Electronic Structure Wave Nature of Light Electromagnetic Radiation






















- Slides: 33

Electronic Structure

Wave Nature of Light Electromagnetic Radiation Gamma Rays, Visible Light Moves Through Vacuum at 3. 00 x 108 m/s (c) Wavelength (λ, m) = distance between successive peaks or troughs Frequency (f, s-1) = how often a wave passes through a particular point c = f · λ, or v · λ

Wavelength Practice The brilliant red colors seen in fireworks are due to the emission of light with wavelengths around 650 nm when strontium salts such as Sr(NO 3)2 and Sr. CO 3 are heated. Calculate the frequency of red light of wavelength 6. 50 x 102 nm. A FM radio station broadcasts electromagnetic radiation at a frequency of 103. 4 MHz. Calculate the wavelength of this radiation. (1 MHz=106 s-1)

Quantize Energy and Photons Wave model explains much of the behavior of light but not all: Black body radiation – Emission of light from hot objects Photoelectric Effect – Emission of electrons from metal surfaces Emission Spectra – Emission of light from excited atoms

Black Body Radiation

Hot Objects and Quantization of Energy When objects are heated they emit light Red hot (cooler) → white hot (hotter) Light only emitted at certain wavelengths Max Planck declared that energy can only be emitted or absorbed in packets (quanta, photon) E= h · v h = Planck's Constant 6. 63 x 10 -34 J·s Energy emitted at whole number multiples of hv Think walking up and down stairs Why don't we notice this? Why does energy

Photoelectric Effect and Photons When light shines on an object, electrons are emitted Light has to have a specific energy, frequency and wavelength in order for e- to be emitted Photons are absorbed Too little energy – nothing happens Just right amount – electrons are emitted A little too much – electrons are emitted and excess used as kinetic energy

Calculation Practice The blue color of fireworks is often achieved by heating copper (I) chloride to about 1200°C. Then the compound emits blue light having a wavelength of 450 nm. Calculate the frequency and quantum of energy that is emitted at 4. 50 x 102 nm by Cu. Cl.

Spectra Radiation emitted from a source contains various λ's When separated into its different λ's a spectrum is formed Two types of spectrum Continuous Line

