Electronic UVvisible Spectroscopy Electronic XPS UPS UVvisible 1

Electronic (UV-visible) Spectroscopy | Electronic | XPS UPS UV-visible 1
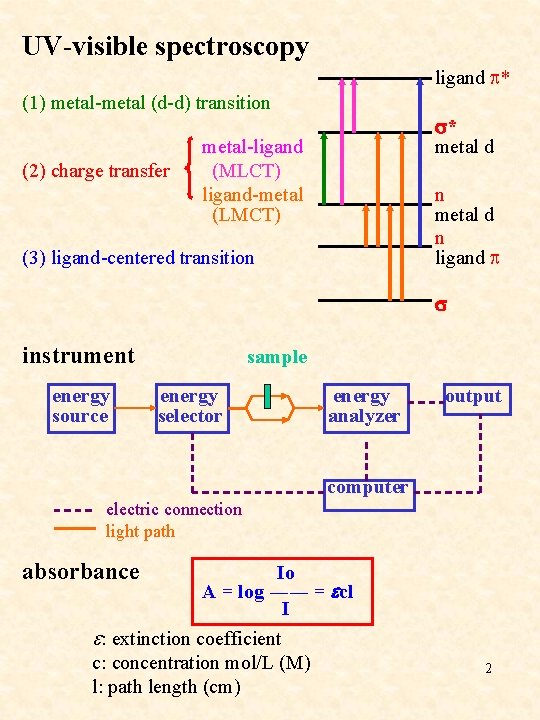
UV-visible spectroscopy ligand p* (1) metal-metal (d-d) transition (2) charge transfer metal-ligand (MLCT) ligand-metal (LMCT) (3) ligand-centered transition instrument s* metal d n ligand p s sample energy output source selector analyzer computer electric connection light path absorbance Io A = log ―― = ecl I e: extinction coefficient c: concentration mol/L (M) l: path length (cm) 2
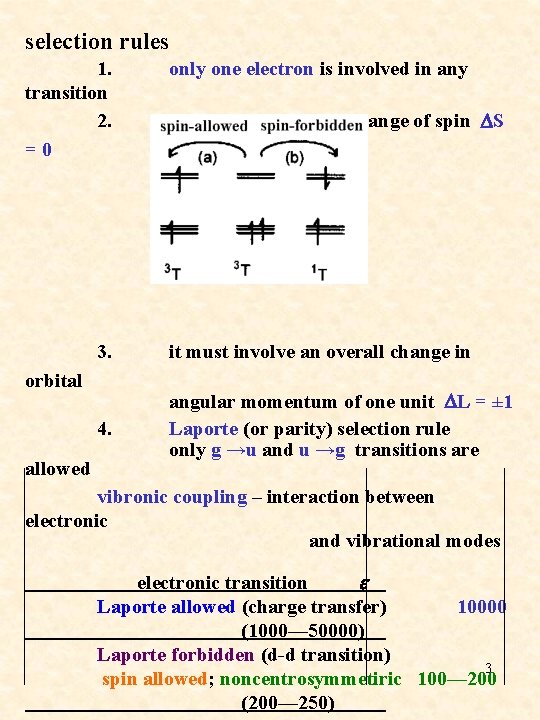
selection rules 1. transition 2. = 0 only one electron is involved in any 3. it must involve an overall change in 4. angular momentum of one unit DL = ± 1 Laporte (or parity) selection rule only g →u and u →g transitions are orbital allowed there must be no net change of spin DS vibronic coupling – interaction between electronic and vibrational modes electronic transition e Laporte allowed (charge transfer) 10000 (1000— 50000) Laporte forbidden (d-d transition) 3 spin allowed; noncentrosymmetiric 100— 200 (200— 250)
