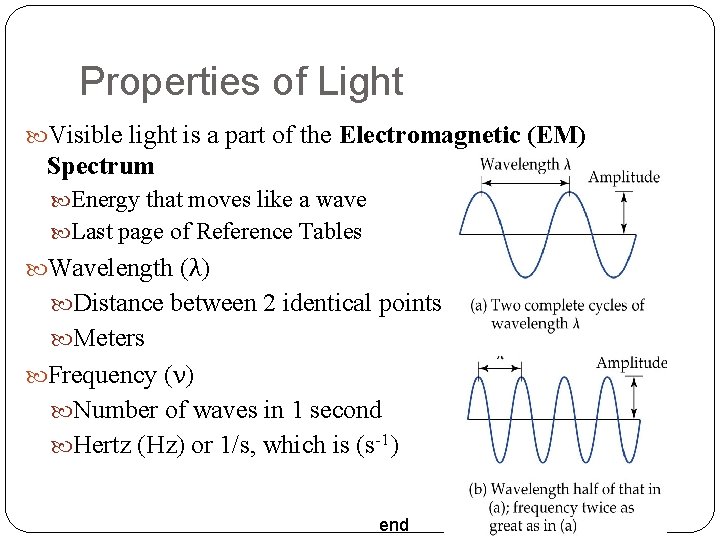
Properties of Light Visible light is a part of the Electromagnetic (EM) Spectrum Energy that moves like a wave Last page of Reference Tables Wavelength (λ) Distance between 2 identical points Meters Frequency (ν) Number of waves in 1 second Hertz (Hz) or 1/s, which is (s-1) end

Properties of Light All EM waves travel at the Speed of Light (c) c = 3. 0 x 108 m/s As frequency goes up: v Wavelength goes down Energy goes up end E λ
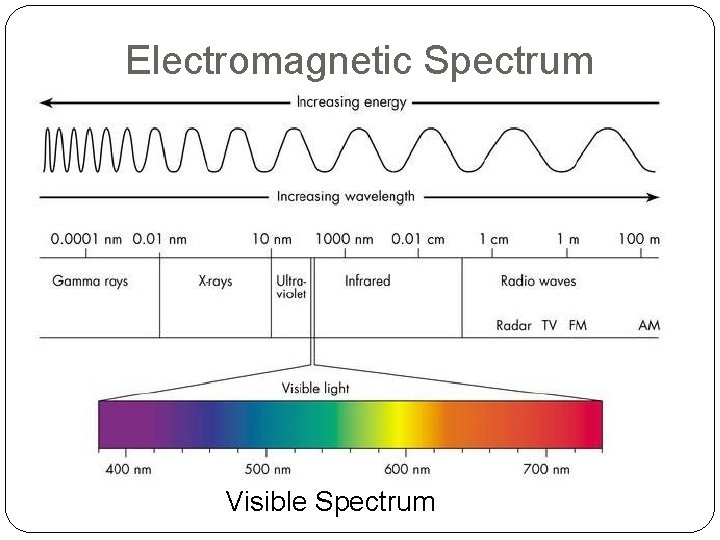
Electromagnetic Spectrum Visible Spectrum

Properties of Light 1. A gamma ray has a wavelength of 7 x 10 -13 m. What is the frequency of this ray? 2. A ray has a wavelength of 5. 5 x 10 -7 m. What is the frequency of this ray? What type of EM wave is this? 3. A ray has a frequency of 7. 65 x 10 -2 Hz. What is the wavelength of this ray? What type of EM wave is this? 4. HONORS: A satellite transmits on the EM Spectrum at a frequency of 9. 4 x 109 Hz. What is the signal’s wavelength? 1. What type of wave is being transmitted? 5. HONORS: A beam of visible light has a frequency of 4. 5 x 1014 Hz. 1. What is the wavelength of this light? 2. What color is this light? end

Properties of Light does not always act like a wave The Photoelectric Effect Metal gives off electrons when light shines on it Depends on the frequency of the light Not the amount of light Max Planck (1900) Matter gains/loses energy in specific amounts known as Quanta Quantum The minimum amount of energy that can be gained/lost Frequency Energy Planck’s Constantend (6. 626 x 10 -34 J·s)