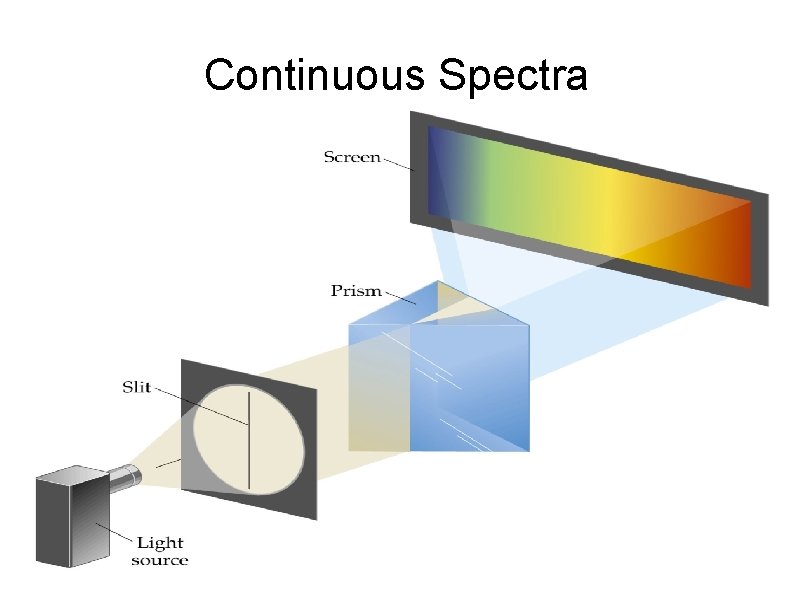
Continuous Spectra

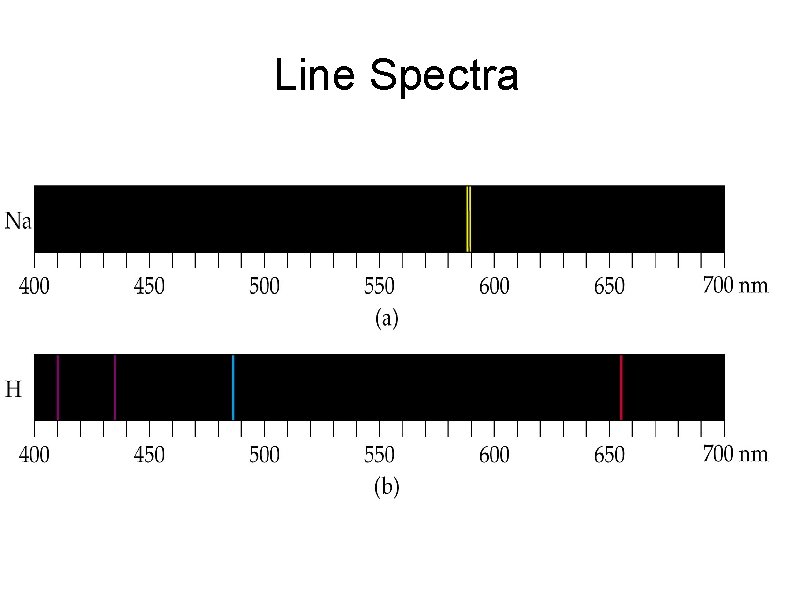
Line Spectra

Bohr Model Assumed electrons orbit nucleus in circular patterns To move between levels energy is absorbed or emitted Ground State – electron at lowest energy Excited State – electron is at higher energy state Bohr model only accurately explains hydrogen

Wave Behavior of Matter Lights is both a wave and a particle Louis de Broglie believed matter could have wave properties Applied idea to electrons λ=h/m·v Works for all matter so why don't we observed this in our everyday lives?

Uncertainty Principle If matter can act as a wave we should be able to calculate position and velocity Heisenberg determined that we cannot know both position and velocity of subatomic matter. Solving Schrödinger's equation gives use probabilities of location. The solutions correspond to the orbitals

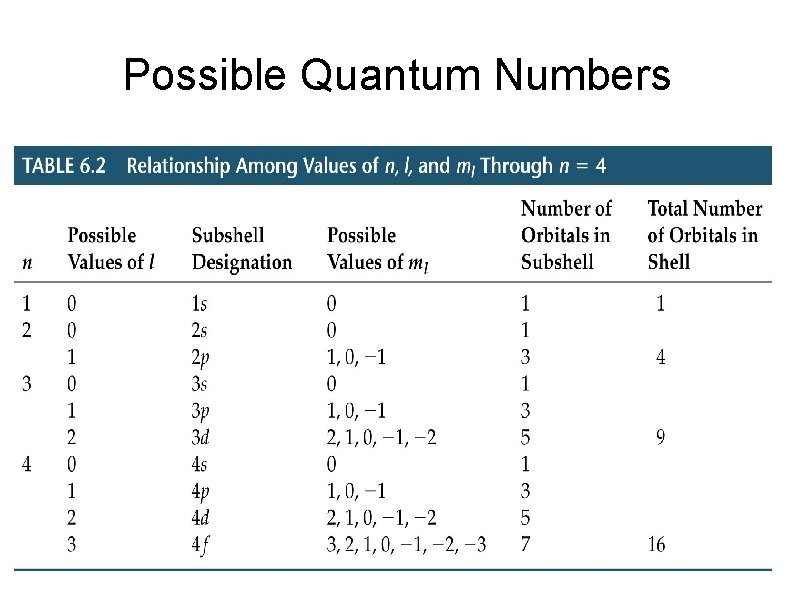
Quantum Numbers n – principle quantum number Whole numbers – 1, 2, 3, . . l – azimuthal quantum number From 0 to n-1 Determines shape of orbital 0 → s = shape 1 → p = principle 2 → d = diffuse 3 → f = fundamental

Quantum Numbers Cont. ml – magnetic quantum number Goes from -l to l including 0 Determines orientation of orbital ms – spin quantum number Two values +1/2 and -1/2 Determines spin of electron No two electrons can have the same four quantum numbers – Pauli Exclusion Principle

Possible Quantum Numbers

Quantum Numbers Example Which of the following sets of quantum numbers are not allowed? For each incorrect set, state why it is incorrect. n = 3, l = 3, ml = 0, ms = -1/2 n = 4, l = 3, ml = 2, ms = -1/2 n = 4, l = 1, ms = +1/2

Quantum Numbers Practice Which of the following sets of quantum numbers are not allowed? For each incorrect set, state why it is incorrect. n = 2, l = 1, ml = -1, ms = -1 n = 5, l = -4, ml = 2, ms = +1/2 n = 3, l =1, ml = 2, ms = -1/2

Quantum Number Practice What is the designation for the subshell with n = 5 and l = 1? How many orbitals are in this subshell? Indicate the values of ml for each of these orbitals.

