Electrons and Chemical Bonds Part 1 G 8

Electrons and Chemical Bonds Part 1 G 8 Science Chapter 8. 2

Our Story So Far… • What is the difference between a compound a mixture? • How are covalent bonds formed? • How are ionic bonds formed? • Give one reason why chemical bonds are formed. • Give the other reason.
![Valence Electron • Valence electrons are in the outermost [valence] shell of an atom. Valence Electron • Valence electrons are in the outermost [valence] shell of an atom.](http://slidetodoc.com/presentation_image_h/137259b1247f5472c3b9a240fa145b83/image-3.jpg)
Valence Electron • Valence electrons are in the outermost [valence] shell of an atom. • For the purpose of making chemical bonds the valence shell is the only thing that matters.

8 is a magic number • For elements Boron (5) or greater, the valence shell is considered full if it has 8 electrons. • This is the same number of valence electrons as most noble gasses. • For elements less than Boron, the valence shell is considered full if it has 2 electrons. • Atoms will get rid of, gain, or share electrons to get a full valence shell.

Hydrogen is special • Hydrogen has one valence electron. • It may get rid of the electron and have a positive charge. • It may gain an electron and have a negative charge. • It may share electrons to get two valence electrons. What kind of bond is this?
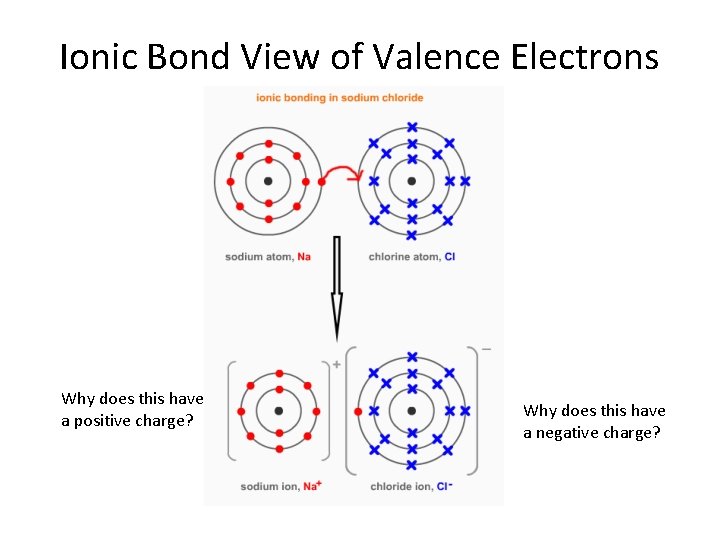
Ionic Bond View of Valence Electrons Why does this have a positive charge? Why does this have a negative charge?

Periodic Table View of Valence Electrons • As you go L-R on the table, each group [except transition metals] has one more valence electron. • The rule is: – Period 1 has 1 valence electron – Period 2 has 2 valence electrons – Period 13 -18 have (group # minus 10) valence electrons • As you go top to bottom on the table, each period has one more shell.

Your Turn • How many valence electrons does carbon have? • How many valence electrons does sodium have? • How many valence electrons does neon have? • How many valence electrons does chlorine have?
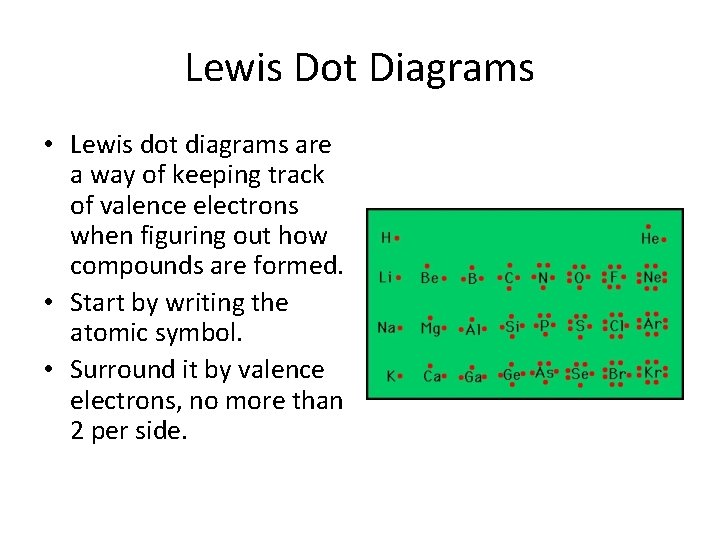
Lewis Dot Diagrams • Lewis dot diagrams are a way of keeping track of valence electrons when figuring out how compounds are formed. • Start by writing the atomic symbol. • Surround it by valence electrons, no more than 2 per side.
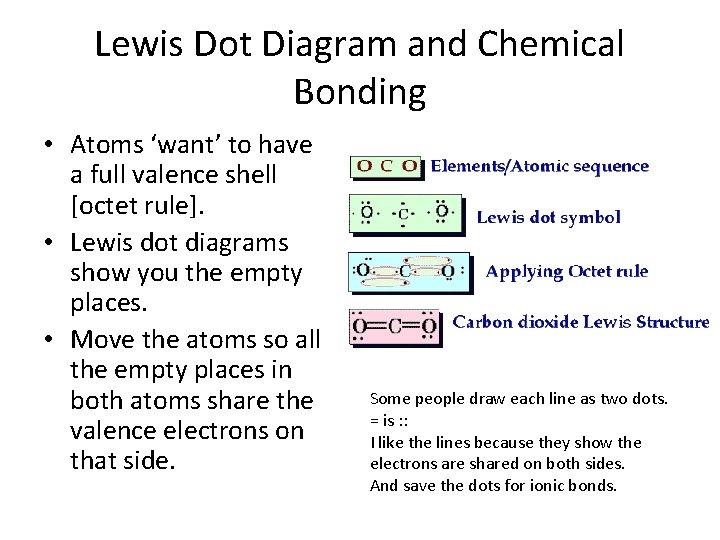
Lewis Dot Diagram and Chemical Bonding • Atoms ‘want’ to have a full valence shell [octet rule]. • Lewis dot diagrams show you the empty places. • Move the atoms so all the empty places in both atoms share the valence electrons on that side. Some people draw each line as two dots. = is : : I like the lines because they show the electrons are shared on both sides. And save the dots for ionic bonds.

Your Turn • Draw the Lewis dot diagram for F 2. What kind of bond is this? • Draw the Lewis dot diagram for Be. O. What kind of bond is this? • Draw the Lewis dot diagram for H 2 O. What kind of bond is this?

Activity • Handout • Side 1 is just drawing the Lewis dot diagrams. • Side 2 is using Lewis dot diagrams to draw ionic and covalent bonds. • Due at end of class. • If you get done early, work on homework – do not socialize.
- Slides: 12