Properties of Light 1. What is the energy of an EM wave with a frequency of 6. 32 x 1026 Hz? 2. What is the energy of an EM wave with a frequency of 4. 56 x 1015 Hz? 3. What is the energy of an EM wave with a frequency of 9. 5 x 1030 Hz? 4. HONORS: What energy would a beam of microwaves have if their wavelength is 5. 6 x 10 -2 m? (2 step problem) 5. HONORS: A beam of visible light has a wavelength of 5. 8 x 10 -7 m. What is its energy and what color is it? (2 step problem) end

Properties of Light Albert Einstein (1905) Wave-Particle Duality All matter and energy can act as both a wave and a particle Photon A particle of light The energy of a photon depends on the frequency end

Line-Emission Ground State: The lowest energy state of an atom Excited State: The highest energy state of an atom How can we see the change between states? You can use a spectroscope When a narrow beam of light is shined through a prism, it is separated into a series of specific wavelengths of visible light.
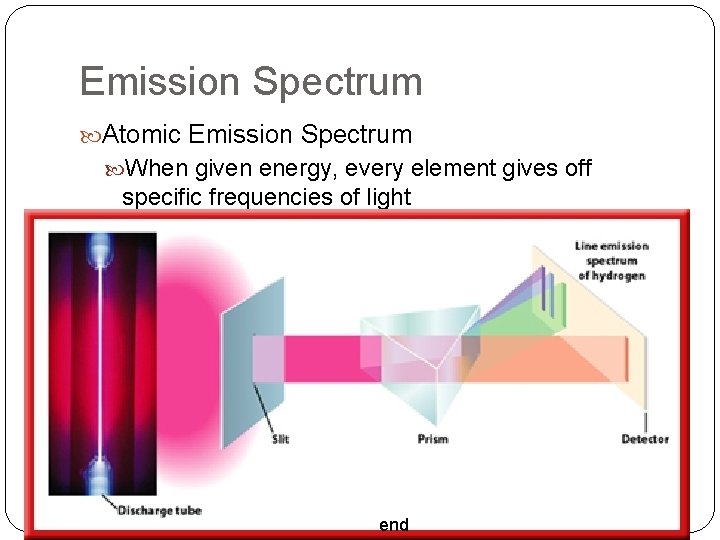
Emission Spectrum Atomic Emission Spectrum When given energy, every element gives off specific frequencies of light end
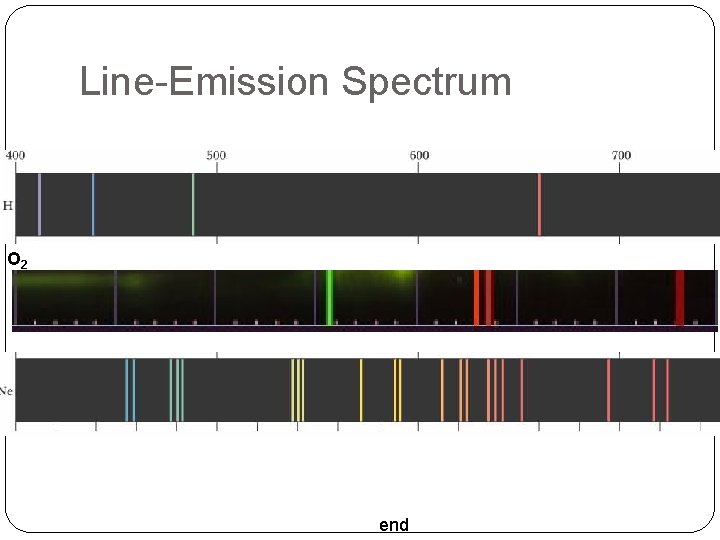
Line-Emission Spectrum O 2 end