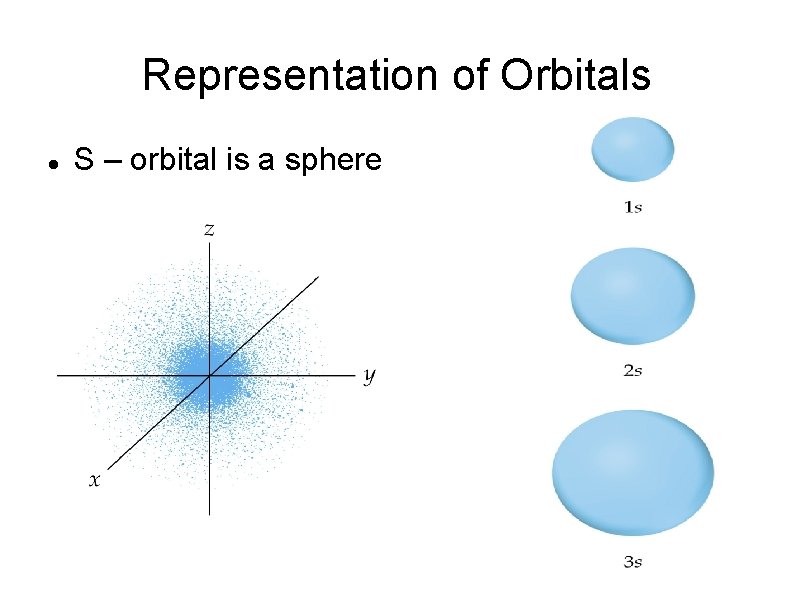
Representation of Orbitals S – orbital is a sphere

