Electrons in Atoms and The Periodic Table Chapters

Electrons in Atoms and The Periodic Table Chapters 4 and 5

Unanswered Questions Where are the electrons placed around the nucleus? Why are the electrons NOT pulled into the nucleus? Opposites attract Answers came from a new understanding of LIGHT. end
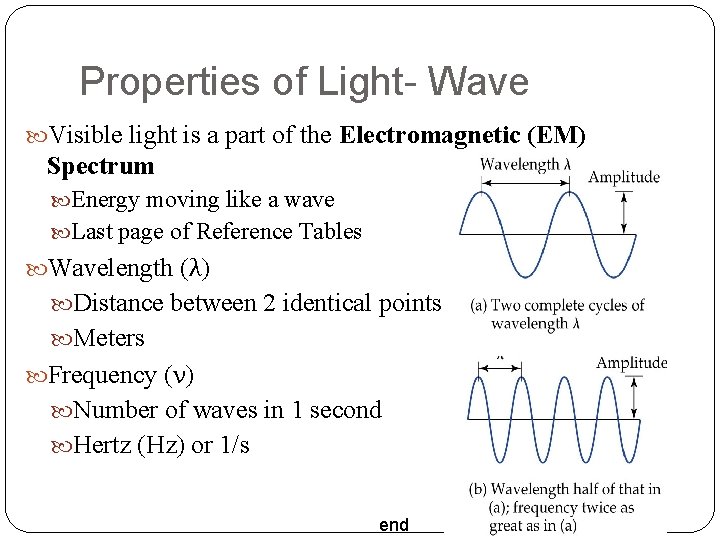
Properties of Light- Wave Visible light is a part of the Electromagnetic (EM) Spectrum Energy moving like a wave Last page of Reference Tables Wavelength (λ) Distance between 2 identical points Meters Frequency (ν) Number of waves in 1 second Hertz (Hz) or 1/s end
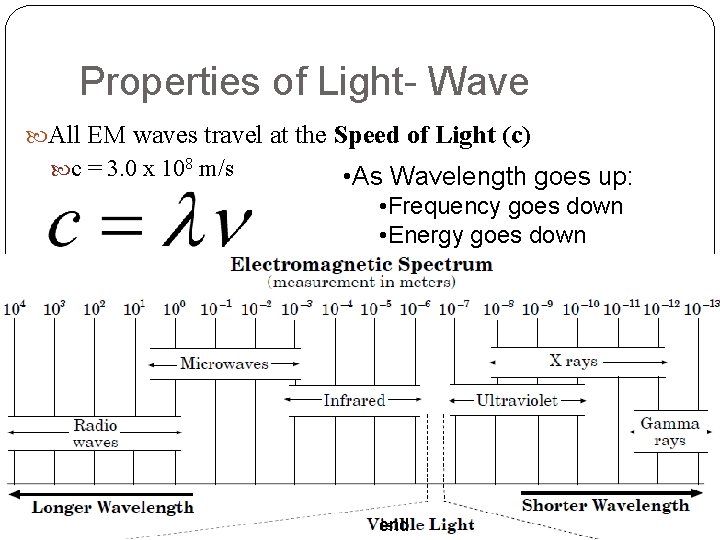
Properties of Light- Wave All EM waves travel at the Speed of Light (c) c = 3. 0 x 108 m/s • As Wavelength goes up: • Frequency goes down • Energy goes down end

Properties of Light- Wave A gamma ray has a wavelength of 7 x 10 -13 m. What is the frequency of this ray? A satellite transmits on the EM Spectrum at a frequency of 9. 4 x 109 Hz. What is the signal’s wavelength? What type of wave is being transmitted? A beam of visible light has a frequency of 4. 5 x 1014 Hz. What is the wavelength of this light? What color is this light? end

Properties of Light- Particle Light does not always act like a wave The Photoelectric Effect Metal gives off electrons when light shines on it Depends on the frequency Not the amount Max Planck Energy is gained/lost energy in specific amounts known as Quanta Frequency Energy Planck’s Constant (6. 626 x 10 -34 J·s) end

Properties of Light- Particle What is the energy of an EM wave with a frequency of 6. 32 x 1026 Hz? What energy would a beam of microwaves have if their wavelength is 5. 6 x 10 -2 m? A beam of visible light has a wavelength of 5. 8 x 10 -7 m. What is its energy and what color is it? end

