Electrons Atomic theory Overview 1 The Humble Beginnings

Electrons

Atomic theory Overview 1) The Humble Beginnings Democritus (460 -370 BC) and Leucippus (~500 BC) • The atom is an indestructible thing, it is the smallest piece that any substance can be broken in to. • It is indivisible, that is, it cannot be broken down any further.

2) Thousands of years passed: John Dalton (1808) • Atoms are the smallest part that any sample of element can be broken into. • Atoms of the same element have the same atomic mass, atoms of different elements have different atomic mass.

3) Not so much time passed: a Crookes Tube in J. J. Thomson (1897) • The atom is a sphere made of a diffuse (thin) positive charge, in which negatively charged electrons are embedded (stuck). • He called his model the “plum pudding” model, but who eats plum pudding anymore? It’s more like a “chocolate chip cookie dough” model, where the atom is a positively charged cookie dough ball with negative chocolate chip electrons stuck in it.

4) But then Ernest Rutherford discovered the alpha particle and HAD to play with it! (1911) • The atom is made of a small, dense, positively charged nucleus with electrons orbiting outside the nucleus at a distance with empty space making up the rest of the atom. • The majority of an atom’s volume is empty space, and the majority of the atom’s mass is in the nucleus.

5) He saw the light! Broken up into bright lines though a spectroscope! Neils Bohr! (1913) • Bohr observed the light given off when several elements are heated and give off light. Different elements gave off different colors of light. • When this light was passed through a prism, the light was broken up into lines of color. Each element’s lines were different. • Bohr figured that electrons falling from high energy levels to low energy levels were causing the light. • Each element’s spectrum of colored lines was different, meaning that the energy levels of different elements have a different amount of energy. • This process, called spectroscopy, is useful for identifying element samples.

6) Werner Heisenberg may have slept here: we’re uncertain! The Quantum-Mechanical Model • The atom contains a small, dense positive nucleus surrounded by electrons that travel in a wave-like motion around the nucleus. • This motion is modified by mass and charge interactions between electrons and the nucleus. • The interactions and the fast speed of the electron make it impossible to know with any certainty both where an electron is and where it is going in any particular instant. • All we can know is the general area of space in which the electron might be found. They very from the most general location to the most specific. • Electrons travel in principal energy levels, which are made up of sublevels, which are made up of orbitals that contain up to two electrons each. If two electrons are in the same orbital, they will spin in opposite directions.


4 Types of Electron Configuration of Elements
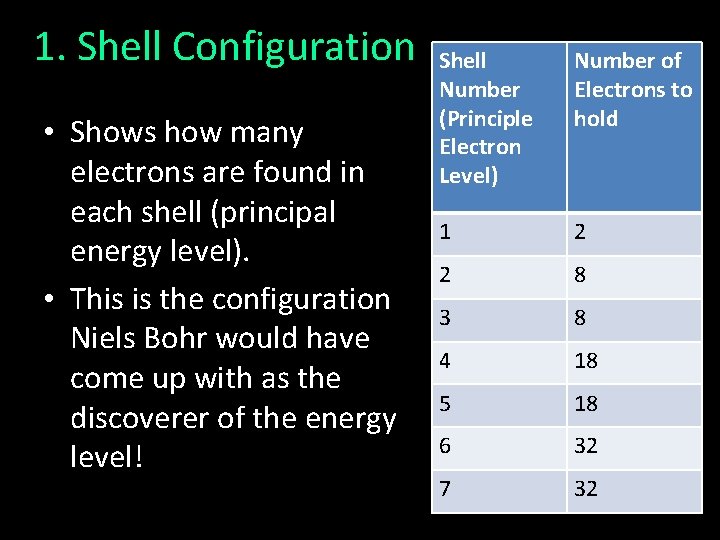
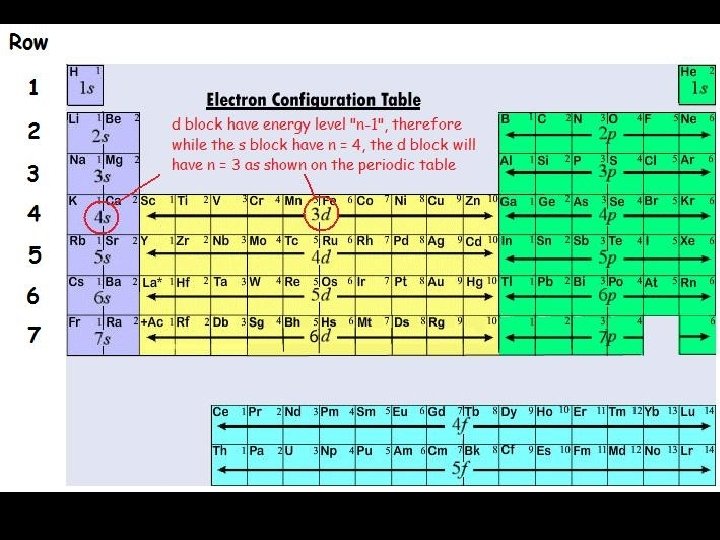
1. Shell Configuration • Shows how many electrons are found in each shell (principal energy level). • This is the configuration Niels Bohr would have come up with as the discoverer of the energy level! Shell Number (Principle Electron Level) Number of Electrons to hold 1 2 2 8 3 8 4 18 5 18 6 32 7 32

Shell Configuration (Bohr Diagrams) C 1) Draw a nucleus with the element symbol inside. 2) Carbon is in the 2 nd period, so it has two energy levels, or shells. 3) Draw the shells around the nucleus.