Atomic Theories- Bohr Niels Bohr (1913) Electrons are placed into Energy Levels Circular orbits around the nucleus Number of electrons in each orbit = 2 n 2 Cannot be between energy Levels Gain energy, electrons move up energy levels To an Excited State Release energy, electrons move down energy levels To the Ground State Bigger Move = More Energy end
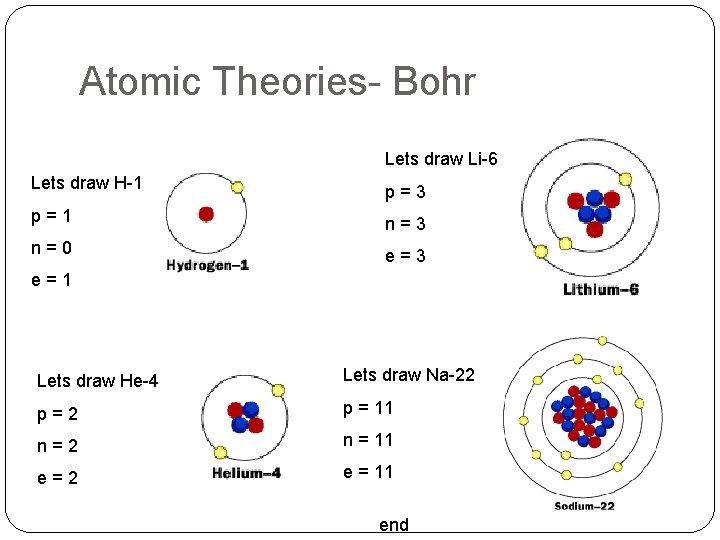
Atomic Theories- Bohr Lets draw Li-6 Lets draw H-1 p=3 p=1 n=3 n=0 e=3 e=1 Lets draw He-4 Lets draw Na-22 p=2 p = 11 n=2 n = 11 e=2 e = 11 end
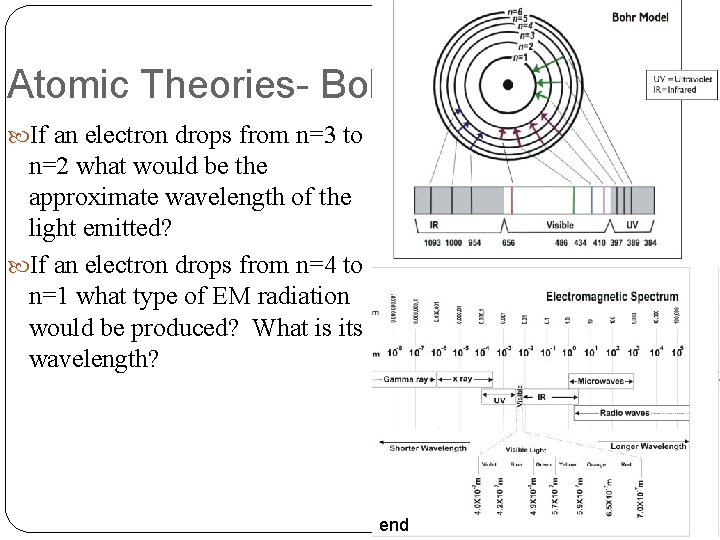
Atomic Theories- Bohr If an electron drops from n=3 to n=2 what would be the approximate wavelength of the light emitted? If an electron drops from n=4 to n=1 what type of EM radiation would be produced? What is its wavelength? end

Atomic Theories- Bohr Determine the wavelength, frequency, and type of EM wave for each of the following transitions. n = 4 n = 1 n = 5 n = 2 n = 6 n = 3 end
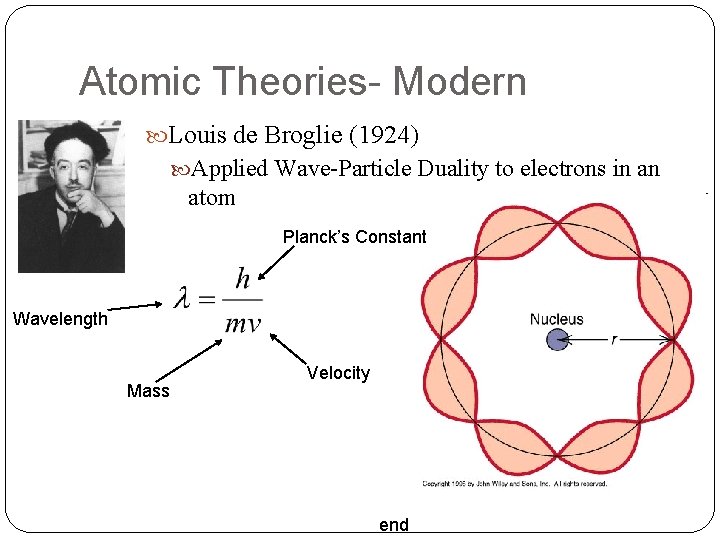
Atomic Theories- Modern Louis de Broglie (1924) Applied Wave-Particle Duality to electrons in an atom Planck’s Constant Wavelength Mass Velocity end

