Electronic Structure of Alkoxy Radical Isomers from Anion






- Slides: 43

Electronic Structure of Alkoxy Radical Isomers from Anion PEI Spectroscopy Kellyn M. Patros, Marissa A. Dobulis, Jennifer E. Mann, Caroline Chick Jarrold ISMS June 20, 2018

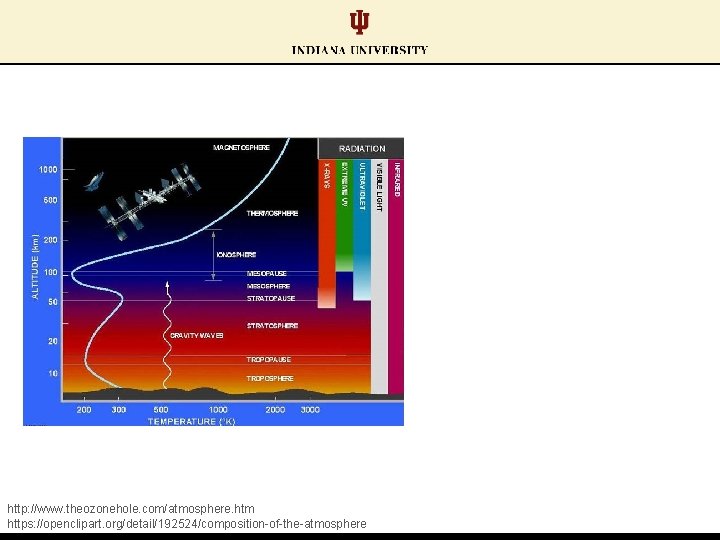
http: //www. theozonehole. com/atmosphere. htm https: //openclipart. org/detail/192524/composition-of-the-atmosphere

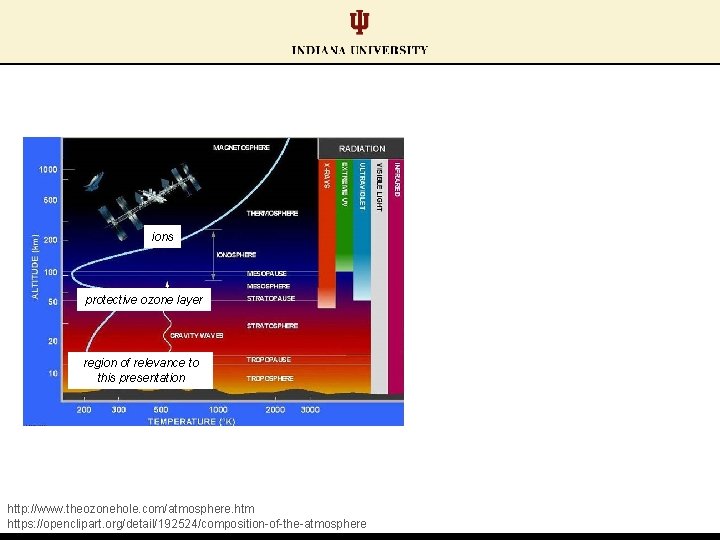
ions protective ozone layer region of relevance to this presentation http: //www. theozonehole. com/atmosphere. htm https: //openclipart. org/detail/192524/composition-of-the-atmosphere

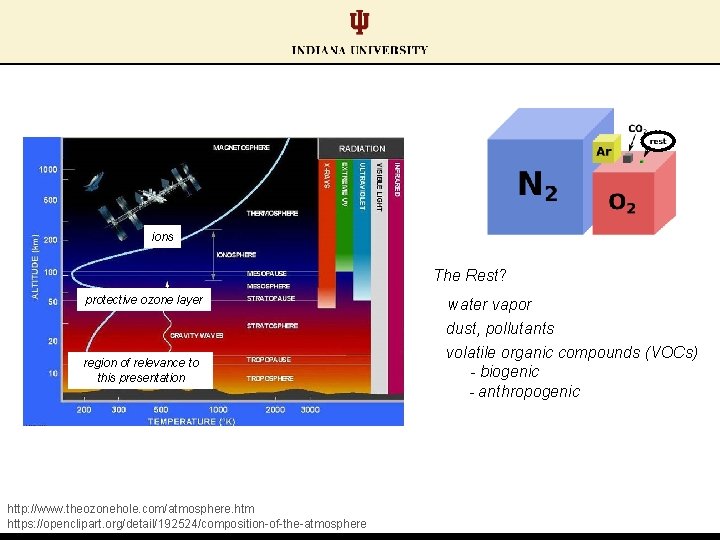
ions The Rest? protective ozone layer water vapor dust, pollutants region of relevance to this presentation http: //www. theozonehole. com/atmosphere. htm https: //openclipart. org/detail/192524/composition-of-the-atmosphere volatile organic compounds (VOCs) - biogenic - anthropogenic