Shell Configuration (Bohr Diagrams) C 1) Add the electrons. 2) Carbon has 6 electrons. 3) The first shell can only hold 2 electrons.

Shell Configuration (Bohr Diagrams) C 1) Since you have 2 electrons already drawn, you need to add 4 more. 2) These go in the 2 nd shell. 3) Add one at a time starting on the right side and going counter clock-wise.

Shell Configuration (Bohr Diagrams) C 1) Check your work. 2) You should have 6 total electrons for Carbon. 3) Only two electrons can fit in the 1 st shell. 4) The 2 nd shell can hold up to 8 electrons. 5) The 3 rd shell can hold 18, but the elements in the first few periods only use 8 electrons.

2. Sublevel Electron Configuration • Principal energy levels are made up of sublevels, much as a town is made up of streets. • The expanded configuration tells you how many electrons are found in each sublevel of each PEL. • Most of the time (and for all of the configurations you will be responsible for), one sublevel must fill up completely before the next one can get any electrons.
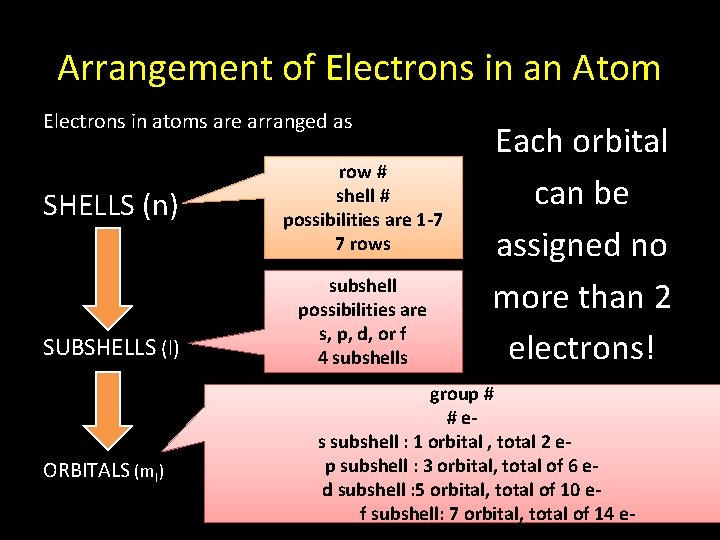
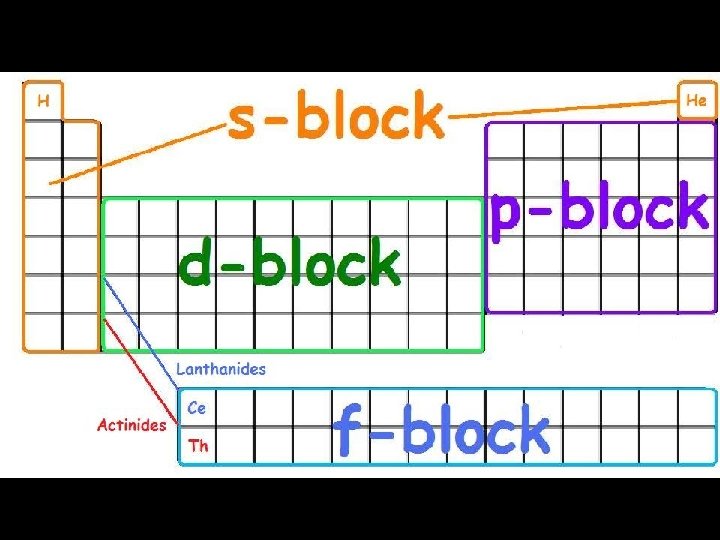