Atomic Theories- Modern Werner Heisenberg (1927) The Heisenberg Uncertainty Principle It is IMPOSSIBLE to know BOTH the position and velocity of a particle at the same time end

Atomic Theories- Modern Erwin Schrödinger (1926) Derived an equation for describing the Hydrogen atom’s electrons as waves Predicts the shapes of an electron’s Atomic Orbital Equation has only been solved for the Hydrogen atom end

Atomic Orbitals & Quantum Numbers From the new research scientists created the Electron Cloud Model Nucleus with electrons orbiting in an area of high probability Atomic Orbital A 3 -D area where you have a 95% chance of finding an electron The Principal Quantum Number (n) Indicates the Energy Level 1, 2, 3, … 7 Bigger Numbers = Higher Energy Each Energy Level has sublevels end
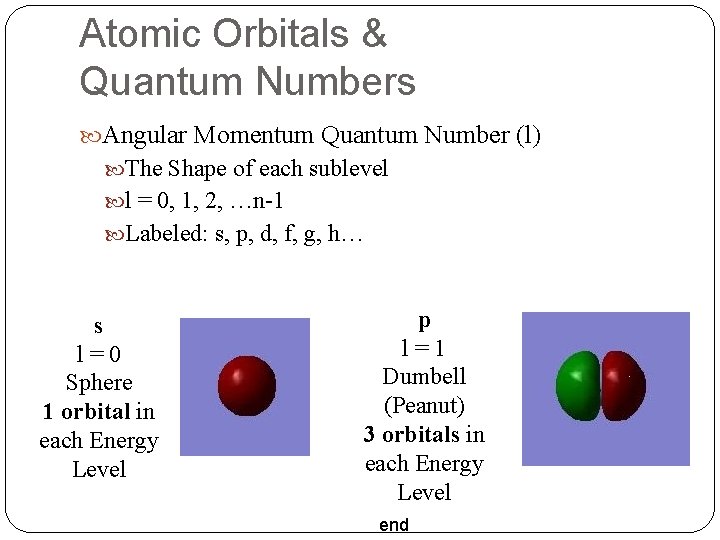
Atomic Orbitals & Quantum Numbers Angular Momentum Quantum Number (l) The Shape of each sublevel l = 0, 1, 2, …n-1 Labeled: s, p, d, f, g, h… s l=0 Sphere 1 orbital in each Energy Level p l=1 Dumbell (Peanut) 3 orbitals in each Energy Level end
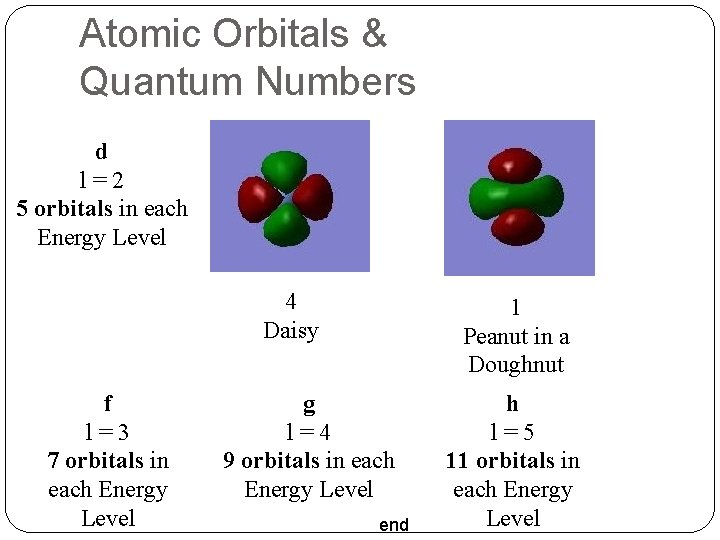
Atomic Orbitals & Quantum Numbers d l=2 5 orbitals in each Energy Level 4 Daisy f l=3 7 orbitals in each Energy Level 1 Peanut in a Doughnut g l=4 9 orbitals in each Energy Level end h l=5 11 orbitals in each Energy Level
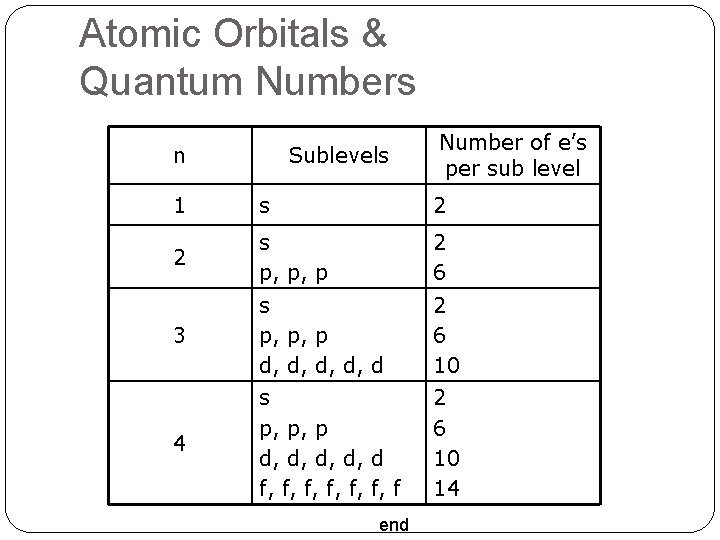
Atomic Orbitals & Quantum Numbers n Sublevels Number of e’s per sub level 1 s 2 2 s p, p, p 2 6 3 s p, p, p d, d, d 2 6 10 4 s p, p, p d, d, d f, f, f, f 2 6 10 14 end