![[Co. Cl 4]2 - [Co(H 2 O)6]2+ [Mn(H 2 O)6]2+ 4 [Co. Cl 4]2 - [Co(H 2 O)6]2+ [Mn(H 2 O)6]2+ 4](http://slidetodoc.com/presentation_image/49609b366ee4a73b5d89c2322dc09f91/image-4.jpg)
[Co. Cl 4]2 - [Co(H 2 O)6]2+ [Mn(H 2 O)6]2+ 4
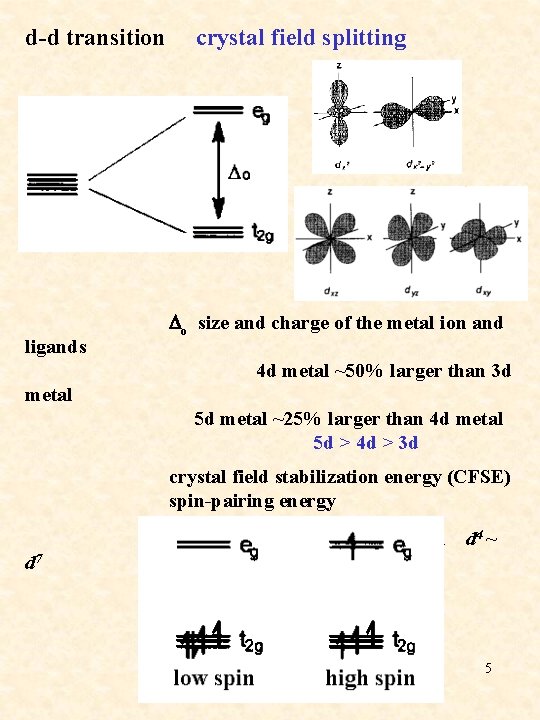
d-d transition crystal field splitting ligands Do size and charge of the metal ion and 4 d metal ~50% larger than 3 d metal 5 d metal ~25% larger than 4 d metal 5 d > 4 d > 3 d crystal field stabilization energy (CFSE) spin-pairing energy high-spin/low spin configuration d 4 ~ d 7 d 4 5
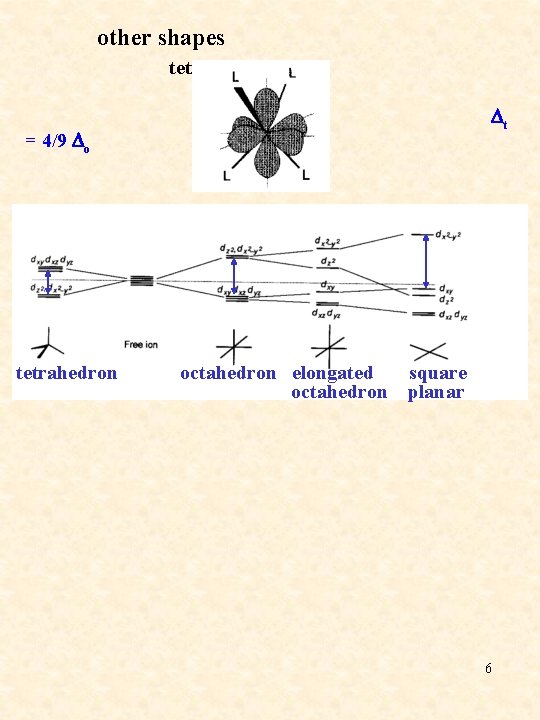
other shapes tetrahedral = 4/9 Do Dt tetrahedron octahedron elongated square octahedron planar 6
![d 1 [Ti(H 2 O)6]3+ hu = D o hole formalism d 2 possible d 1 [Ti(H 2 O)6]3+ hu = D o hole formalism d 2 possible](http://slidetodoc.com/presentation_image/49609b366ee4a73b5d89c2322dc09f91/image-7.jpg)