Arrangement of Electrons in an Atom Electrons in atoms are arranged as SHELLS (n) SUBSHELLS (l) ORBITALS (ml) row # shell # possibilities are 1 -7 7 rows subshell possibilities are s, p, d, or f 4 subshells Each orbital can be assigned no more than 2 electrons! group # # es subshell : 1 orbital , total 2 ep subshell : 3 orbital, total of 6 ed subshell : 5 orbital, total of 10 ef subshell: 7 orbital, total of 14 e-
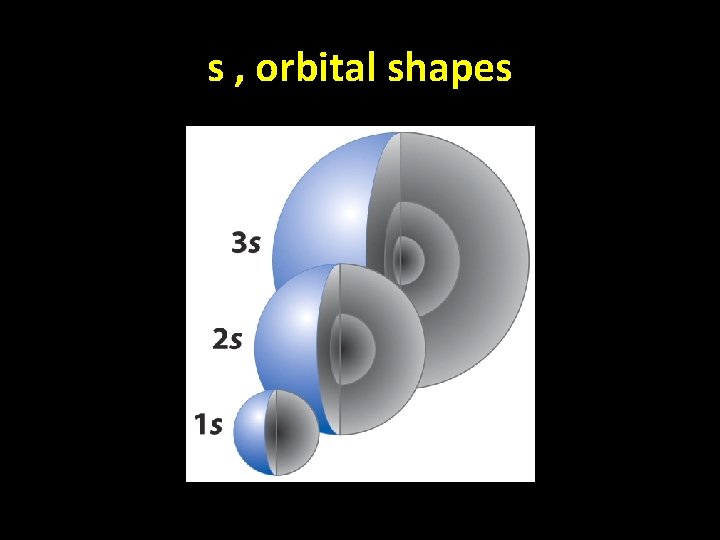
s , orbital shapes
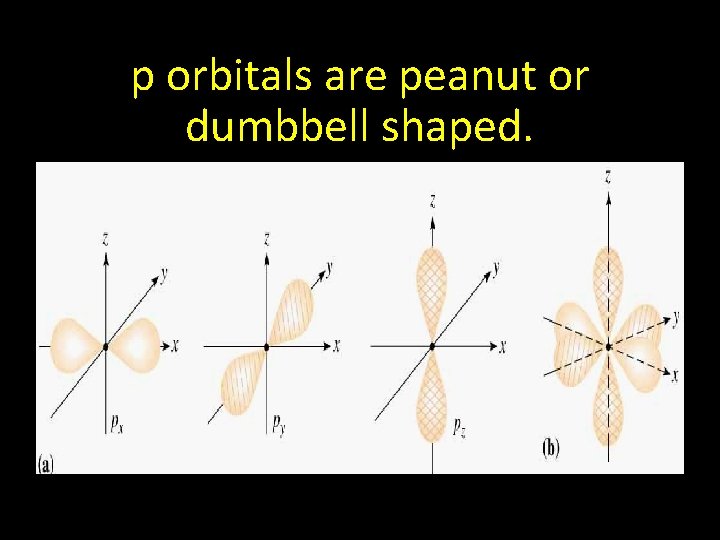
p orbitals are peanut or dumbbell shaped.
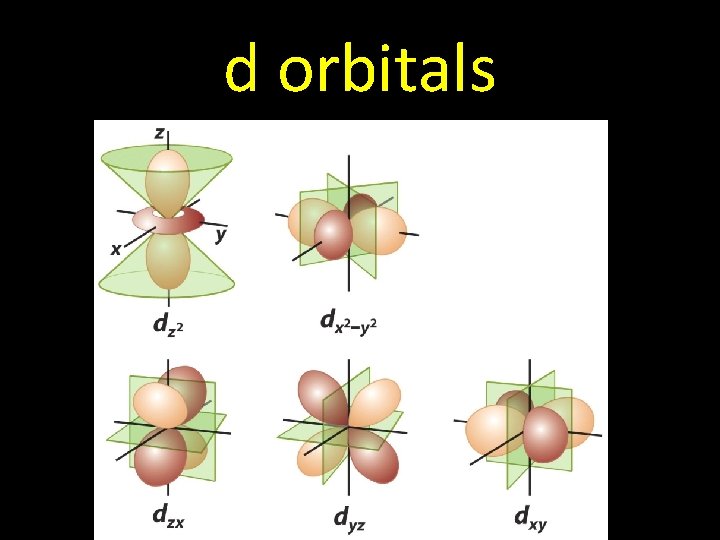
d orbitals
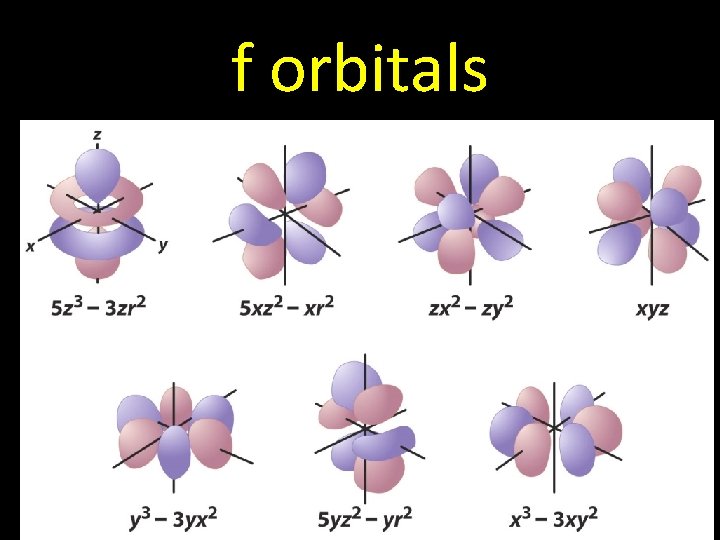
f orbitals
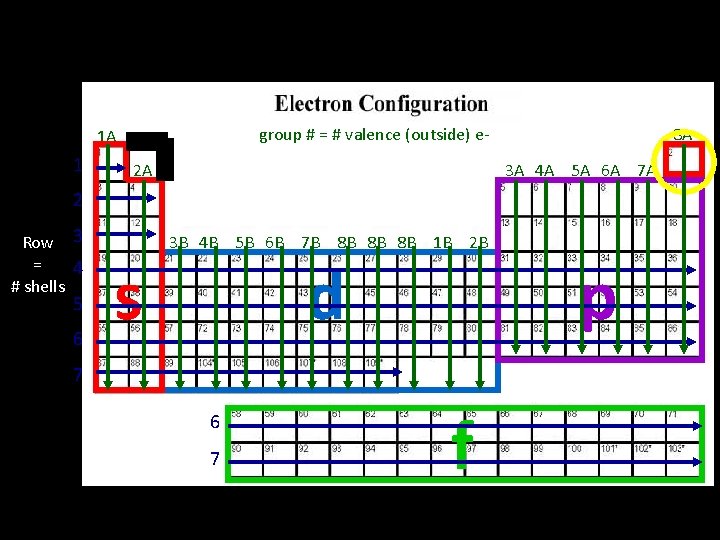
group # = # valence (outside) e- 1 A 1 2 A 8 A 3 A 4 A 5 A 6 A 7 A 2 Row 3 = 4 # shells 5 6 s 3 B 4 B 5 B 6 B 7 B 8 B 8 B 8 B 1 B 2 B d 7 6 7 f p
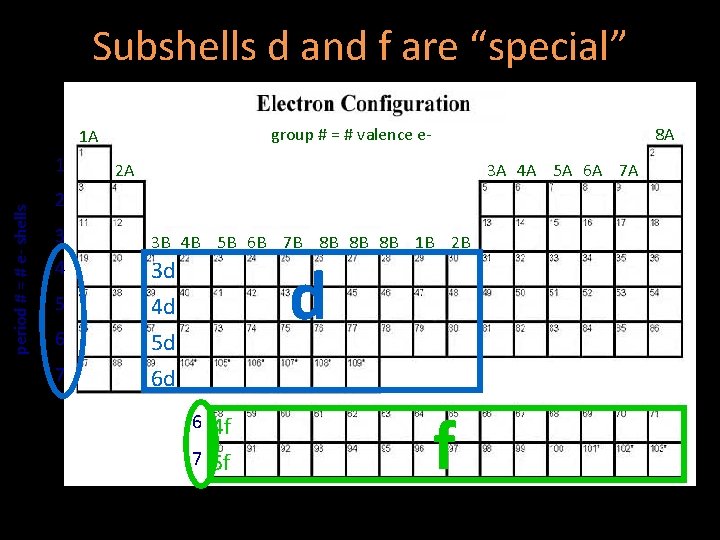
Subshells d and f are “special” group # = # valence e- 1 A period # = # e- shells 1 8 A 2 A 3 A 4 A 5 A 6 A 7 A 2 3 3 B 4 B 5 B 6 B 7 B 8 B 8 B 8 B 1 B 2 B 4 3 d 4 d 5 d 6 d 5 6 7 d 6 4 f 7 5 f f