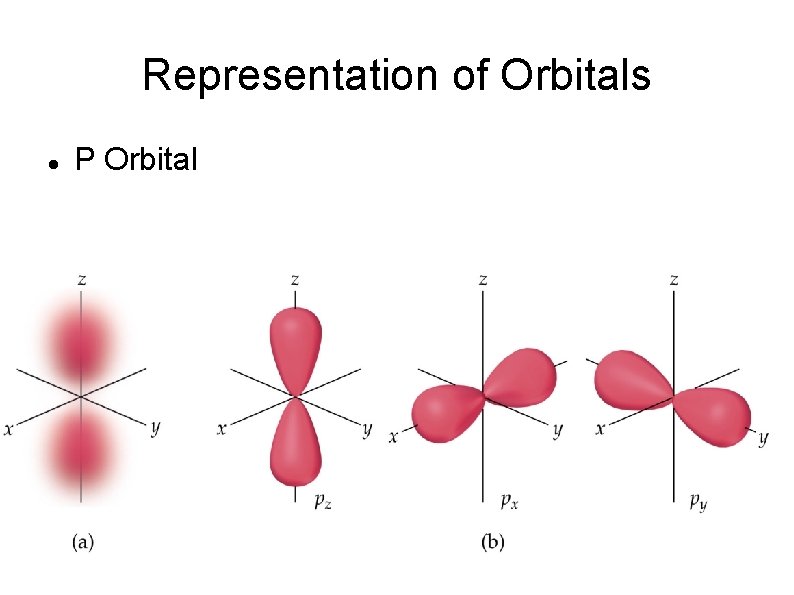
Representation of Orbitals P Orbital

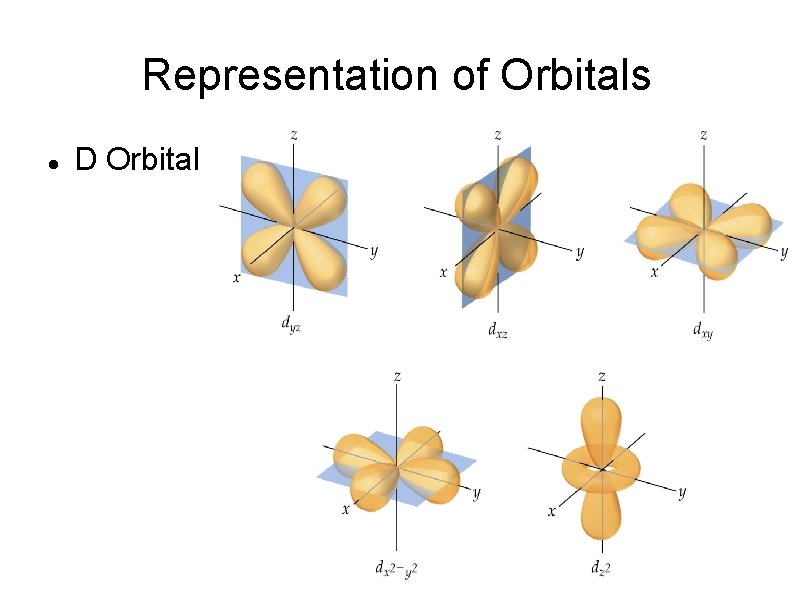
Representation of Orbitals D Orbital

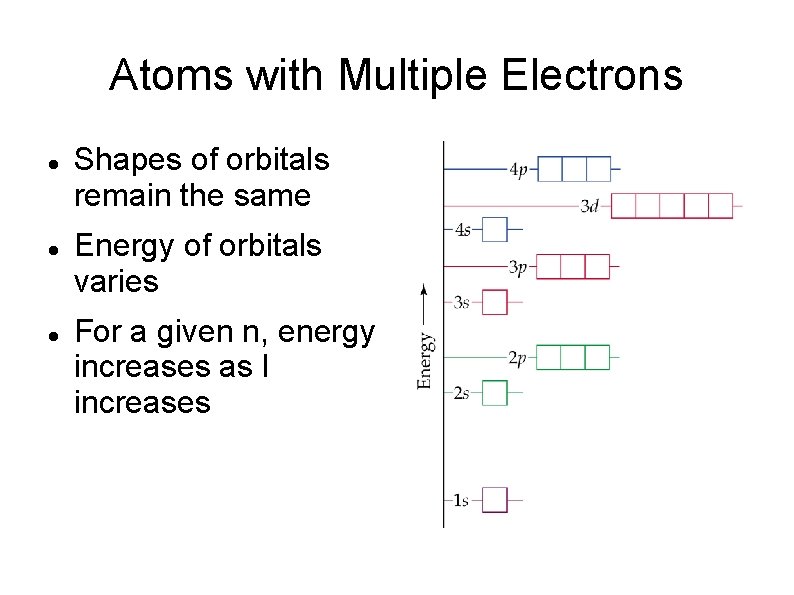
Atoms with Multiple Electrons Shapes of orbitals remain the same Energy of orbitals varies For a given n, energy increases as l increases

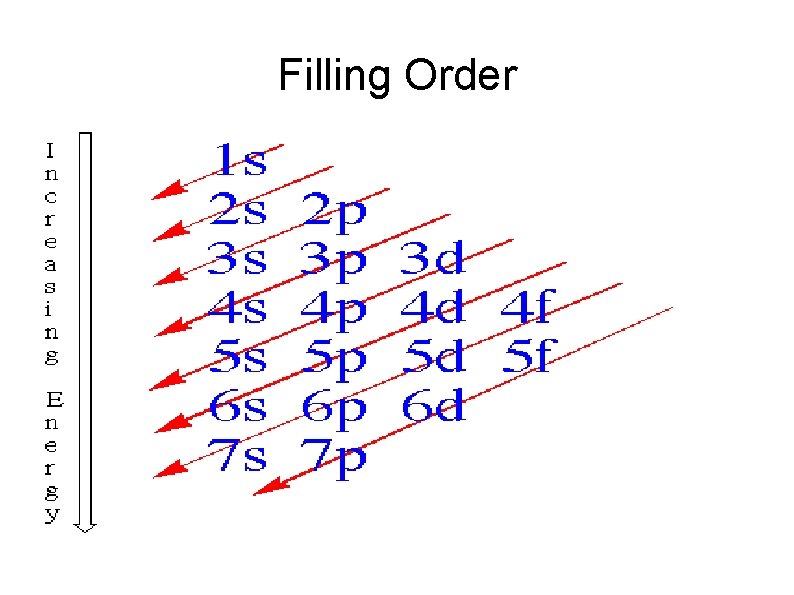
Electron Configuration Governed by three rules Pauli Exclusion Hund's Rule – for orbitals with same energy, lowest energy is attained when the number of electrons with the same spin is maximized AUFBAU – Energy shells are filled from lowest energy to highest energy