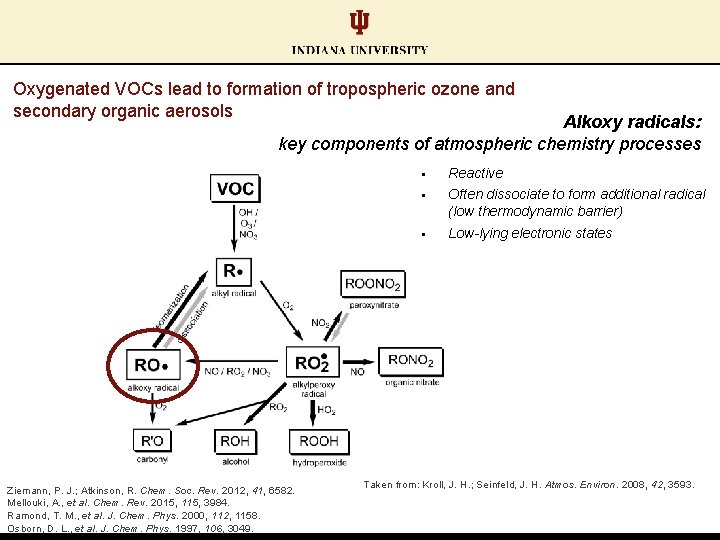
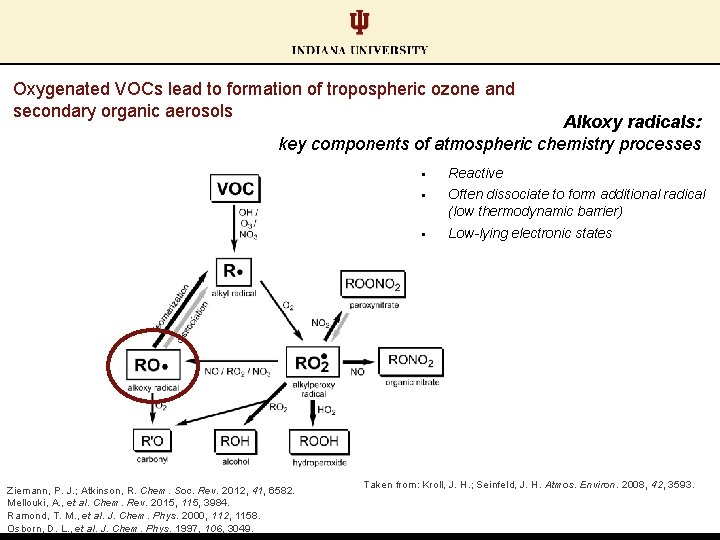
Oxygenated VOCs lead to formation of tropospheric ozone and secondary organic aerosols

Oxygenated VOCs lead to formation of tropospheric ozone and secondary organic aerosols Alkoxy radicals: key components of atmospheric chemistry processes Ziemann, P. J. ; Atkinson, R. Chem. Soc. Rev. 2012, 41, 6582. Mellouki, A. , et al. Chem. Rev. 2015, 115, 3984. Ramond, T. M. , et al. J. Chem. Phys. 2000, 112, 1158. Osborn, D. L. , et al. J. Chem. Phys. 1997, 106, 3049. § Reactive § Often dissociate to form additional radical (low thermodynamic barrier) § Low-lying electronic states Taken from: Kroll, J. H. ; Seinfeld, J. H. Atmos. Environ. 2008, 42, 3593.

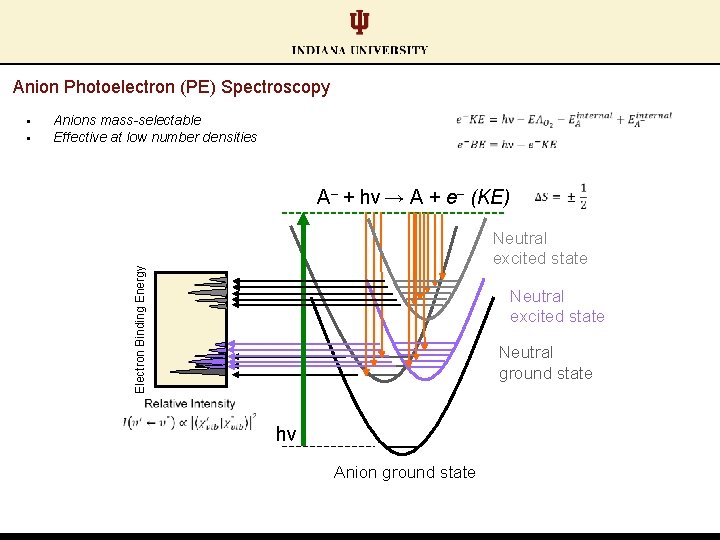
Anion Photoelectron (PE) Spectroscopy § Anions mass-selectable Effective at low number densities A‒ + hν → A + e‒ (KE) Neutral excited state Electron Binding Energy § Neutral excited state Neutral ground state hν Anion ground state

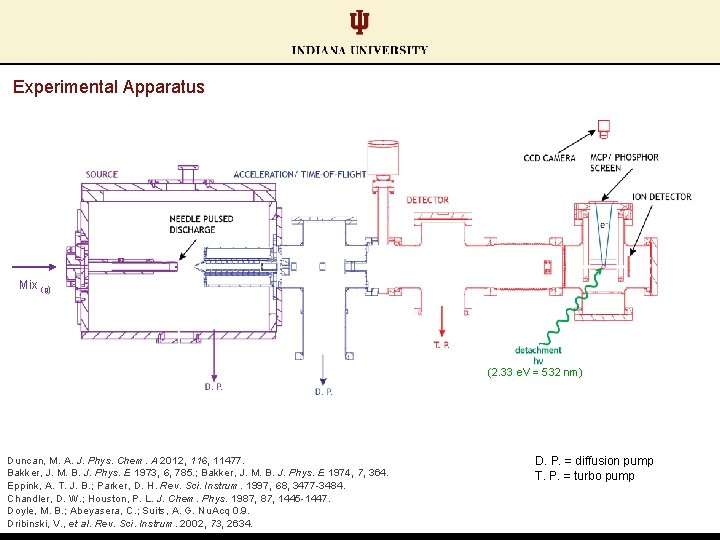
Experimental Apparatus Mix (g) (2. 33 e. V = 532 nm) Duncan, M. A. J. Phys. Chem. A 2012, 116, 11477. Bakker, J. M. B. J. Phys. E 1973, 6, 785. ; Bakker, J. M. B. J. Phys. E 1974, 7, 364. Eppink, A. T. J. B. ; Parker, D. H. Rev. Sci. Instrum. 1997, 68, 3477 -3484. Chandler, D. W. ; Houston, P. L. J. Chem. Phys. 1987, 1445 -1447. Doyle, M. B. ; Abeyasera, C. ; Suits, A. G. Nu. Acq 0. 9. Dribinski, V. , et al. Rev. Sci. Instrum. 2002, 73, 2634. D. P. = diffusion pump T. P. = turbo pump