Electron Configuration Electrons are arranged in a specific way according to 3 rules: RULE 1: Aufbau Principle Electrons will occupy the LOWEST energy level possible Energy Levels = 1 is the lowest Sublevels = s < p < d < f Sublevels “d” and “f” overlap with the next Energy Level end

Electron Configuration RULE 2: Pauli Exclusion Principle ONLY 2 electrons in each ORBITAL Electrons MUST have opposite Spins end
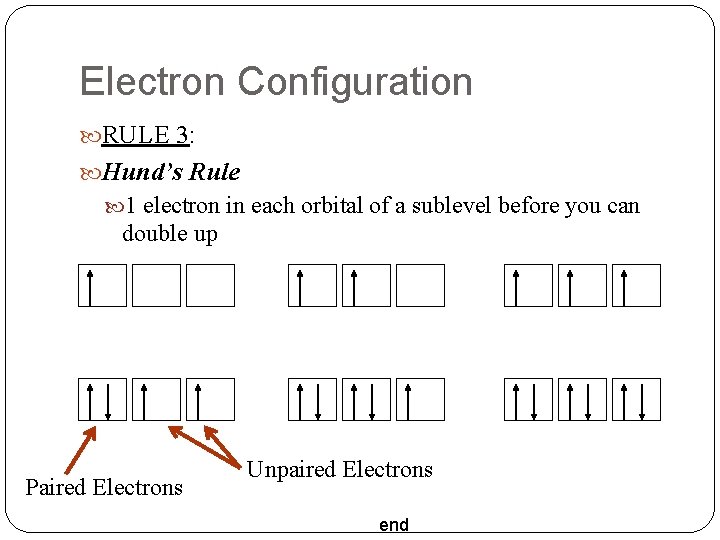
Electron Configuration RULE 3: Hund’s Rule 1 electron in each orbital of a sublevel before you can double up Paired Electrons Unpaired Electrons end

Electron Configuration Orbital Notation Draw a line and label each Energy Level and Sublevel Add arrows to represent electrons Li O Cl Ne Ca V end

Electron Configuration Notation Instead of drawing all of the lines, use superscripts Be F Mg He Ar B end

The Periodic Table By 1860 scientists had discovered 63 elements But no good way to organize them Scientists memorized everything This was changed by Mendeleev end
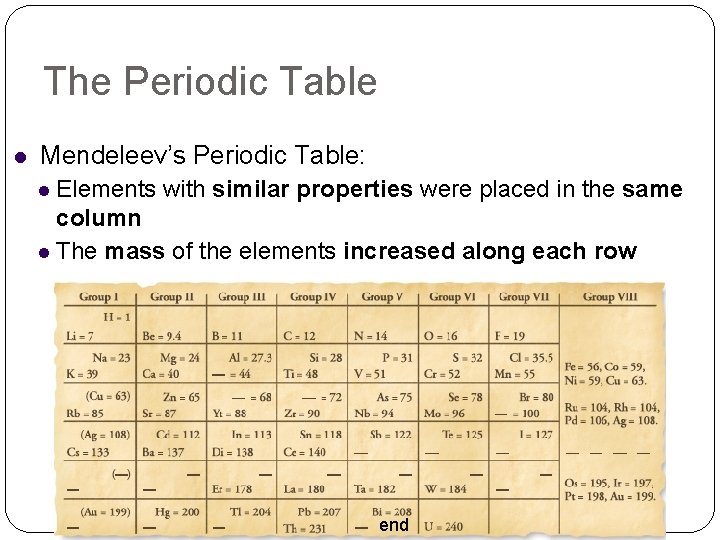
The Periodic Table Mendeleev’s Periodic Table: Elements with similar properties were placed in the same column The mass of the elements increased along each row end