d 1 [Ti(H 2 O)6]3+ hu = D o hole formalism d 2 possible electron possible arrangements of electrons transitions 7
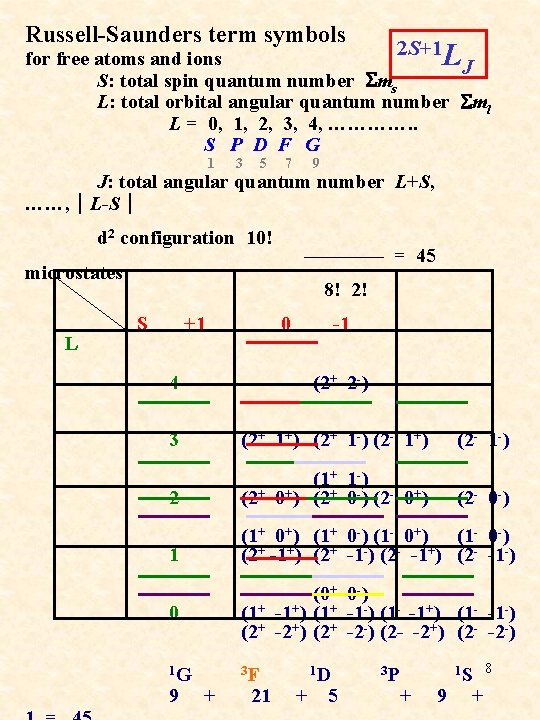
Russell-Saunders term symbols 2 S+1 L for free atoms and ions J S: total spin quantum number Sms L: total orbital angular quantum number Sml L = 0, 1, 2, 3, 4, …………. . S P D F G 1 3 5 7 9 J: total angular quantum number L+S, ……, │L-S│ d 2 configuration 10! ———— = 45 microstates 8! 2! L S +1 4 0 -1 (2+ 2 -) 3 (2+ 1+) (2+ 1 -) (2 - 1+) (2 - 1 -) 2 (1+ 1 -) (2+ 0+) (2+ 0 -) (2 - 0+) (2 - 0 -) 1 (1+ 0+) (1+ 0 -) (1 - 0+) (1 - 0 -) (2+ -1+) (2+ -1 -) (2 - -1+) (2 - -1 -) 0 (0+ 0 -) (1+ -1+) (1+ -1 -) (1 - -1+) (1 - -1 -) (2+ -2+) (2+ -2 -) (2 - -2+) (2 - -2 -) 1 G 3 F 1 D 3 P 1 S 8 9 + 21 + 5 + 9 +
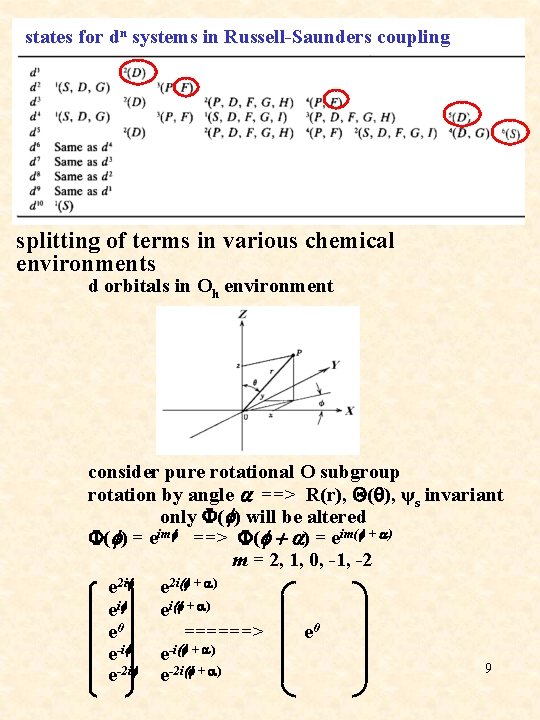
states for dn systems in Russell-Saunders coupling splitting of terms in various chemical environments d orbitals in Oh environment consider pure rotational O subgroup rotation by angle a ==> R(r), Q(q), ψs invariant only F(f) will be altered F(f) = eimf ==> F(f + a) = eim(f + a) m = 2, 1, 0, -1, -2 e 2 if e 2 i(f + a) eif ei(f + a) e 0 ======> e 0 e-if e-i(f + a) 9 e-2 if e-2 i(f + a)