Properties of Light- Both? ? ? Albert Einstein Wave-Particle Duality EM waves can act as both a wave and a particle Photon A particle of light end
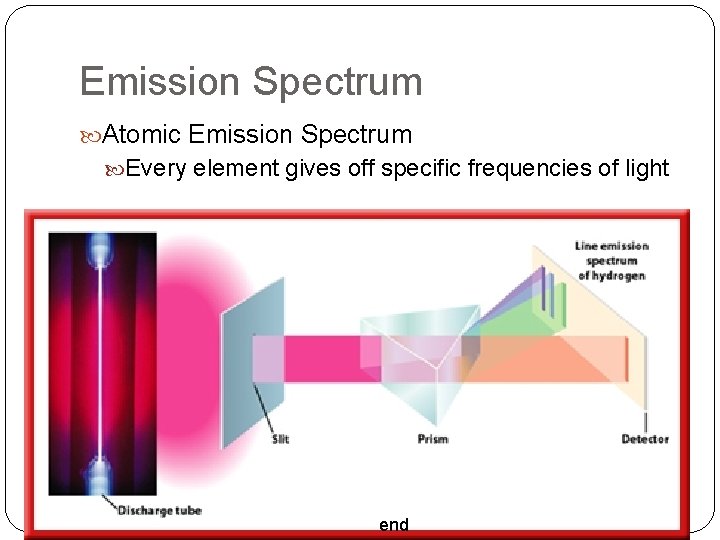
Emission Spectrum Atomic Emission Spectrum Every element gives off specific frequencies of light end
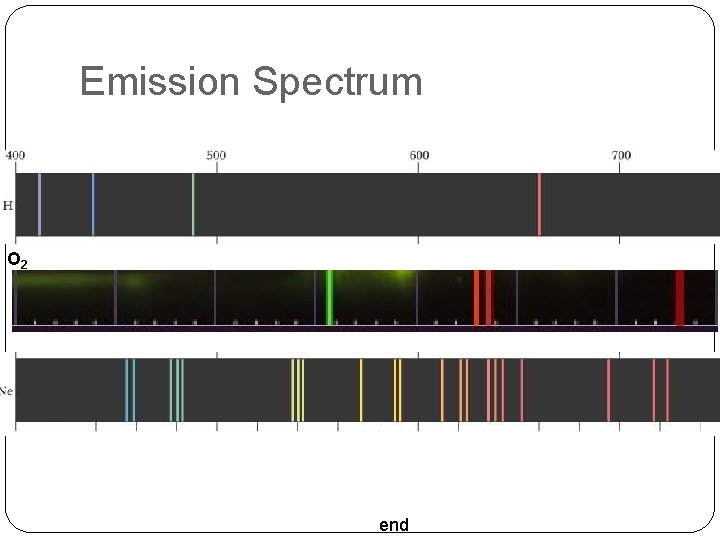
Emission Spectrum O 2 end

Properties of Light Wavelength (meters) 3. 0 x 108 m/s Frequency (Hertz- Hz) A gamma ray has a wavelength of 7 x 10 -13 m. What is the frequency of this ray? A satellite transmits on the EM Spectrum at a frequency of 9. 4 x 109 Hz. What is the signal’s wavelength? What type of wave is being transmitted? A beam of visible light has a frequency of 4. 5 x 1014 Hz. What is the wavelength of this light? end

Properties of Light Energy Frequency (Joules- J) Planck’s Constant (6. 626 x 10 -34 J·s) What is the energy of an EM wave with a frequency of 6. 32 x 1026 Hz? What energy would a beam of microwaves have if their wavelength is 5. 6 x 10 -2 m? A beam of visible light has a wavelength of 5. 8 x 10 -7 m. What is its energy and what color is it?

Bell Ringer: Light can be a particle and wave energy. True/False The Frequency of a Light is 7. 5 x 1014 Hz. What is its wavelength? What type of light is it? What is light as a particle called? Compare Frequency, Energy, and Wavelength of a Gamma Ray, and a Microwave. You need your ipad, & reference table out today!

Atomic Theories- Bohr Niels Bohr e- placed into Energy Levels (n) Rings around the nucleus # of electrons in each orbit = 2 n 2 Cannot be between energy Levels Gain Energy = e- move up levels To an Excited State Release Energy = e- move down levels To the Ground State Bigger Move = More Energy end

BOHR MODEL PRACTICE
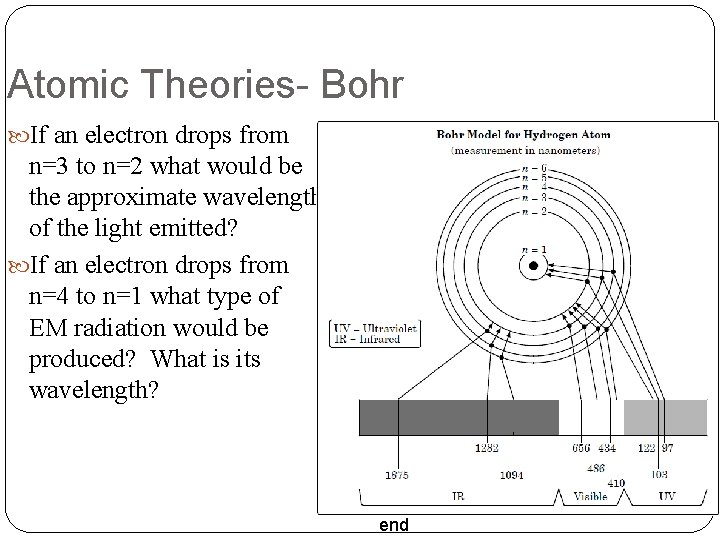
Atomic Theories- Bohr If an electron drops from n=3 to n=2 what would be the approximate wavelength of the light emitted? If an electron drops from n=4 to n=1 what type of EM radiation would be produced? What is its wavelength? end