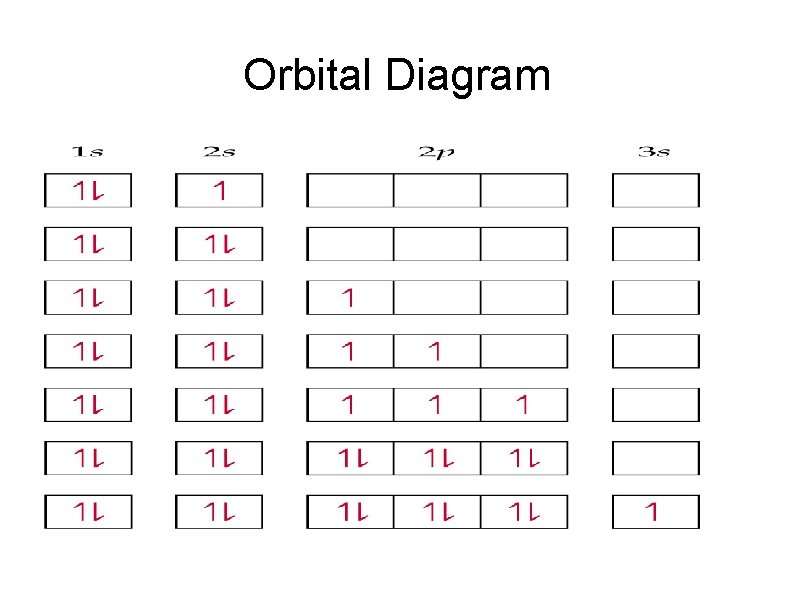
Orbital Diagram

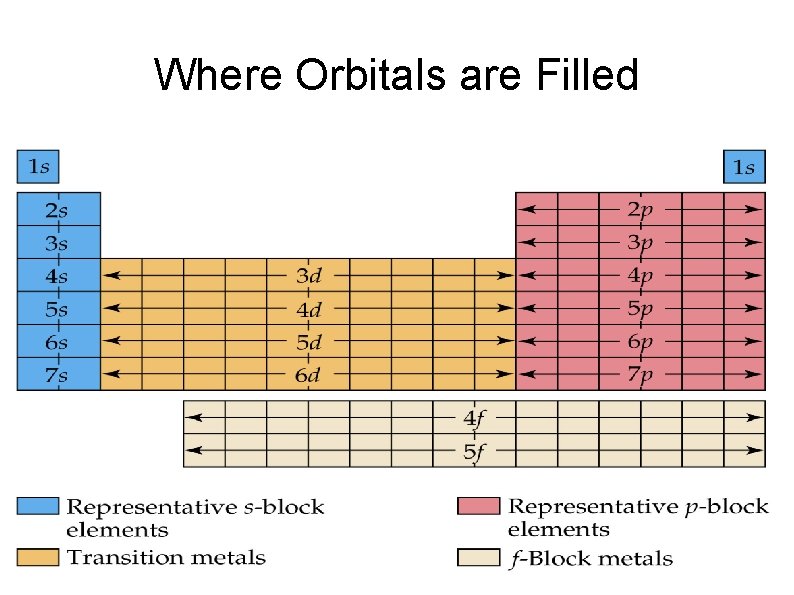
Where Orbitals are Filled

Filling Order

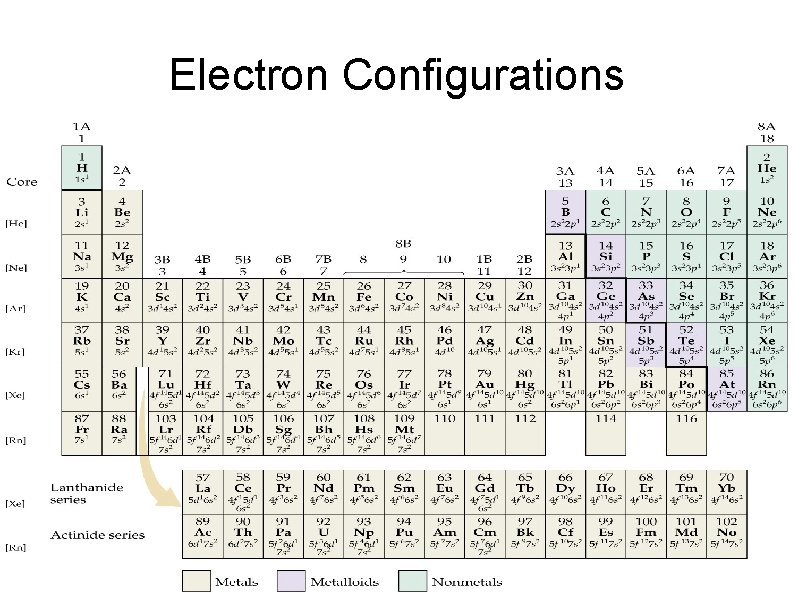
Electron Configuration Example Write the electron configuration for the following elements: Silicon Chromium Iodine

Electron Configuration Practice Write the electron configuration for the following elements: Copper Sulfur Tin

Exceptions to Filling Rules Chromium Copper Silver Molybdenum

Homework 2, 6, 10, 14, 20, 22, 26, 46, 52, 54, 60, 68

Electron Configurations