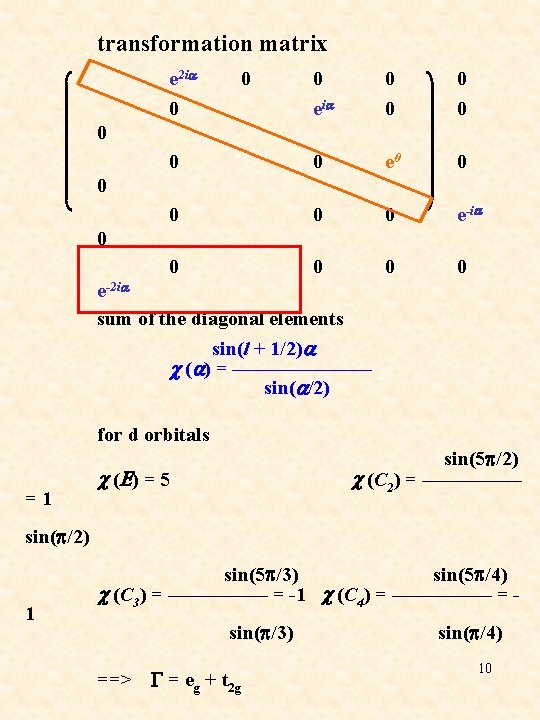
transformation matrix e 2 ia 0 0 0 eia 0 0 0 e-ia 0 0 0 0 e-2 ia sum of the diagonal elements sin(l + 1/2)a c (a) = ——————— sin(a/2) = 1 sin(p/2) for d orbitals sin(5 p/2) c (E) = 5 c (C 2) = ————— 1 sin(5 p/3) sin(5 p/4) c (C 3) = ————— = -1 c (C 4) = ————— = sin(p/3) sin(p/4) ==> G = eg + t 2 g 10
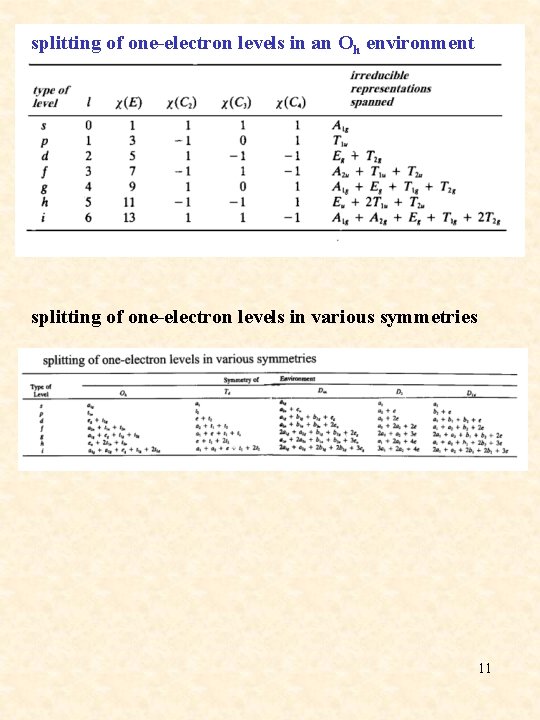
splitting of one-electron levels in an Oh environment splitting of one-electron levels in various symmetries 11
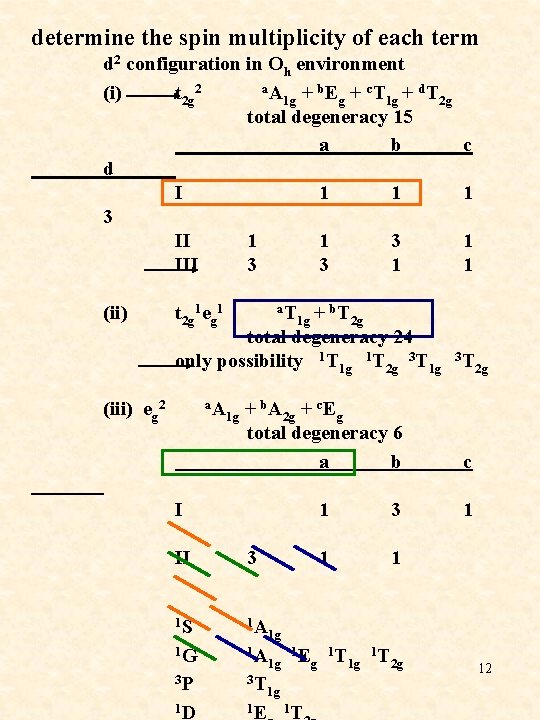
determine the spin multiplicity of each term d 2 configuration in Oh environment (i) t 2 g 2 a. A 1 g + b. Eg + c. T 1 g + d. T 2 g total degeneracy 15 a b d I 1 1 3 III 3 3 1 c 1 1 1 (ii) t 2 g 1 eg 1 a. T 1 g + b. T 2 g total degeneracy 24 only possibility 1 T 1 g 1 T 2 g 3 T 1 g 3 T 2 g (iii) eg 2 a. A 1 g + b. A 2 g + c. Eg total degeneracy 6 a b I II 3 1 S 1 A 1 3 1 1 c 1 1 G 3 P 1 D 1 g 1 A 1 1 g Eg T 1 g T 2 g 3 T 1 g 1 E 1 T 12