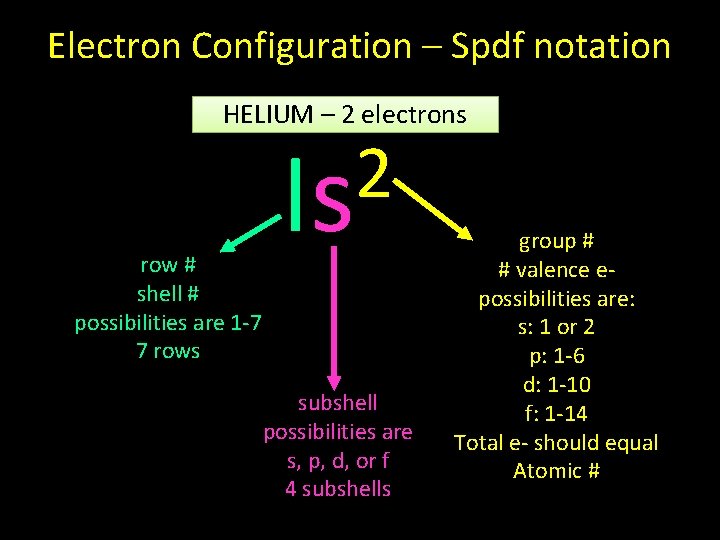
Electron Configuration – Spdf notation HELIUM – 2 electrons row # shell # possibilities are 1 -7 7 rows 2 Is subshell possibilities are s, p, d, or f 4 subshells group # # valence epossibilities are: s: 1 or 2 p: 1 -6 d: 1 -10 f: 1 -14 Total e- should equal Atomic #