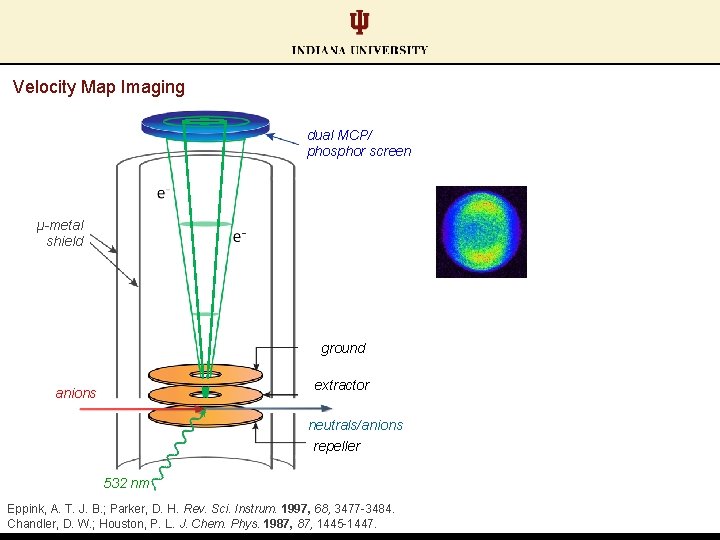
Velocity Map Imaging dual MCP/ phosphor screen μ-metal shield ground extractor anions neutrals/anions repeller 532 nm Eppink, A. T. J. B. ; Parker, D. H. Rev. Sci. Instrum. 1997, 68, 3477 -3484. Chandler, D. W. ; Houston, P. L. J. Chem. Phys. 1987, 1445 -1447.

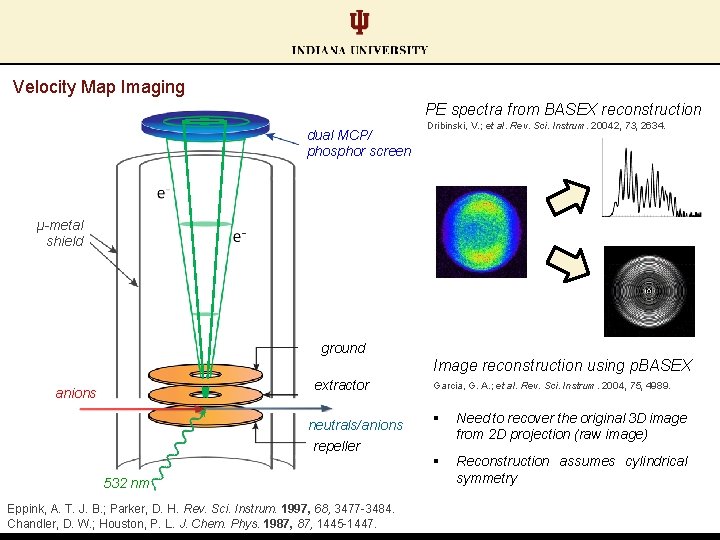
Velocity Map Imaging PE spectra from BASEX reconstruction dual MCP/ phosphor screen Dribinski, V. ; et al. Rev. Sci. Instrum. 20042, 73, 2634. μ-metal shield ground Image reconstruction using p. BASEX extractor anions neutrals/anions repeller 532 nm Eppink, A. T. J. B. ; Parker, D. H. Rev. Sci. Instrum. 1997, 68, 3477 -3484. Chandler, D. W. ; Houston, P. L. J. Chem. Phys. 1987, 1445 -1447. Garcia, G. A. ; et al. Rev. Sci. Instrum. 2004, 75, 4989. § Need to recover the original 3 D image from 2 D projection (raw image) § Reconstruction assumes cylindrical symmetry

Oxygenated VOCs lead to formation of tropospheric ozone and secondary organic aerosols Alkoxy radicals: key components of atmospheric chemistry processes Ziemann, P. J. ; Atkinson, R. Chem. Soc. Rev. 2012, 41, 6582. Mellouki, A. , et al. Chem. Rev. 2015, 115, 3984. Ramond, T. M. , et al. J. Chem. Phys. 2000, 112, 1158. Osborn, D. L. , et al. J. Chem. Phys. 1997, 106, 3049. § Reactive § Often dissociate to form additional radical (low thermodynamic barrier) § Low-lying electronic states Taken from: Kroll, J. H. ; Seinfeld, J. H. Atmos. Environ. 2008, 42, 3593.