method of descending symmetry consider d 2 ion in Oh environment from correlation table for group Oh + ag + bg (i) t 2 g 2 A 1 g Eg T 1 g T 2 g lowering the symmetry to C 2 h t 2 g × t 2 g = possible spin multiplicity t 2 g ag 1 A 1 g 1 Eg 3 T 1 g 1 T 2 g 1 1 3 1 3 1 ˇ 3 1 1 3 corresponding 1 Ag Ag representations 1 Bg 3 Bg 1 Ag in C 2 h 3 Bg 1 Bg ag × ag Ag ====> 1 Ag ag × ag’ Ag ====> 1 Ag 3 Ag ag × bg Bg ====> 1 Bg 3 Bg ag’ × ag’ Ag ====> 1 Ag ag’ × bg Bg ====> 1 Bg 3 Bg 1 1 1 Ag 13

b 1 g 3 B (ii) eg 2 A 1 g A 2 g Eg lowering the symmetry to D 4 h eg a 1 g + a 1 g 2 A 1 g possible spin multiplicity 1 A 1 g a 1 gb 1 g B 1 g possible spin multiplicity 1 B 1 g b 1 g 2 A 1 g possible spin multiplicity 1 A 1 g ==> D 4 h Oh 1 A 1 1 g A 1 g 3 B 3 2 g A 1 g 1 A 1 B 1 1 g Eg (iii) t 2 g 1 eg 1 ? ? consider d 2 ion in Td environment from splitting of energy level in Td symmetry 3 F 3 A 3 T 2 1 D 1 1 E T 2 