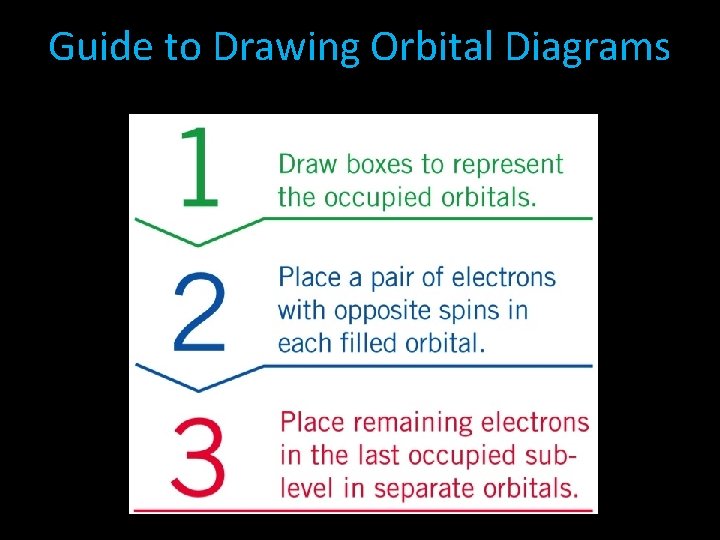
3. Orbital Box Diagram • Shows how many electrons are in each ORBITAL of each sublevel, and what each electron’s SPIN is. • Orbitals are all the same size, they can all fit up to two electrons in them. • The spin of electrons is indicated by arrows up and down. • If the orbital has two electrons in it, the first will have an up spin, and the second will have a down spin. • The number of arrows will equal the number of electrons in the sublevel.
Guide to Drawing Orbital Diagrams

Drawing Orbital Diagram Draw the orbital diagram for nitrogen. Step 1 Draw boxes to represent the occupied orbitals. Nitrogen has an atomic number of seven, which means it has seven electrons. Draw boxes to represent the 1 s, 2 s, and 2 p orbitals.
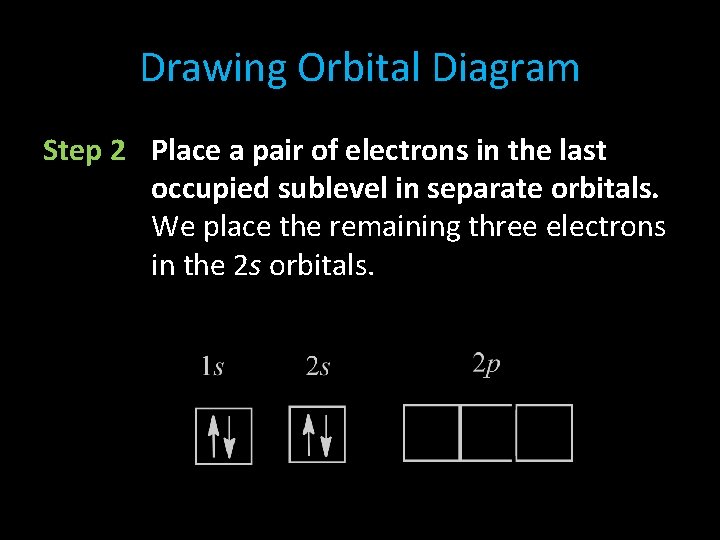
Drawing Orbital Diagram Step 2 Place a pair of electrons in the last occupied sublevel in separate orbitals. We place the remaining three electrons in the 2 s orbitals.

Drawing Orbital Diagram Step 3 Place remaining electrons with opposite spins in each filled orbital. First we place a pair of electrons with opposite spins in the 2 p orbitals, with arrows in the same direction.