The Periodic Table Mendeleev left several blank spaces in his periodic table For elements that had not been discovered yet He correctly predicted the properties of these elements based on the elements around them end
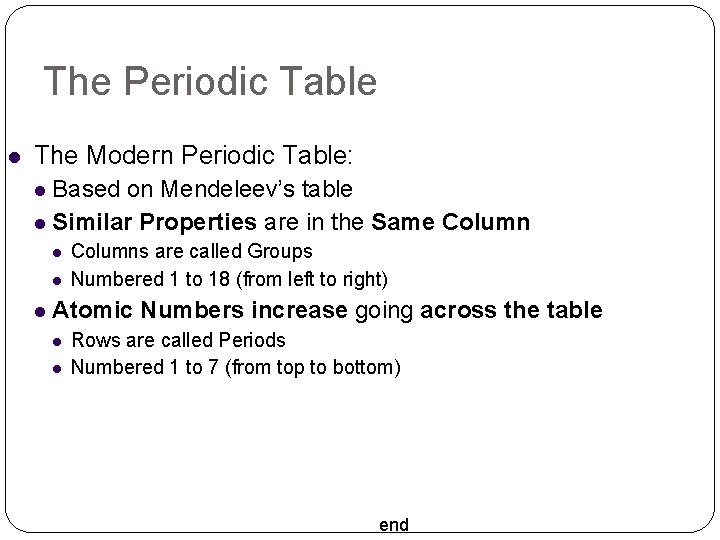
The Periodic Table The Modern Periodic Table: Based on Mendeleev’s table Similar Properties are in the Same Columns are called Groups Numbered 1 to 18 (from left to right) Atomic Numbers increase going across the table Rows are called Periods Numbered 1 to 7 (from top to bottom) end
The Periodic Table • This is the full Periodic Table • As you can see it is REALLY long • Way too long to fit on a page end
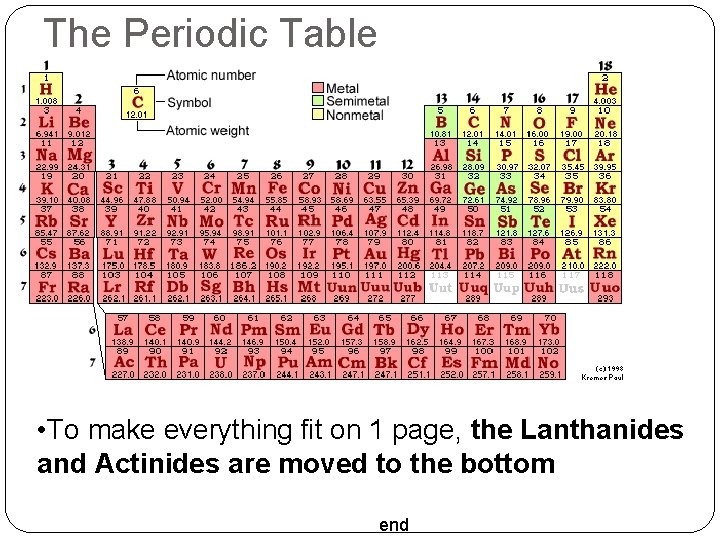
The Periodic Table • To make everything fit on 1 page, the Lanthanides and Actinides are moved to the bottom end
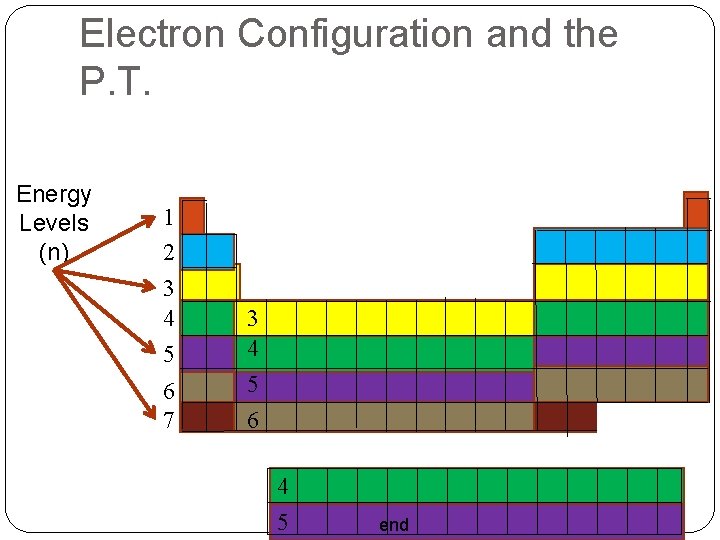
Electron Configuration and the P. T. Energy Levels (n) 1 2 3 4 5 6 7 3 4 5 6 4 5 end
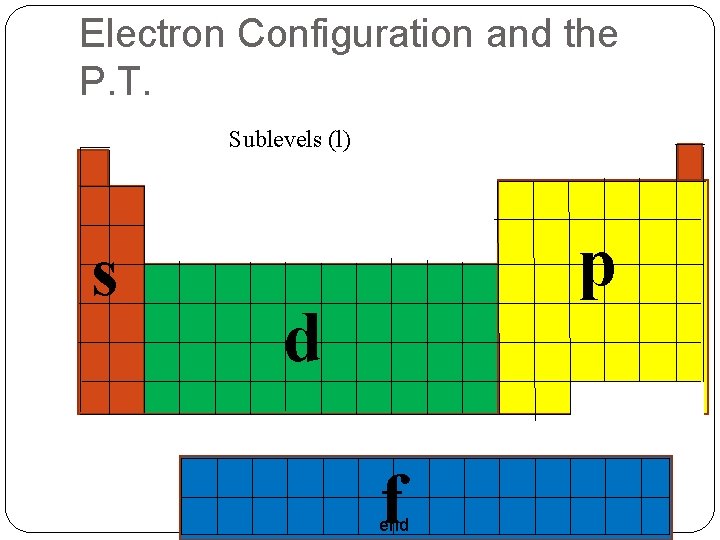
Electron Configuration and the P. T. Sublevels (l) s p d f end