Atomic Theories- Bohr Determine the wavelength, frequency, and type of EM wave for each of the following transitions. n = 4 n = 1 n = 5 n = 2 n = 6 n = 3 end


Atomic Orbitals & Quantum Numbers Electron Cloud Model e- orbit nucleus in Atomic Orbitals Area with 95% chance of finding an e Each orbital holds 2 e e-’s position (address) given with 4 Quantum Numbers The Principal Quantum Number (n) Indicates the Energy Level 1, 2, 3, … 7 Bigger Numbers = Higher Energy Each level has sublevels end
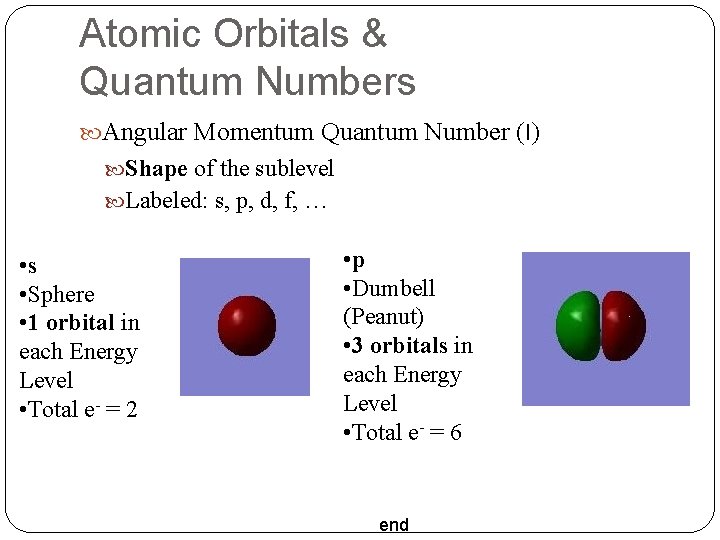
Atomic Orbitals & Quantum Numbers Angular Momentum Quantum Number (l) Shape of the sublevel Labeled: s, p, d, f, … • s • Sphere • 1 orbital in each Energy Level • Total e- = 2 • p • Dumbell (Peanut) • 3 orbitals in each Energy Level • Total e- = 6 end
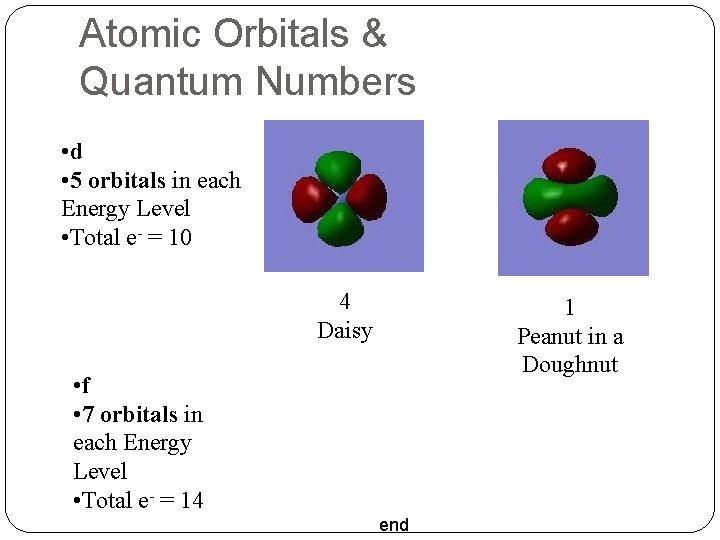
Atomic Orbitals & Quantum Numbers • d • 5 orbitals in each Energy Level • Total e- = 10 4 Daisy 1 Peanut in a Doughnut • f • 7 orbitals in each Energy Level • Total e- = 14 end




Electron Configuration e- are arranged according to 3 rules: Aufbau Principle e- will occupy the LOWEST energy level n = 1 is the lowest Sublevels = s < p < d < f “d” and “f” overlap with the next Energy Level end

Electron Configuration Pauli Exclusion Principle ONLY 2 e- in each ORBITAL e- MUST have opposite Spins end
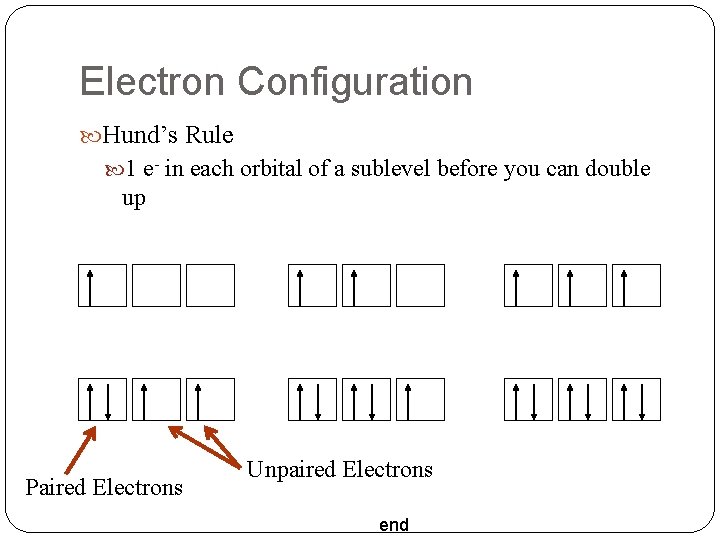
Electron Configuration Hund’s Rule 1 e- in each orbital of a sublevel before you can double up Paired Electrons Unpaired Electrons end