3 P 3 T 1 1 G 1 A 1 E 1 T 1 1 2 1 S 1 A 1 electron configurations e 2 A 1 A 2 E total degeneracy 6 et 2 T 1 T 2 total degeneracy 24 14 t 22 A 1 E T 1 T 2 total degeneracy 15

splitting of the terms for d 2 ion in several point groups 15
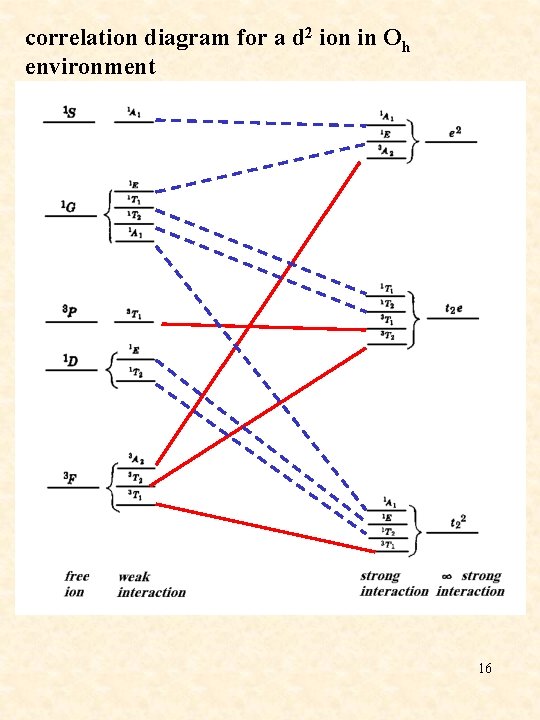
correlation diagram for a d 2 ion in Oh environment 16
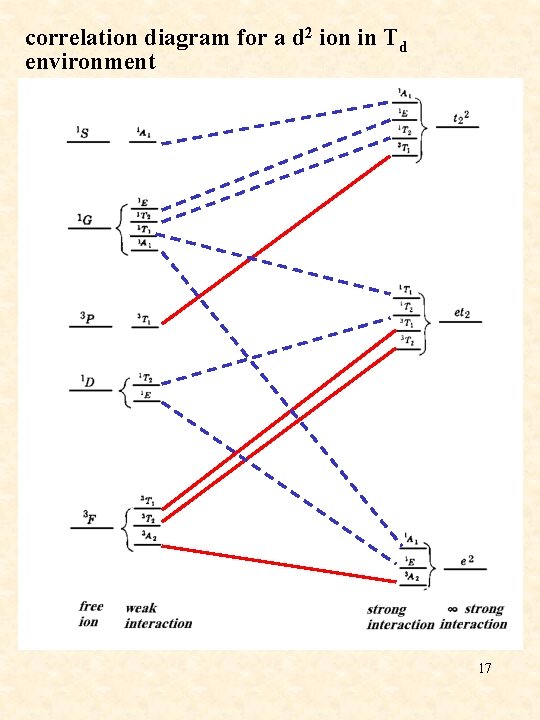
correlation diagram for a d 2 ion in Td environment 17
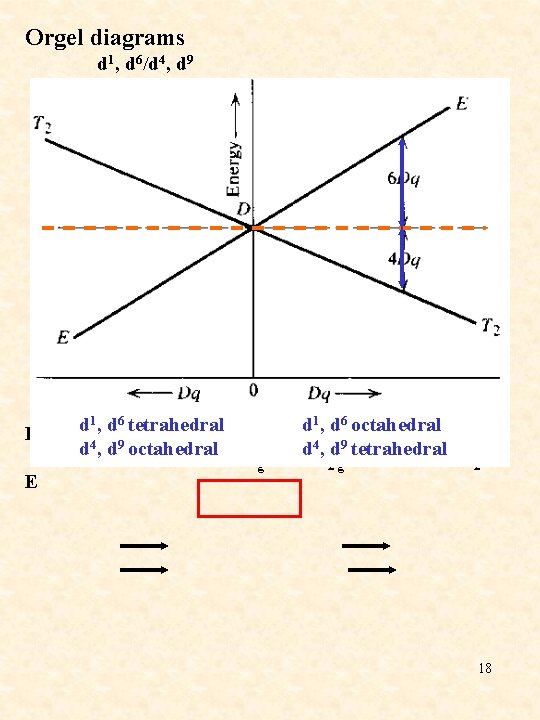
Orgel diagrams d 1, d 6/d 4, d 9 u = 10 Dq Eg E d 1, d 6 tetrahedral d 4, d 9 octahedral E T 2 g 2 6 1 d , d octahedral d 4, d 9 tetrahedral E T g 2 18
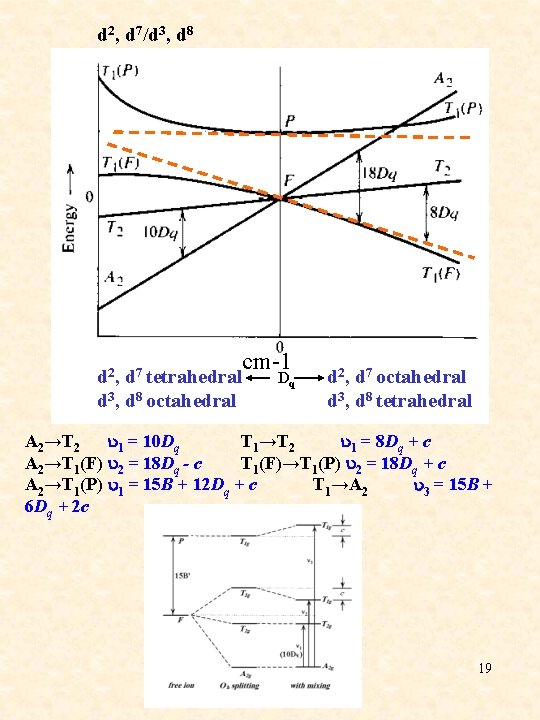
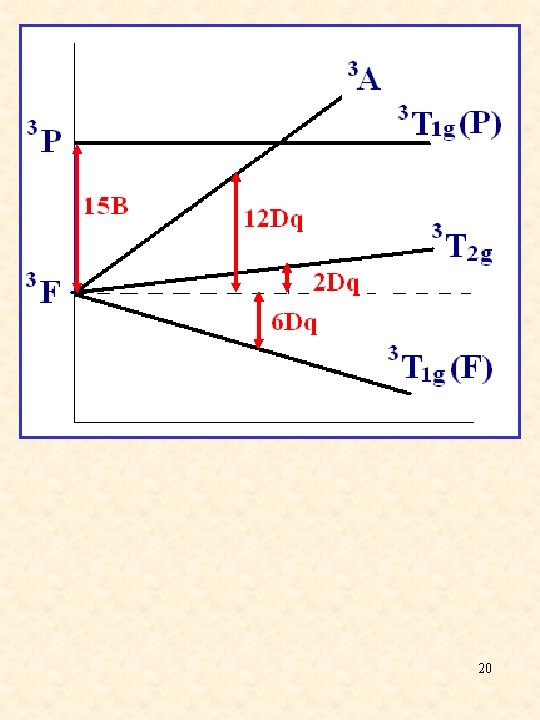
d 2, d 7/d 3, d 8 cm-1 d 2, d 7 tetrahedral Dq d 3, d 8 octahedral d 2, d 7 octahedral d 3, d 8 tetrahedral A 2→T 2 u 1 = 10 Dq T 1→T 2 u 1 = 8 Dq + c A 2→T 1(F) u 2 = 18 Dq - c T 1(F)→T 1(P) u 2 = 18 Dq + c A 2→T 1(P) u 1 = 15 B + 12 Dq + c T 1→A 2 u 3 = 15 B + 6 Dq + 2 c 19
20
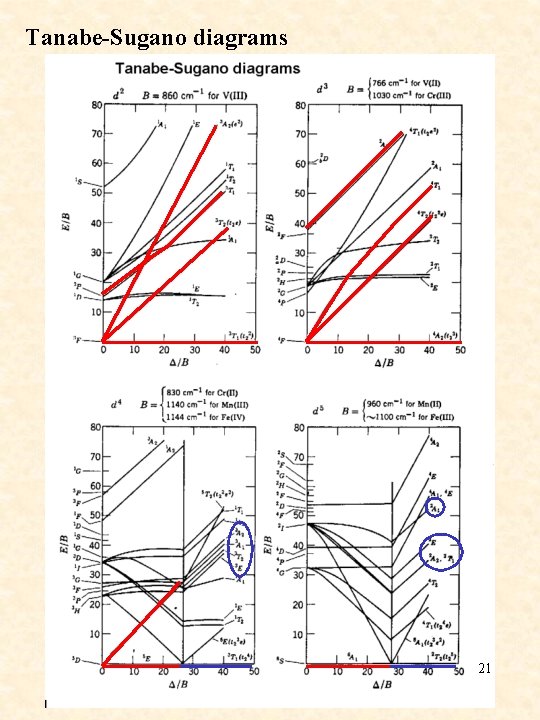