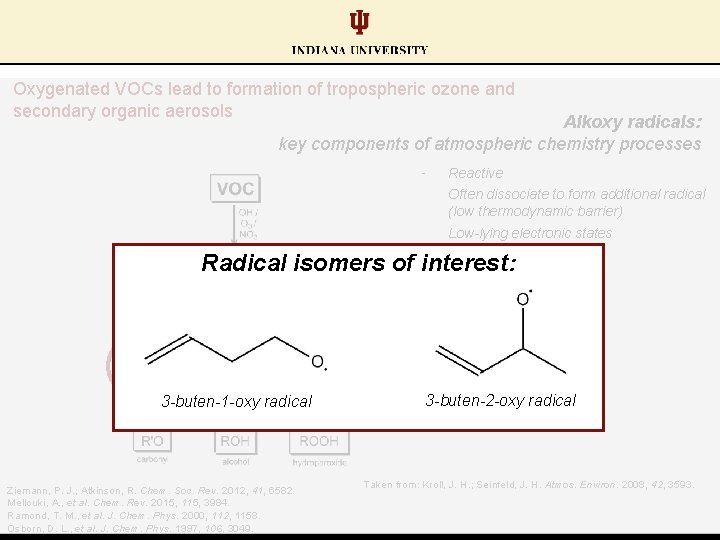
Oxygenated VOCs lead to formation of tropospheric ozone and secondary organic aerosols Alkoxy radicals: key components of atmospheric chemistry processes § Reactive § Often dissociate to form additional radical (low thermodynamic barrier) § Low-lying electronic states Radical isomers of interest: 3 -buten-1 -oxy radical Ziemann, P. J. ; Atkinson, R. Chem. Soc. Rev. 2012, 41, 6582. Mellouki, A. , et al. Chem. Rev. 2015, 115, 3984. Ramond, T. M. , et al. J. Chem. Phys. 2000, 112, 1158. Osborn, D. L. , et al. J. Chem. Phys. 1997, 106, 3049. 3 -buten-2 -oxy radical Taken from: Kroll, J. H. ; Seinfeld, J. H. Atmos. Environ. 2008, 42, 3593.

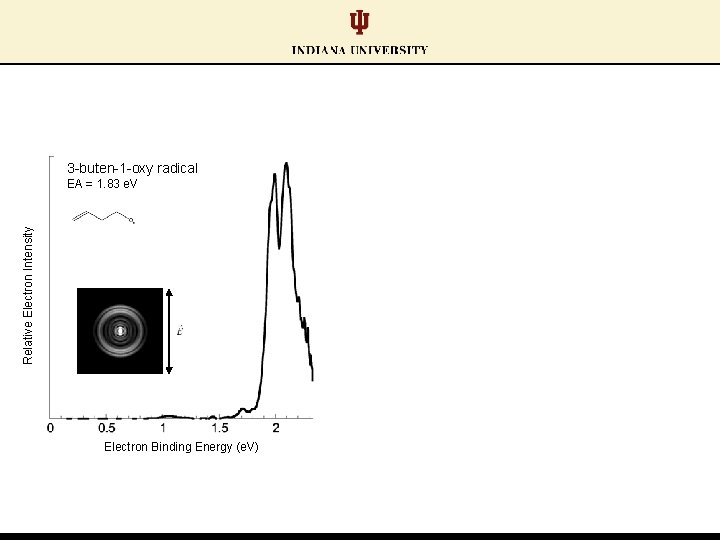
3 -buten-1 -oxy radical Relative Electron Intensity EA = 1. 83 e. V Electron Binding Energy (e. V)

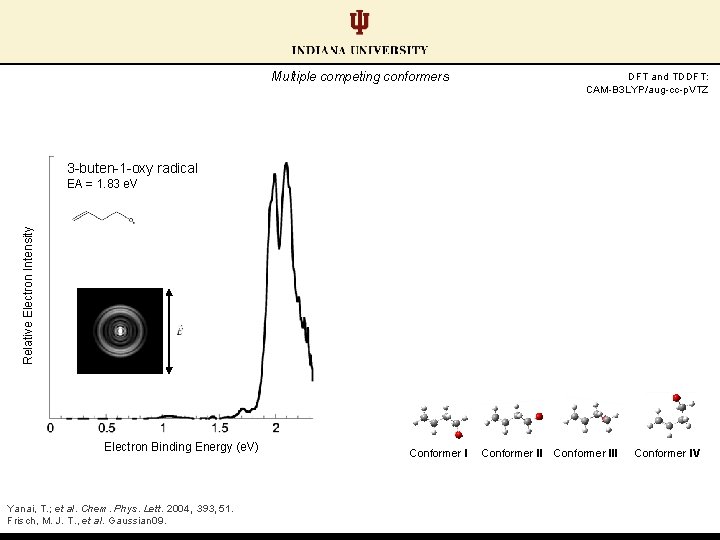
Multiple competing conformers DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 3 -buten-1 -oxy radical Relative Electron Intensity EA = 1. 83 e. V Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer III Conformer IV

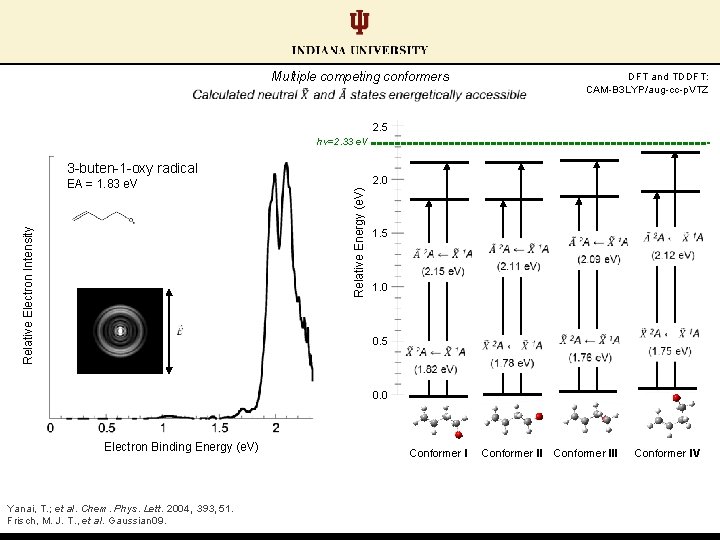
Multiple competing conformers DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer III Conformer IV