Click Below for the Video Lectures Electron Configuration

4. Electron (Lewis) Dot Diagram • VALENCE ELECTRONS – the electrons in the outermost shell (furthest energy level from the nucleus), which is also called the valence shell. – The number of valence electrons that an atom has can be determined by the last number in the basic electron configuration. The number of valence electrons that an atom has determines its physical and chemical properties
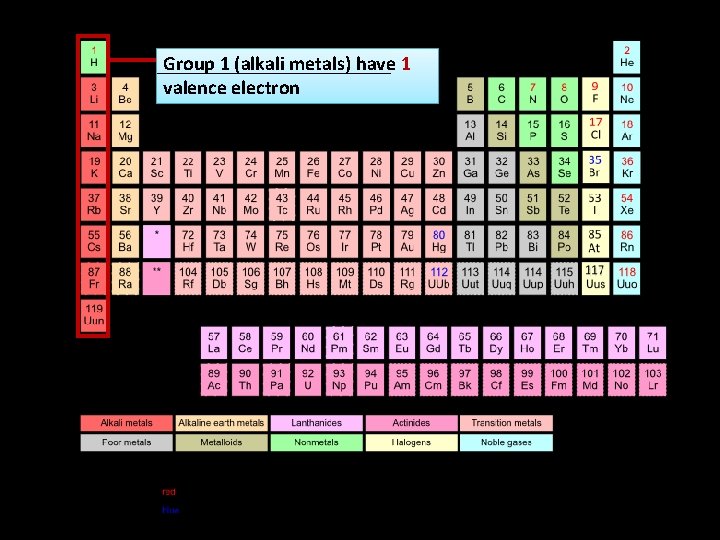
Group 1 (alkali metals) have 1 valence electron
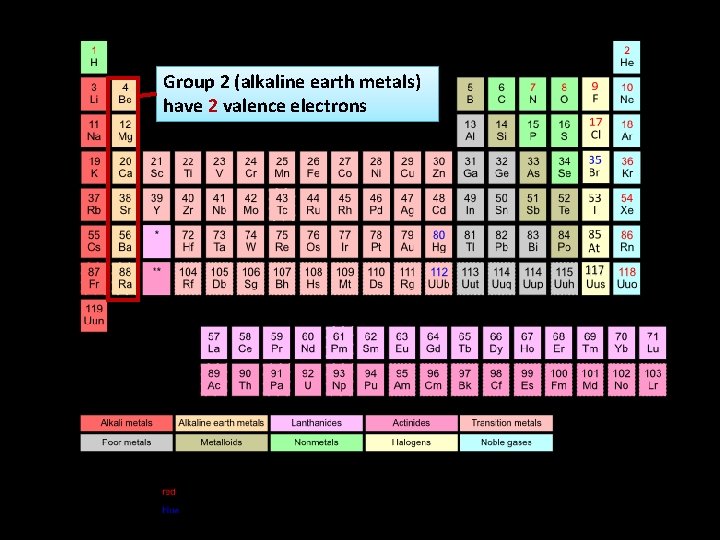
Group 2 (alkaline earth metals) have 2 valence electrons
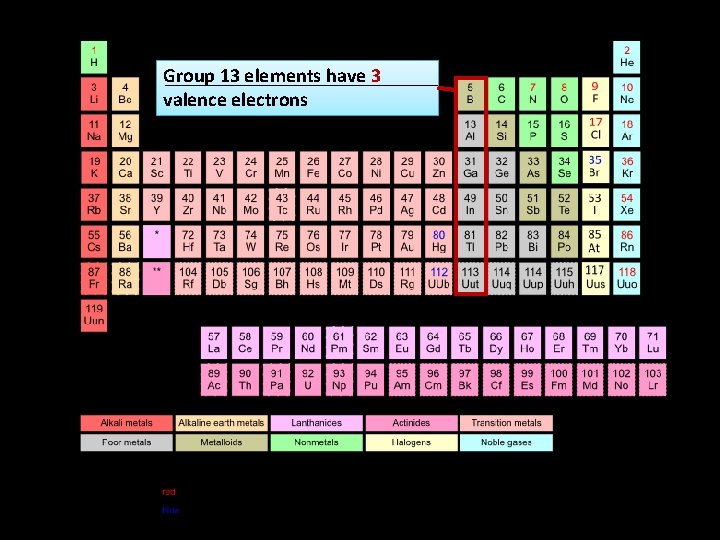