Electron Configuration Orbital Notation Draw line and label each Energy Level and Sublevel Add arrows to represent electrons Li O Cl Ne Ca V end

Electron Configuration Notation Instead of drawing all of the lines, use superscripts Be F Mg He Ar B end
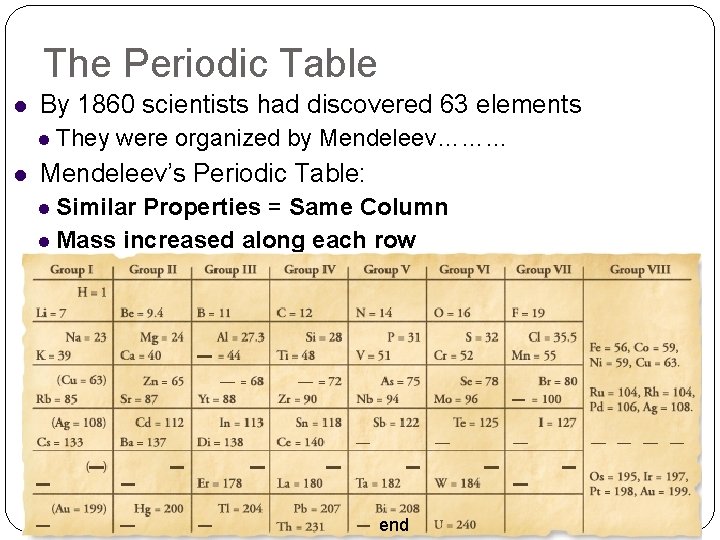
The Periodic Table By 1860 scientists had discovered 63 elements They were organized by Mendeleev……… Mendeleev’s Periodic Table: Similar Properties = Same Column Mass increased along each row end