Electron Configuration and the P. T. Instead of remembering the Aufbau diagram just read the Periodic Table Add 1 electron for each element you pass N Si Ti Mg Mo I end

The Periodic Table • Valence Electrons – Electrons in the highest energy level – Only thing involved in a chemical reaction – Give Elements their Chemical Properties Group: # of 1 1 2 2 13 3 14 4 Valence: end 15 5 16 6 17 7 18 8

The Periodic Table Metals Francium (Fr) is the most reactive Conduct electricity/heat Mostly solids at room temperature High melting/boiling points Can bend without breaking Nonmetals Fluorine (F) is the most reactive nonmetal Poorly conduct heat/electricity Mostly gases at room temperature Low melting/boiling points Break when bent end

The Periodic Table Metalloids Touching the stair-step line Properties between metals and nonmetals Depends on the temperature end
The Periodic Table Alkali Metals EXTREMELY REACTIVE! • Transition Metals – Wide Range of Properties • Lanthanide and Actinide Series – All are radioactive end
The Periodic Table Halogens Highly Reactive Noble Gases Extremely Unreactive THEY DO NOTHING! end

Periodic Trends • Atomic Radius – Francium (Fr) = Largest – Helium (He) = Smallest – Closer to Fr = Larger the Atom Ionic Radius Lose Electron = Positive Charge Cations are Smaller Metals Gain Electron = Negative Charge Anions are Larger Nonmetals end

Periodic Trends Ionization Energy (IE) Energy needed to remove 1 electron Metals like to give up electrons Fr will give electrons away Nonmetals must be forced to give them up He will never give away electrons end

Periodic Trends Electronegativity A measure of how much at atom attracts an electron Metals do NOT attract electrons Low electronegativities Nonmetals LOVE electrons (electrophilic) F has the highest electronegativity NOT He end
- Slides: 46