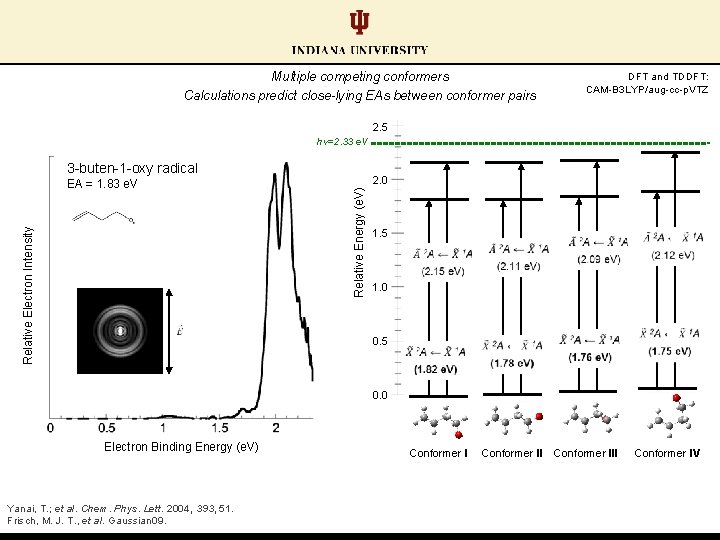
Multiple competing conformers Calculations predict close-lying EAs between conformer pairs DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer III Conformer IV

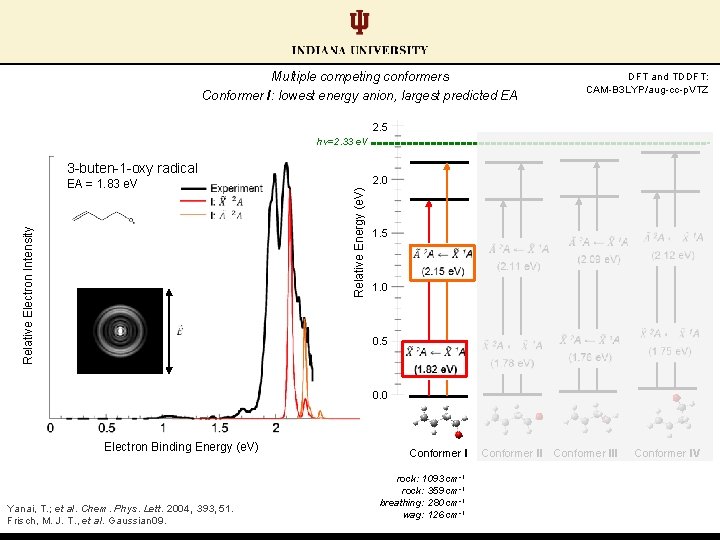
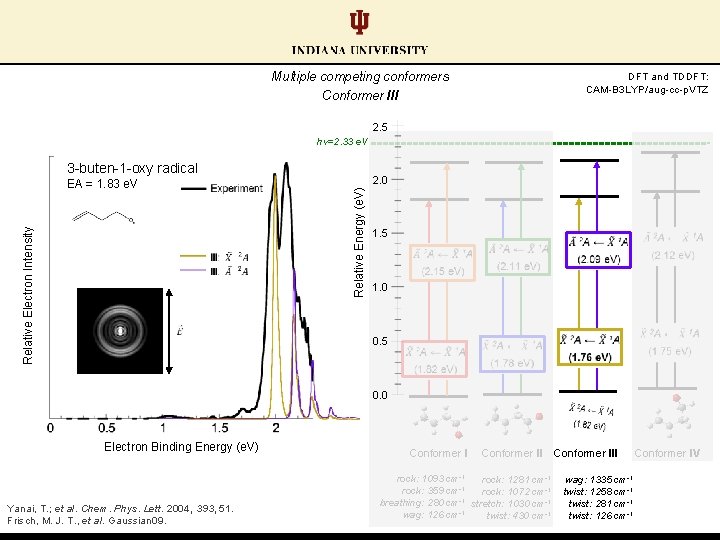
Multiple competing conformers Conformer I: lowest energy anion, largest predicted EA DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer I rock: 1093 cm-1 rock: 359 cm-1 breathing: 280 cm-1 wag: 126 cm-1 Conformer III Conformer IV

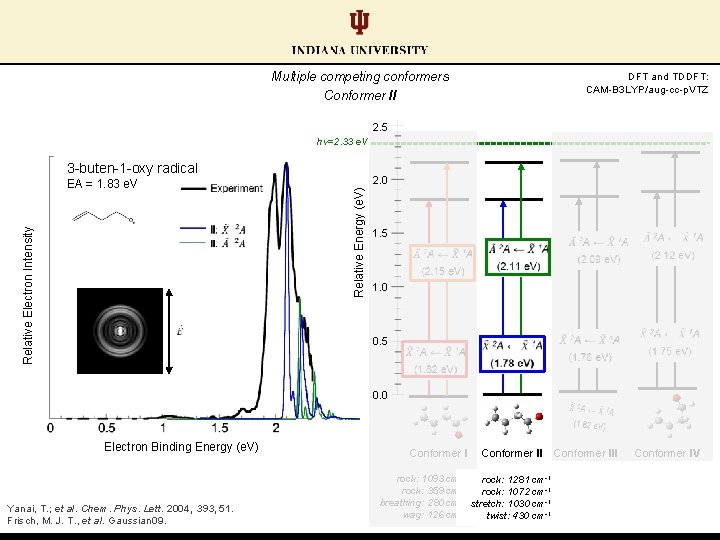
Multiple competing conformers Conformer II DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II rock: 1093 cm-1 rock: 1281 cm-1 -1 rock: 359 cm rock: 1072 cm-1 -1 breathing: 280 cm stretch: 1030 cm-1 wag: 126 cm-1 twist: 430 cm-1 Conformer III Conformer IV