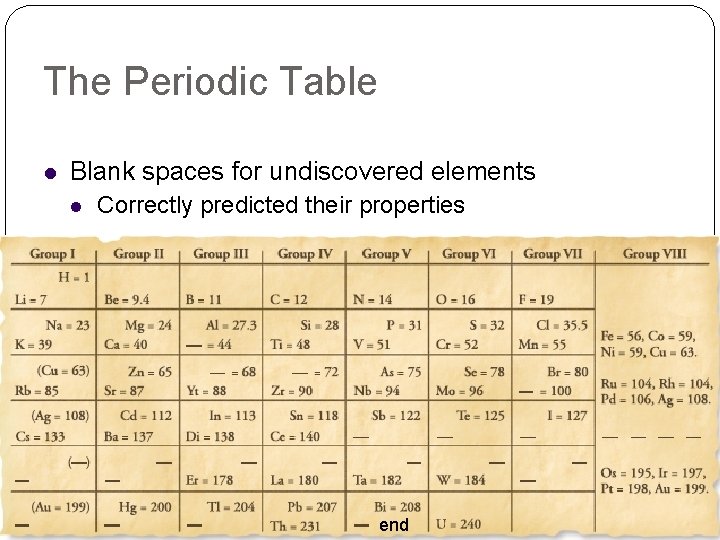
The Periodic Table Blank spaces for undiscovered elements Correctly predicted their properties end
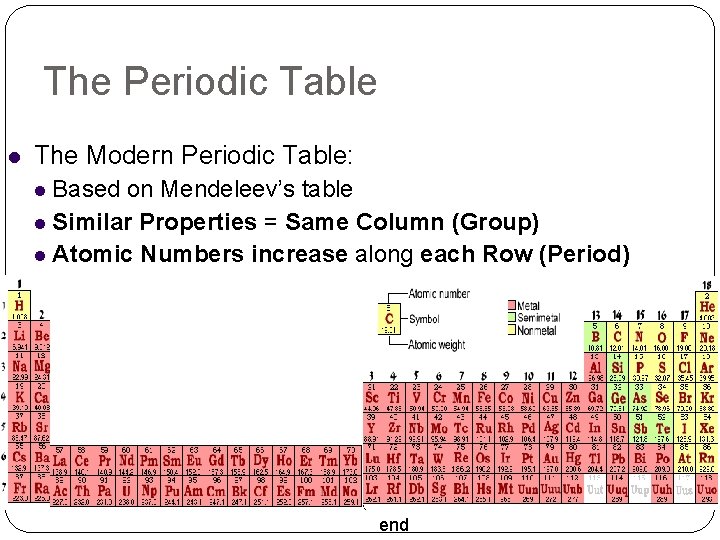
The Periodic Table The Modern Periodic Table: Based on Mendeleev’s table Similar Properties = Same Column (Group) Atomic Numbers increase along each Row (Period) end
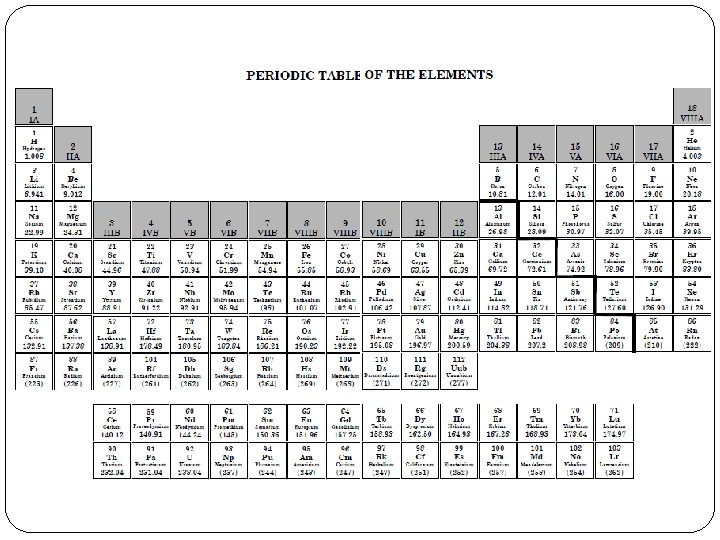