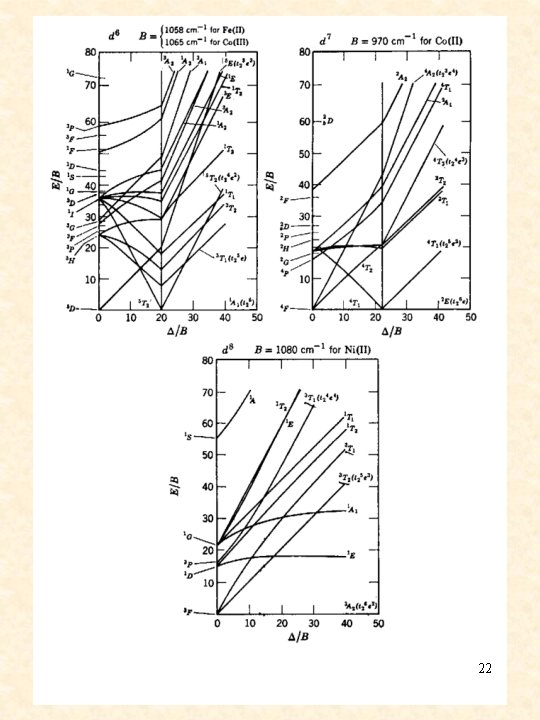
Tanabe-Sugano diagrams 21
22
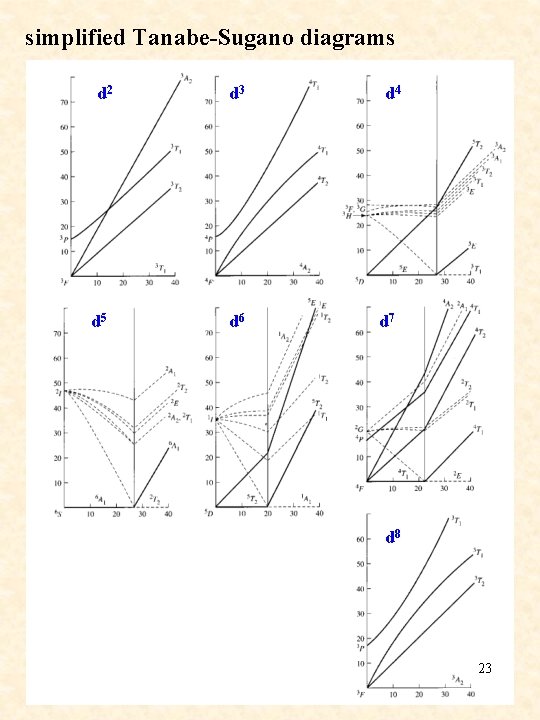
simplified Tanabe-Sugano diagrams d 2 d 5 d 3 d 6 d 4 d 7 d 8 23
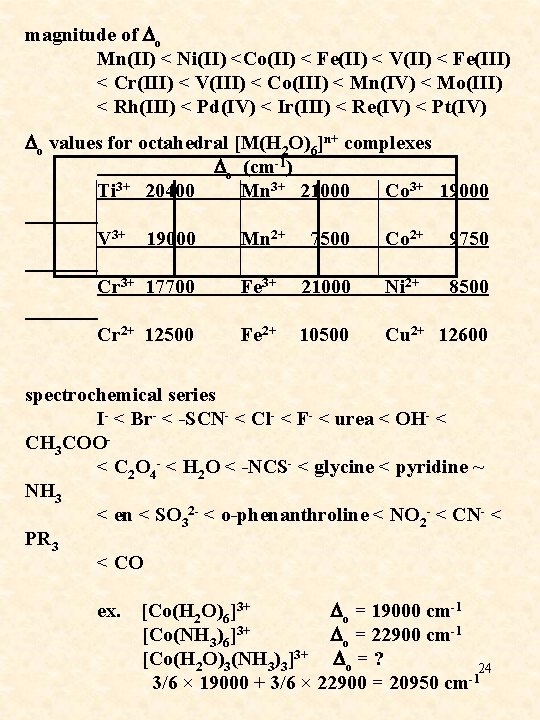
magnitude of Do Mn(II) < Ni(II) <Co(II) < Fe(II) < V(II) < Fe(III) < Cr(III) < V(III) < Co(III) < Mn(IV) < Mo(III) < Rh(III) < Pd(IV) < Ir(III) < Re(IV) < Pt(IV) Do values for octahedral [M(H 2 O)6]n+ complexes Do (cm-1) Ti 3+ 20400 Mn 3+ 21000 Co 3+ 19000 V 3+ 19000 Mn 2+ 7500 Co 2+ 9750 Cr 3+ 17700 Fe 3+ 21000 Ni 2+ 8500 Cr 2+ 12500 Fe 2+ 10500 Cu 2+ 12600 spectrochemical series I- < Br- < -SCN- < Cl- < F- < urea < OH- < CH 3 COO- < C 2 O 4 - < H 2 O < -NCS- < glycine < pyridine ~ NH 3 < en < SO 32 - < o-phenanthroline < NO 2 - < CN- < PR 3 < CO ex. [Co(H 2 O)6]3+ Do = 19000 cm-1 [Co(NH 3)6]3+ Do = 22900 cm-1 [Co(H 2 O)3(NH 3)3]3+ Do = ? 24 -1 3/6 × 19000 + 3/6 × 22900 = 20950 cm