Multiple competing conformers Conformer III DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II rock: 1093 cm-1 rock: 1281 cm-1 -1 rock: 359 cm rock: 1072 cm-1 -1 breathing: 280 cm stretch: 1030 cm-1 wag: 126 cm-1 twist: 430 cm-1 Conformer III wag: 1335 cm-1 twist: 1258 cm-1 twist: 281 cm-1 twist: 126 cm-1 Conformer IV

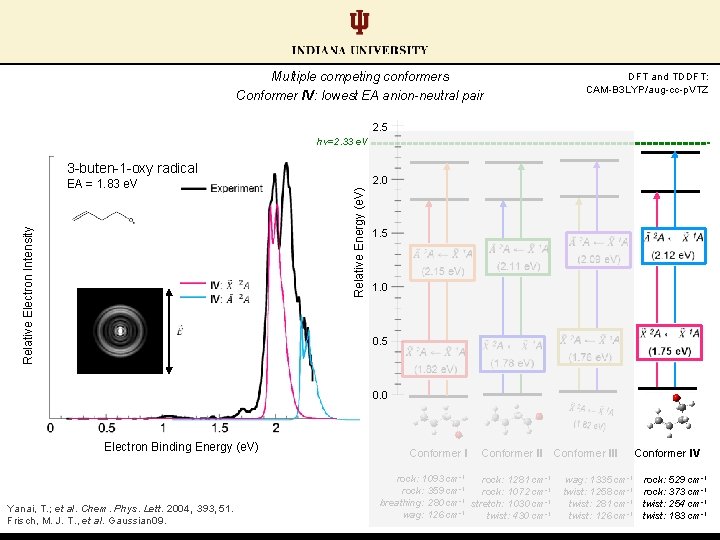
Multiple competing conformers Conformer IV: lowest EA anion-neutral pair DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II rock: 1093 cm-1 rock: 1281 cm-1 -1 rock: 359 cm rock: 1072 cm-1 -1 breathing: 280 cm stretch: 1030 cm-1 wag: 126 cm-1 twist: 430 cm-1 Conformer III Conformer IV wag: 1335 cm-1 rock: 529 cm-1 twist: 1258 cm-1 rock: 373 cm-1 twist: 281 cm-1 twist: 254 cm-1 twist: 126 cm-1 twist: 183 cm-1

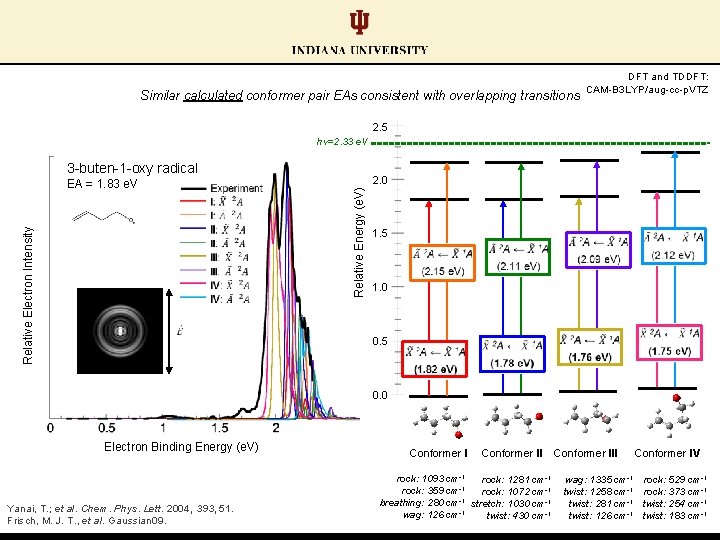
Similar calculated conformer pair EAs consistent with overlapping transitions DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-1 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 83 e. V 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II rock: 1093 cm-1 rock: 1281 cm-1 -1 rock: 359 cm rock: 1072 cm-1 -1 breathing: 280 cm stretch: 1030 cm-1 wag: 126 cm-1 twist: 430 cm-1 Conformer III Conformer IV wag: 1335 cm-1 rock: 529 cm-1 twist: 1258 cm-1 rock: 373 cm-1 twist: 281 cm-1 twist: 254 cm-1 twist: 126 cm-1 twist: 183 cm-1