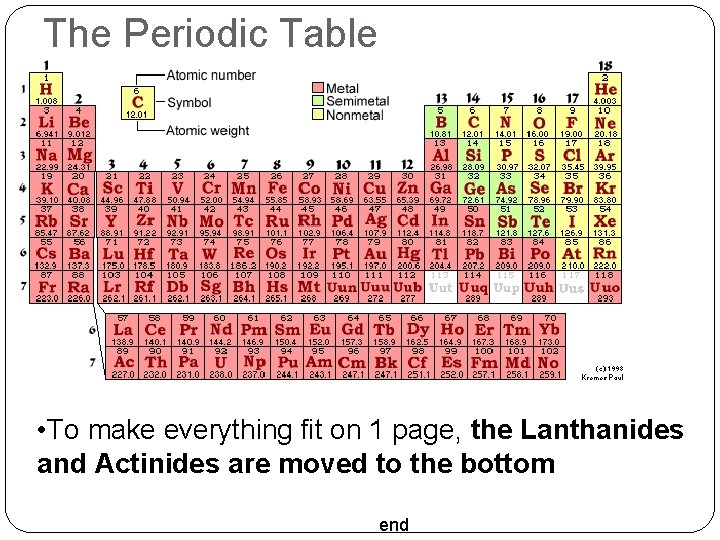
The Periodic Table • To make everything fit on 1 page, the Lanthanides and Actinides are moved to the bottom end
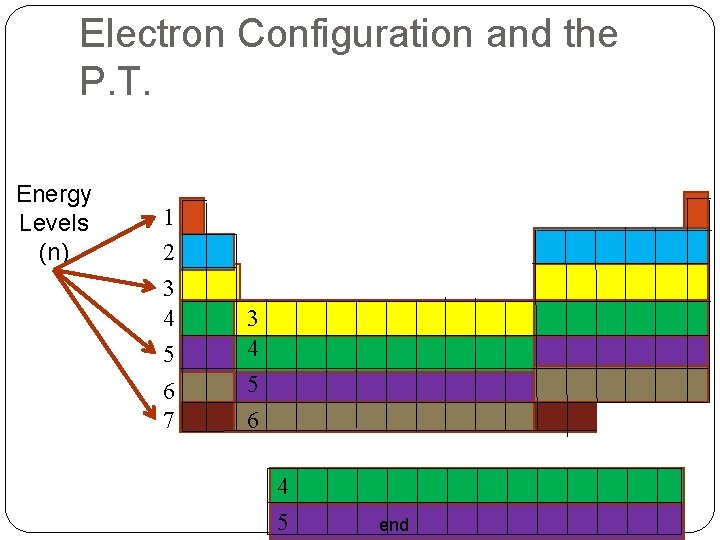
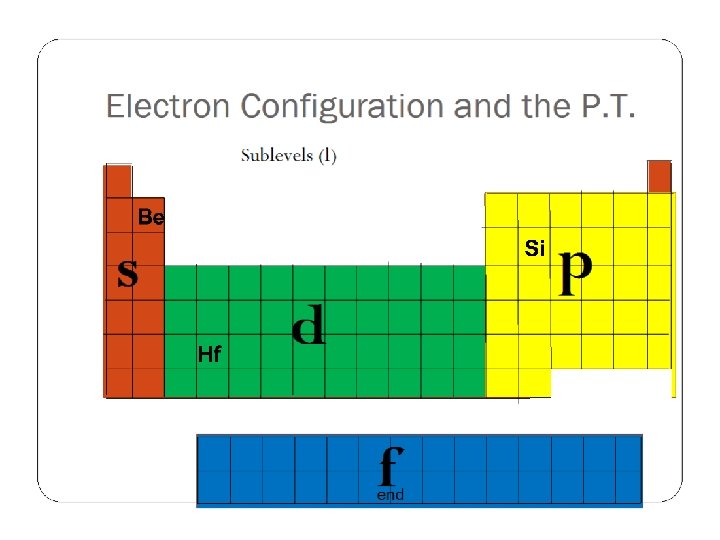
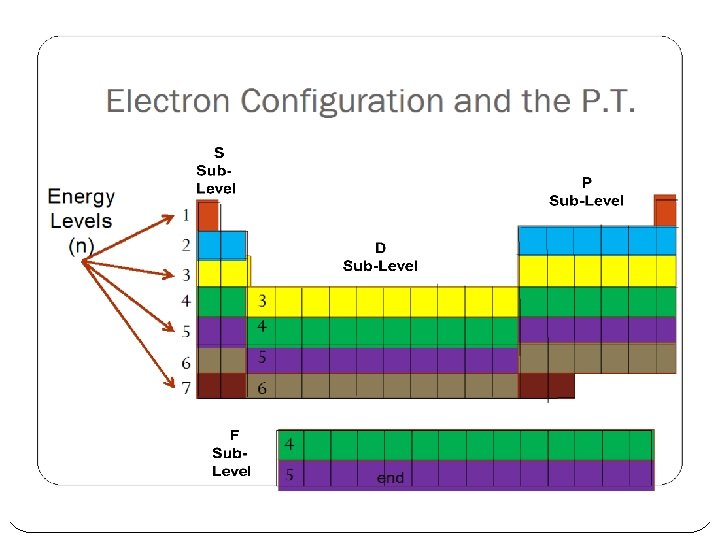
Electron Configuration and the P. T. Energy Levels (n) 1 2 3 4 5 6 7 3 4 5 6 4 5 end
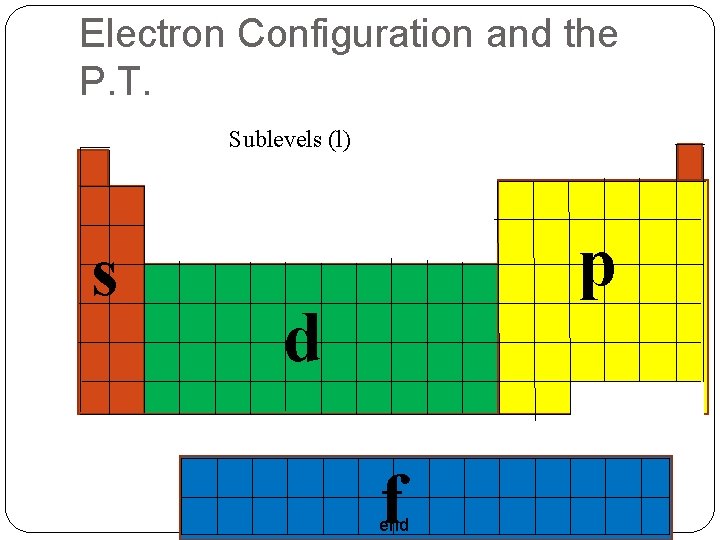
Electron Configuration and the P. T. Sublevels (l) s p d f end