Jørgensen prediction of 10 Dq and B 10 Dq = f · g (cm-1 × 10 -3) B = Bo (1 - h · k) Bo : free ion interelectronic repulsion parameter Jahn-Teller distortions distortion will occur whenever the resulting splitting energy levels yields additional stabilization __ dx -y __ dz eg __ __ dz __ dx -y or __ dxy __ __dxz, dyz t 2 g __ __ __ __ __ dxz, dyz __ dxy 2 2 2 25
![[M(H 2 O)6]n+ Ti 3+ (d 1) Mn 2+ (d 5) V 3+ (d [M(H 2 O)6]n+ Ti 3+ (d 1) Mn 2+ (d 5) V 3+ (d](http://slidetodoc.com/presentation_image/49609b366ee4a73b5d89c2322dc09f91/image-26.jpg)
[M(H 2 O)6]n+ Ti 3+ (d 1) Mn 2+ (d 5) V 3+ (d 2) Fe 2+ (d 6) Co 2+ (d 7) Cr 3+ (d 3) Ni 2+ (d 8) Cu 2+ (d 9) Cr 2+ (d 4) 26
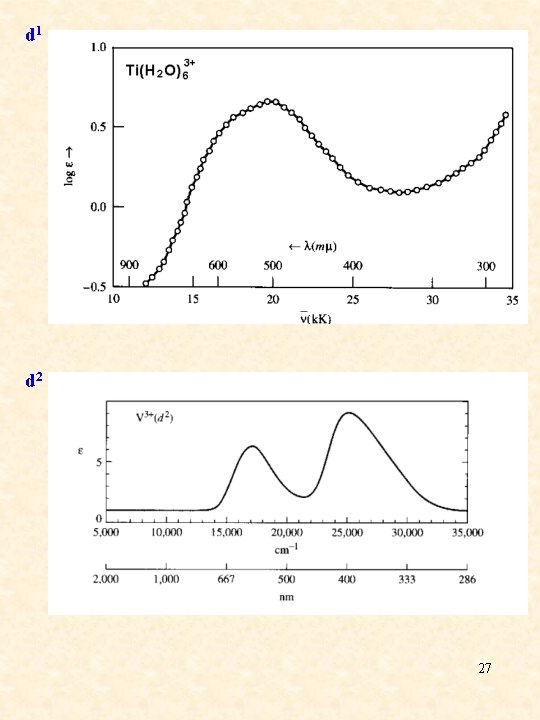
d 1 d 2 27
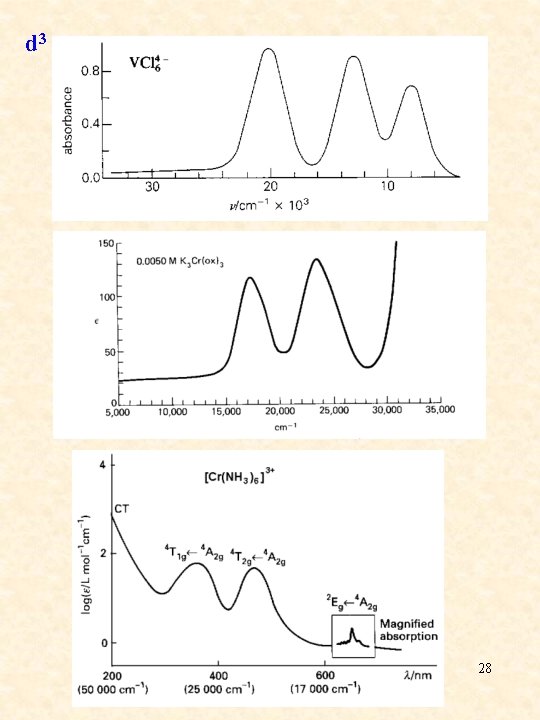
d 3 28
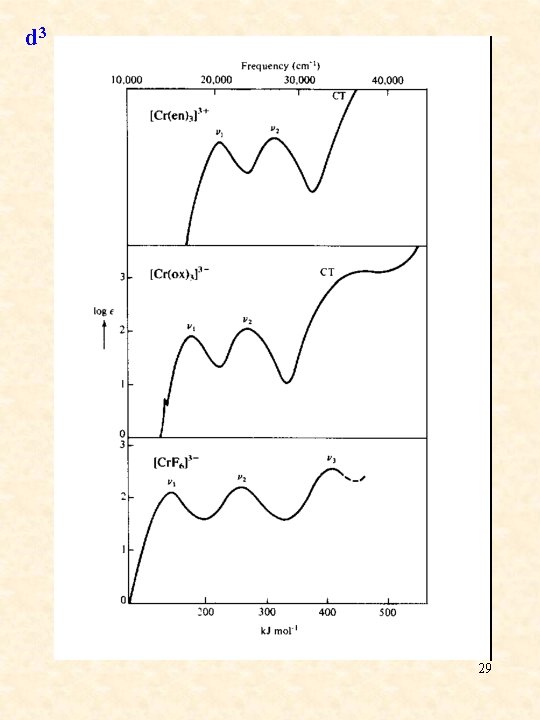
d 3 29
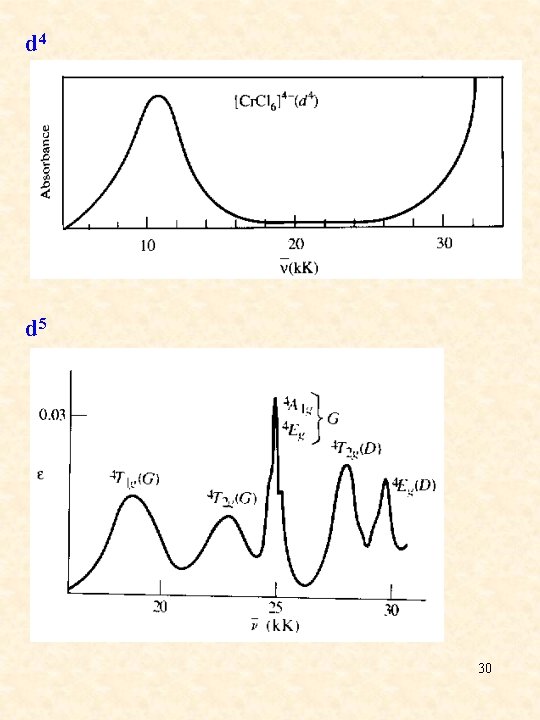
d 4 d 5 30
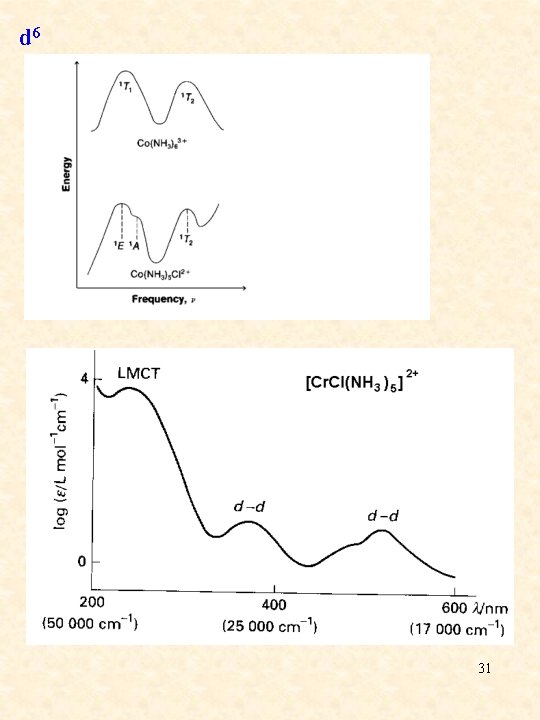
d 6 31
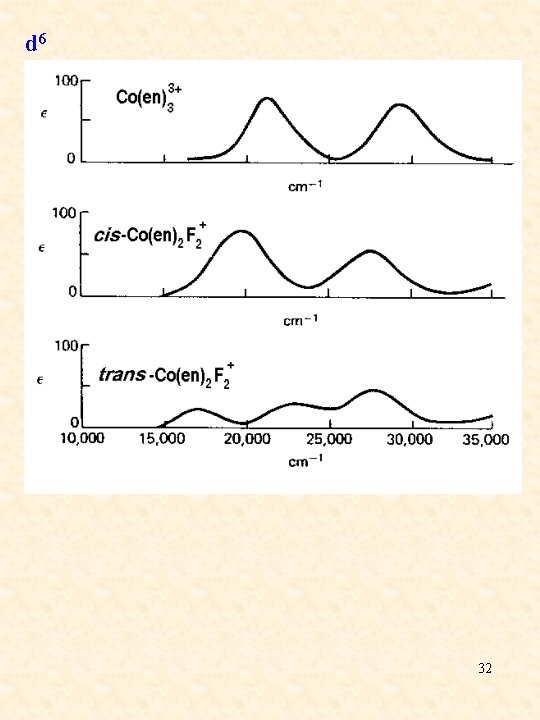
d 6 32
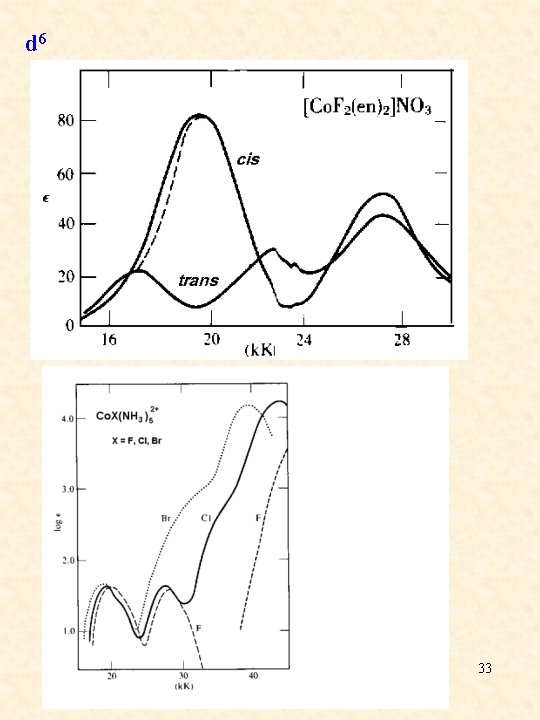
d 6 33
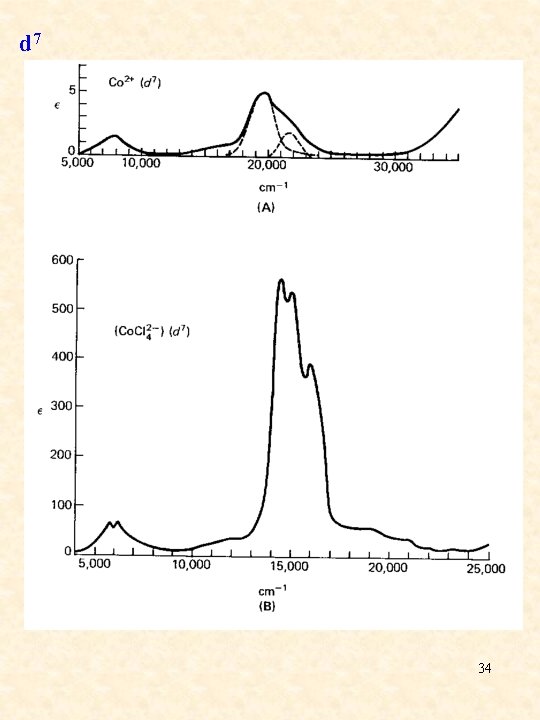
d 7 34
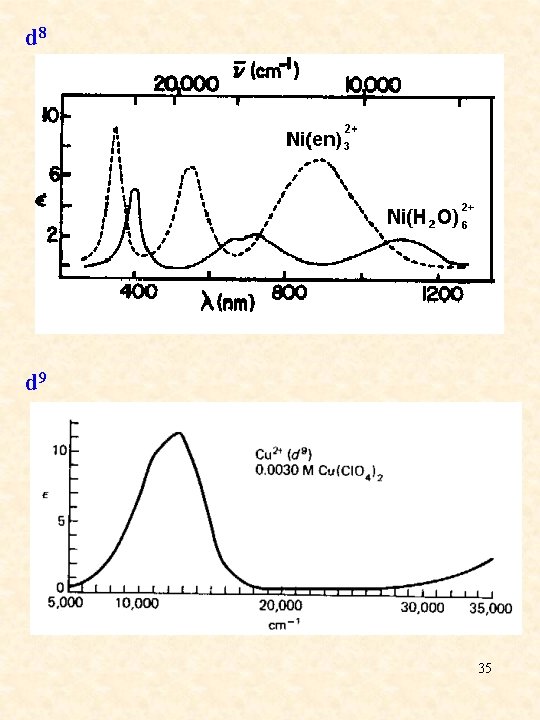
d 8 d 9 35
- Slides: 35