Group 13 elements have 3 valence electrons
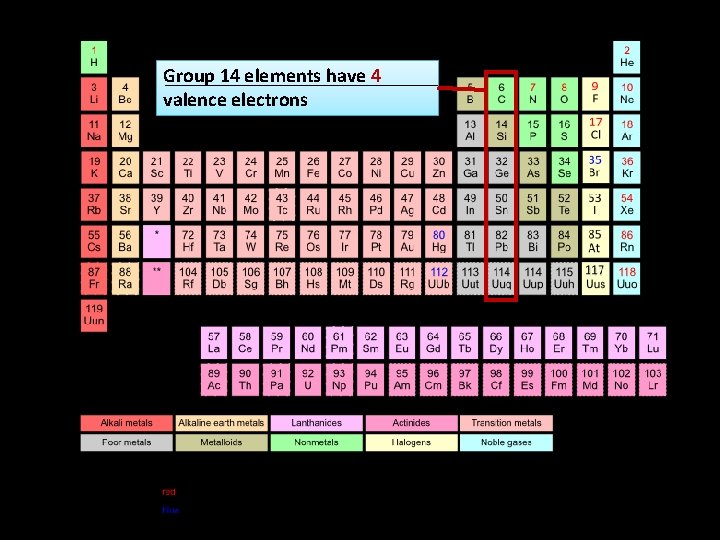
Group 14 elements have 4 valence electrons
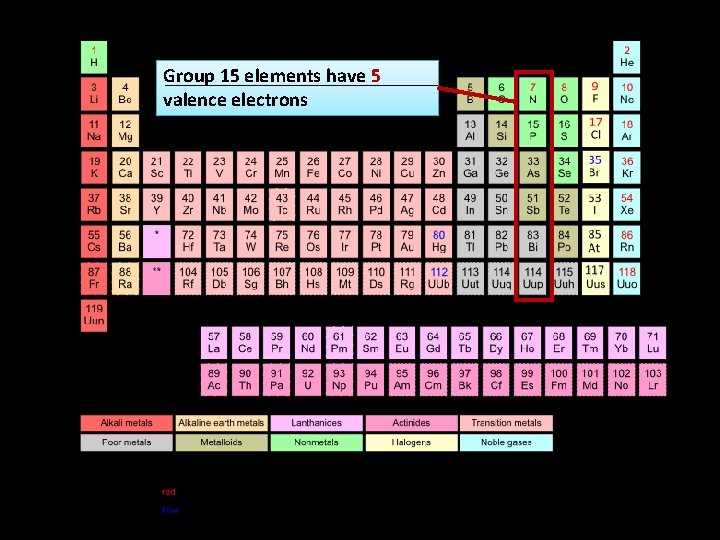
Group 15 elements have 5 valence electrons
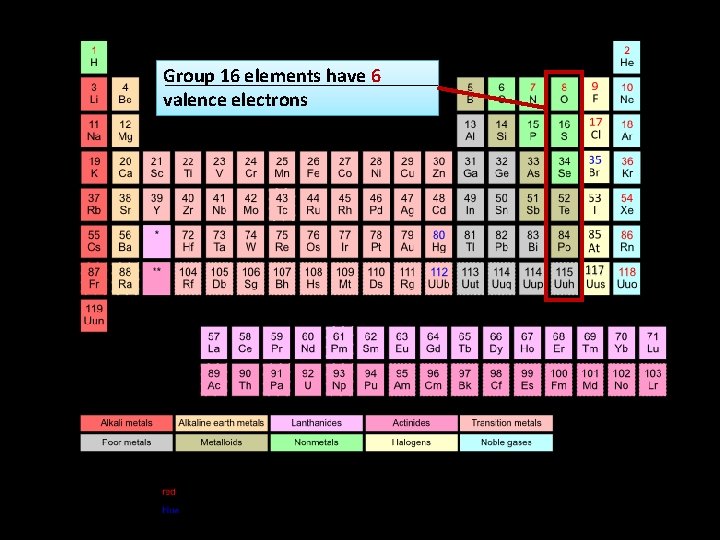
Group 16 elements have 6 valence electrons
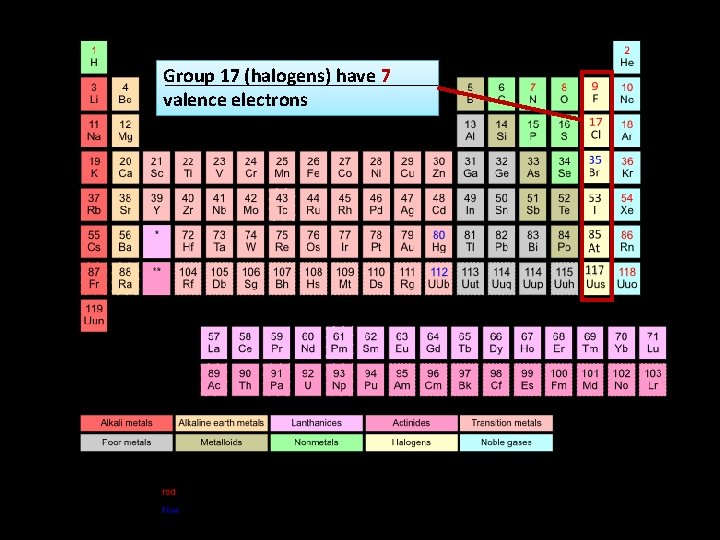
Group 17 (halogens) have 7 valence electrons
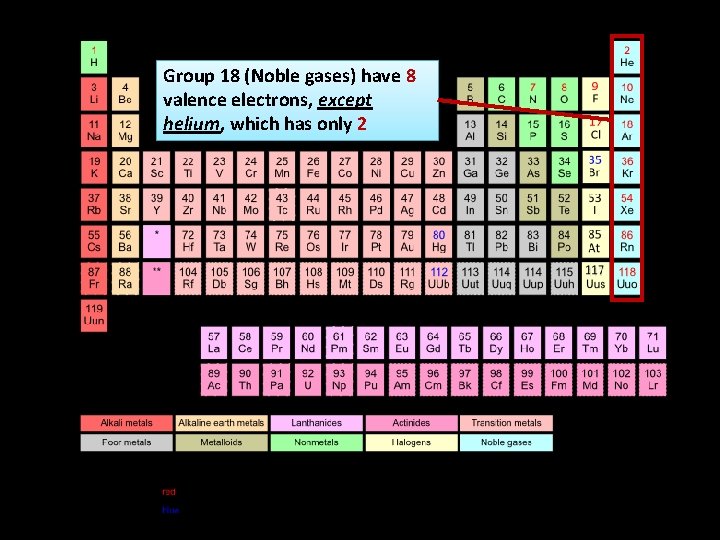
Group 18 (Noble gases) have 8 valence electrons, except helium, which has only 2
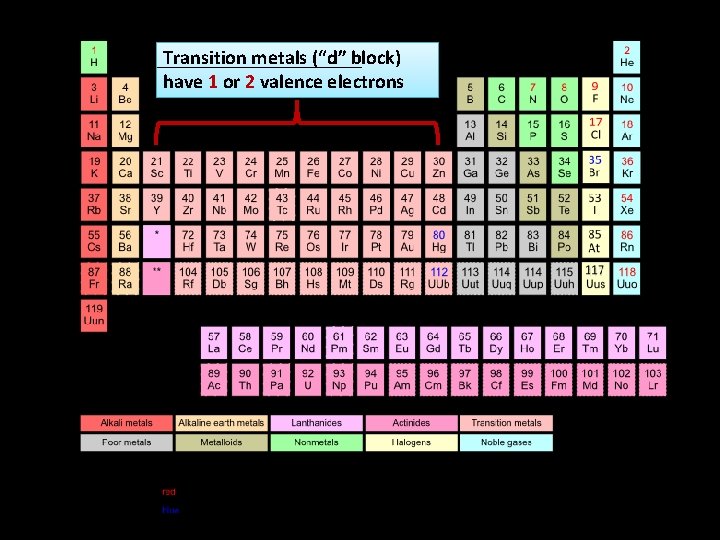
Transition metals (“d” block) have 1 or 2 valence electrons
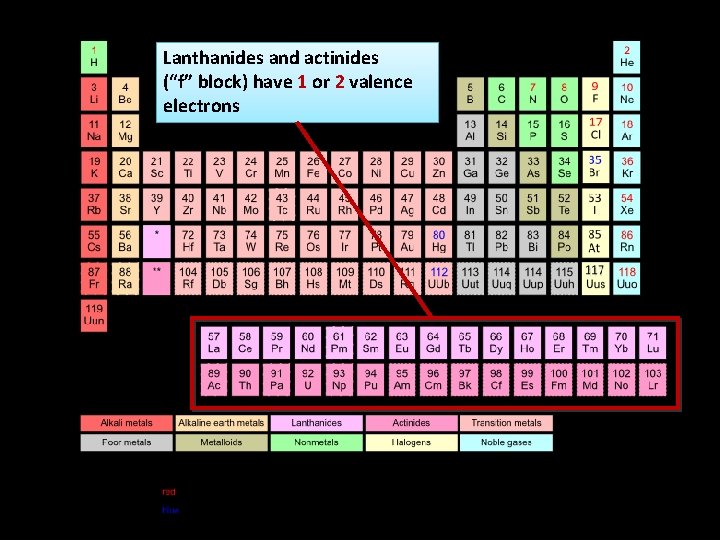
Lanthanides and actinides (“f” block) have 1 or 2 valence electrons