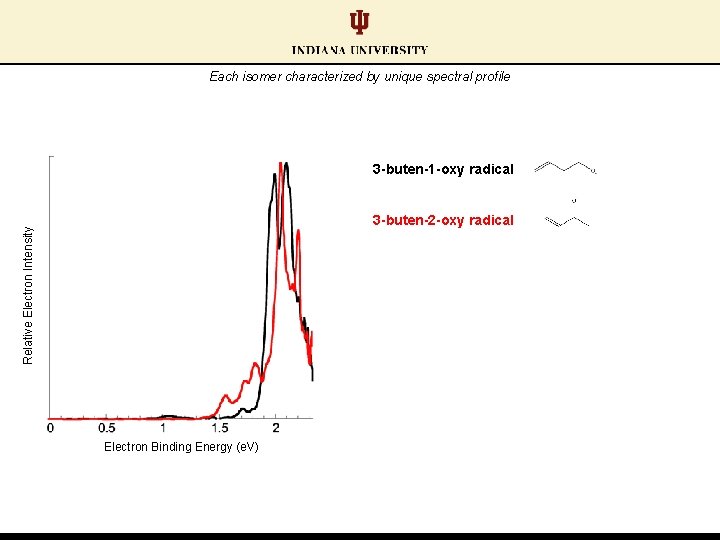
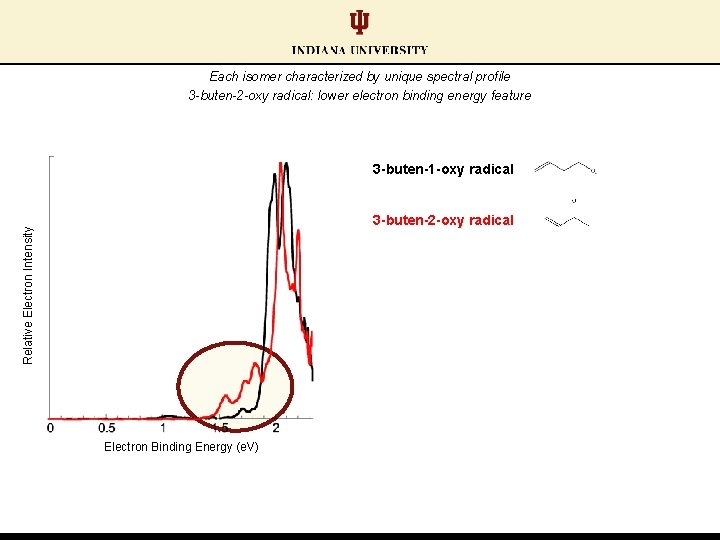
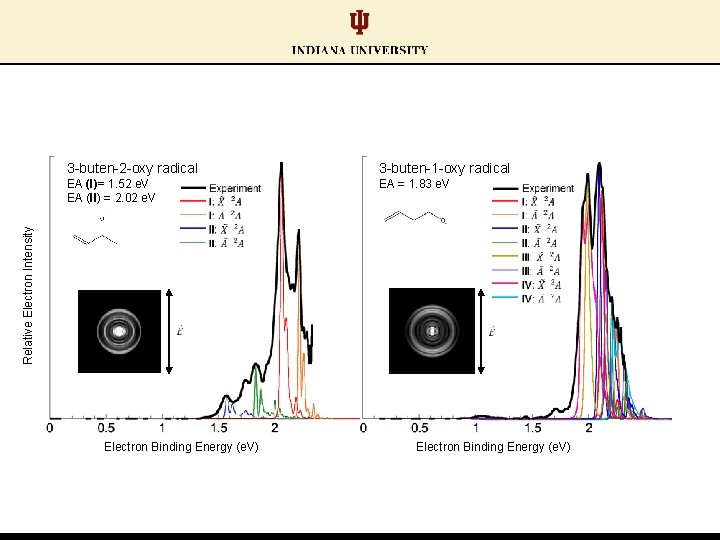
Each isomer characterized by unique spectral profile 3 -buten-1 -oxy radical Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

Each isomer characterized by unique spectral profile 3 -buten-2 -oxy radical: lower electron binding energy feature 3 -buten-1 -oxy radical Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

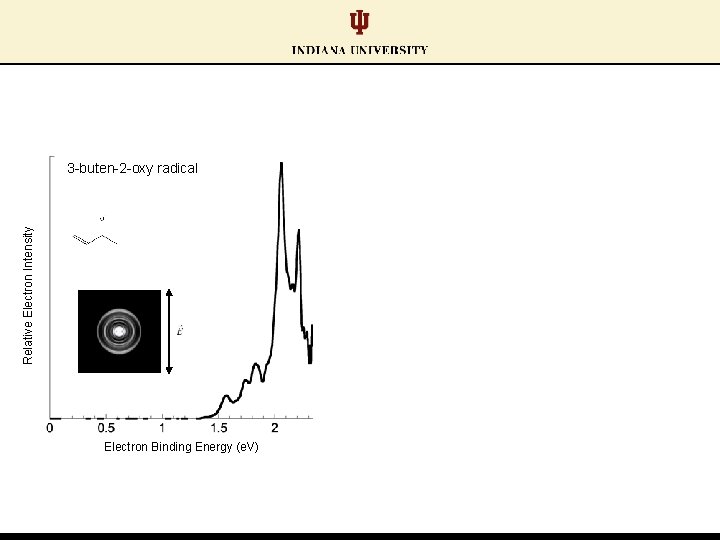
Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

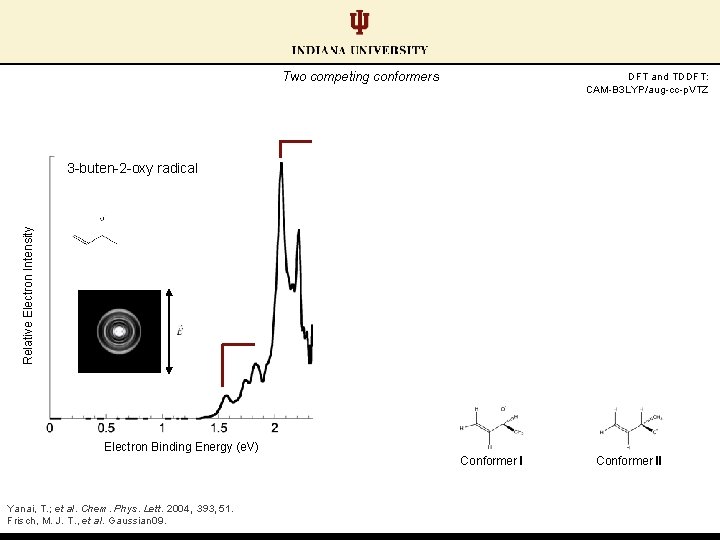
Two competing conformers DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II