Electron Configuration and the P. T. Instead of the Aufbau diagram, read the Periodic Table Add 1 electron for each element you pass N Si Ti Mg Mo I end
![Electron Configuration Noble-Gas Notation Find the Noble Gas before the element Write [Symbol], then Electron Configuration Noble-Gas Notation Find the Noble Gas before the element Write [Symbol], then](http://slidetodoc.com/presentation_image_h2/94e73b65064d11ec1dea1b341f7e84fd/image-43.jpg)
Electron Configuration Noble-Gas Notation Find the Noble Gas before the element Write [Symbol], then continue with the electron configuration Follow the Periodic Table Na Al K S As Sn end

Chapter 5: Periodic Table & Trends

Bell Ringer Draw a Bohr Model for the following elements: Phosporous Write the electron configuration Write the orbital configuration How many electrons are in the highest energy level for this elements?

The Periodic Table • Valence Electrons – Electrons in the highest energy level – Give Elements their Chemical Properties – Atoms want 8 Valence Electrons (an Octet) Filled “s” and “p” orbitals Find valence electrons for: N Mg As Ar K Y end
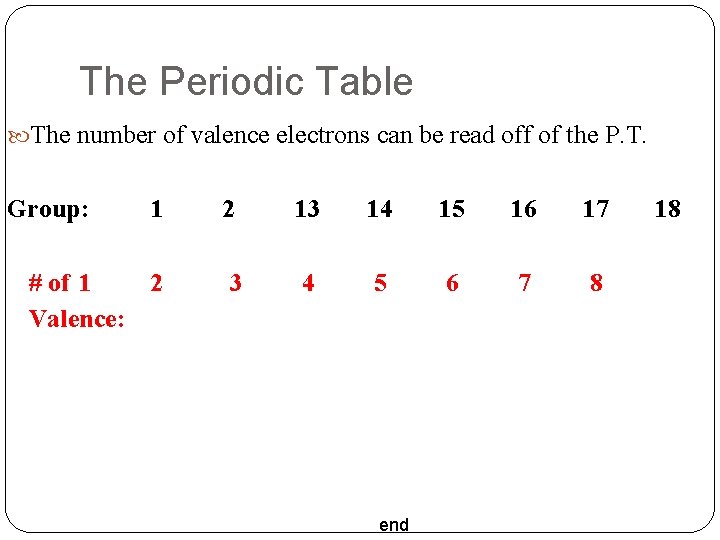
The Periodic Table The number of valence electrons can be read off of the P. T. Group: 1 2 13 14 15 16 17 # of 1 2 Valence: 3 4 5 6 7 8 end 18
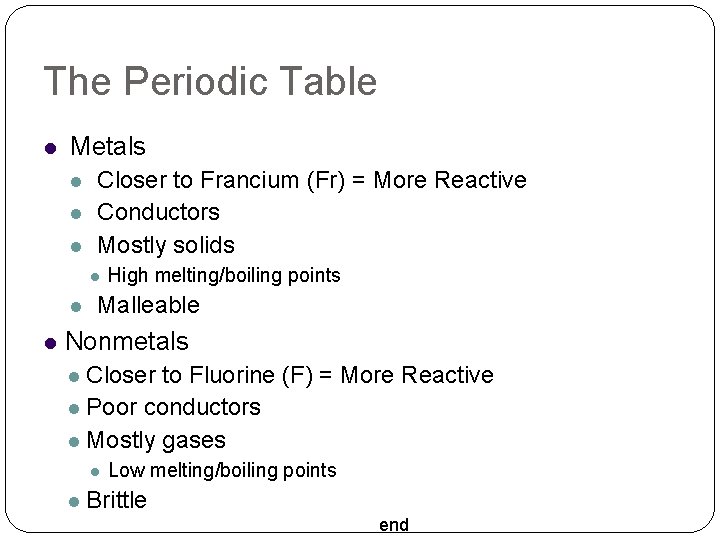
The Periodic Table Metals Closer to Francium (Fr) = More Reactive Conductors Mostly solids High melting/boiling points Malleable Nonmetals Closer to Fluorine (F) = More Reactive Poor conductors Mostly gases Low melting/boiling points Brittle end