Lewis Dot Diagram • using dots in groups of 2 around the symbol of the atom to represent the valence electrons. • For every atom, the valence electrons will occupy only s and p orbitals. • The s electrons fill up first, then the p electrons fill, up electrons first, followed by the downs, just like in the box diagram.

The Electron Dot diagram for Nitrogen has 5 valence electrons. l First we write the symbol. l l. Then add 1 electron at a time to each side. l. Until they are forced to pair up. N
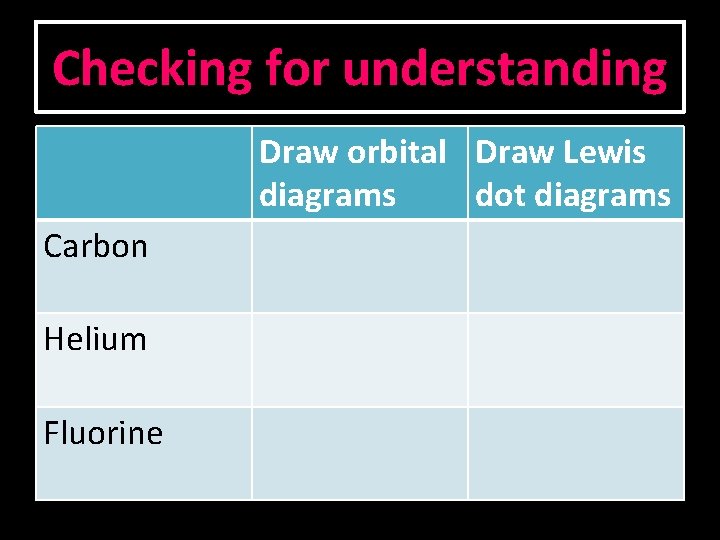
Checking for understanding Draw orbital Draw Lewis diagrams dot diagrams Carbon Helium Fluorine
- Slides: 46