The Periodic Table Metalloids Touching the stair-step line Properties depends on temperature end
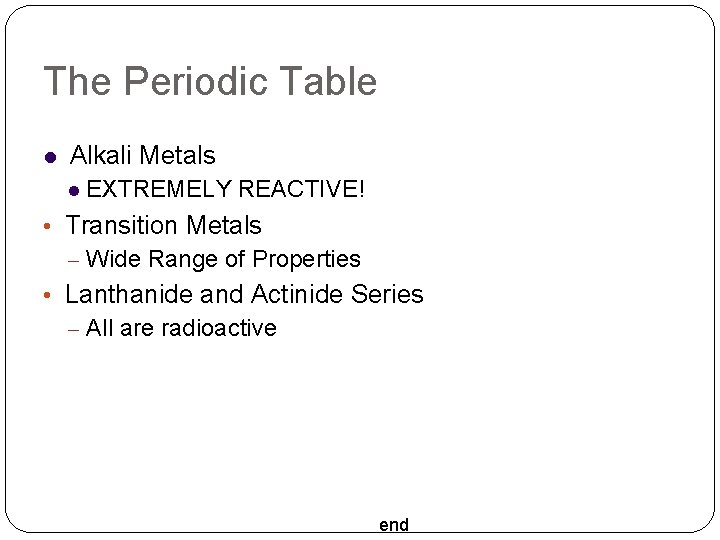
The Periodic Table Alkali Metals EXTREMELY REACTIVE! • Transition Metals – Wide Range of Properties • Lanthanide and Actinide Series – All are radioactive end
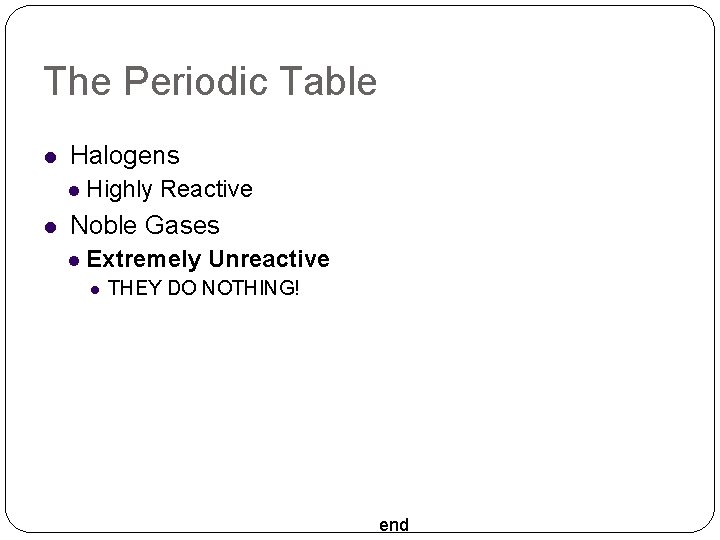
The Periodic Table Halogens Highly Reactive Noble Gases Extremely Unreactive THEY DO NOTHING! end
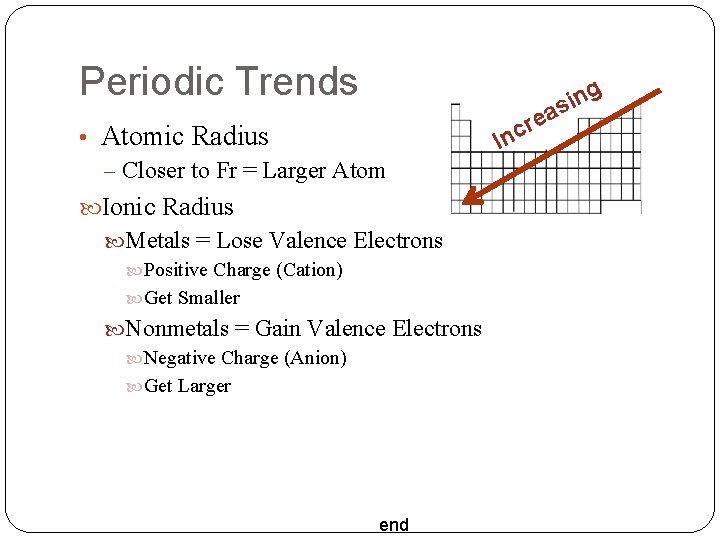
Periodic Trends • Atomic Radius – Closer to Fr = Larger Atom Ionic Radius Metals = Lose Valence Electrons Positive Charge (Cation) Get Smaller Nonmetals = Gain Valence Electrons Negative Charge (Anion) Get Larger end re c In g n i as
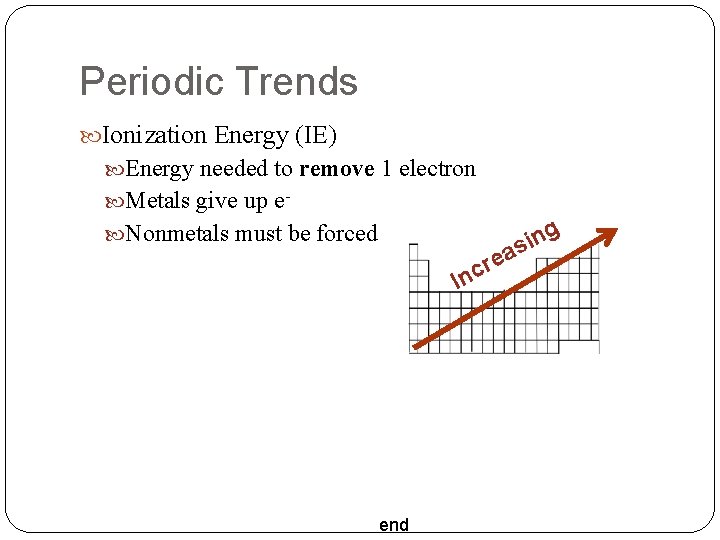
Periodic Trends Ionization Energy (IE) Energy needed to remove 1 electron Metals give up e- g n i as Nonmetals must be forced re c In end
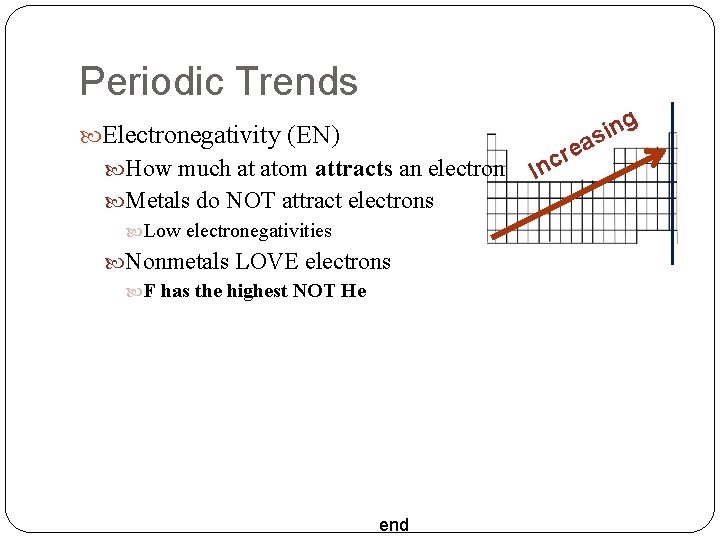
Periodic Trends Electronegativity (EN) How much at atom attracts an electron Metals do NOT attract electrons Low electronegativities Nonmetals LOVE electrons F has the highest NOT He end re c In g n i as
- Slides: 55