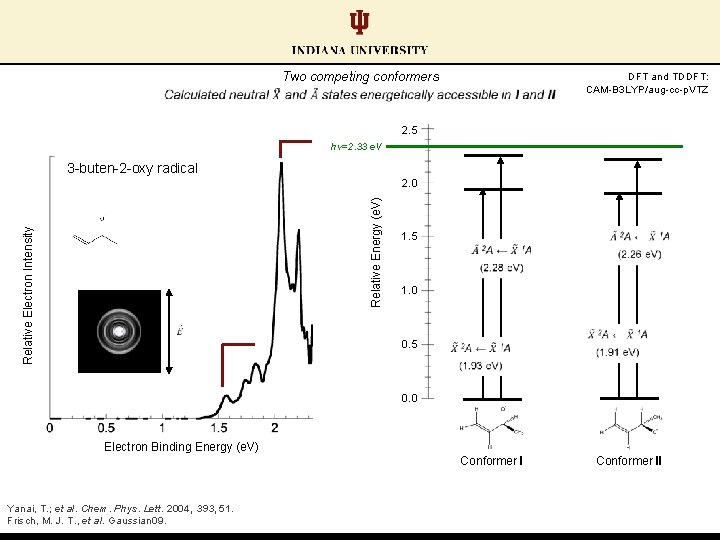
Two competing conformers DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II

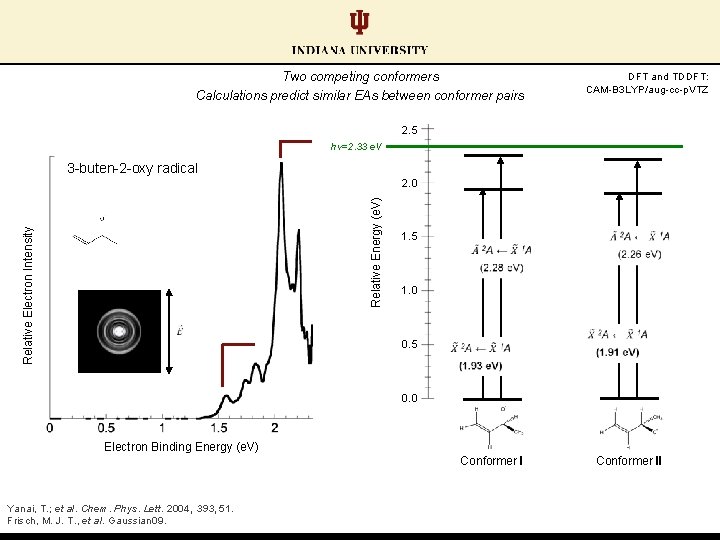
Two competing conformers Calculations predict similar EAs between conformer pairs DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II

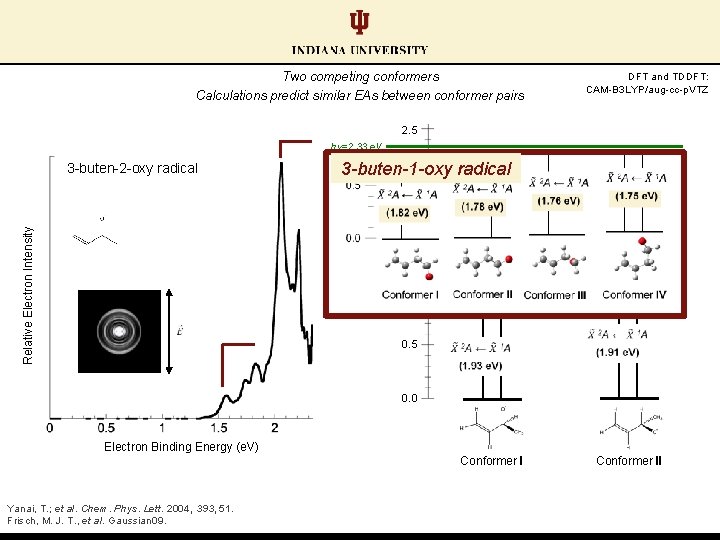
Two competing conformers Calculations predict similar EAs between conformer pairs DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical 3 -buten-1 -oxy radical Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. Conformer II

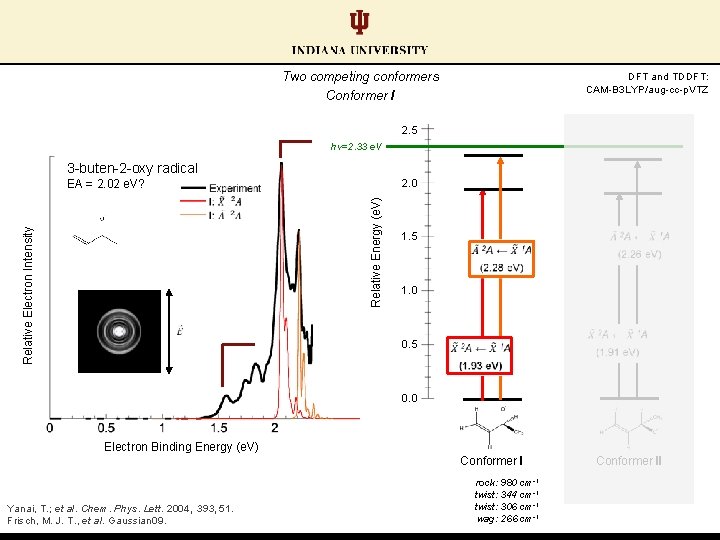
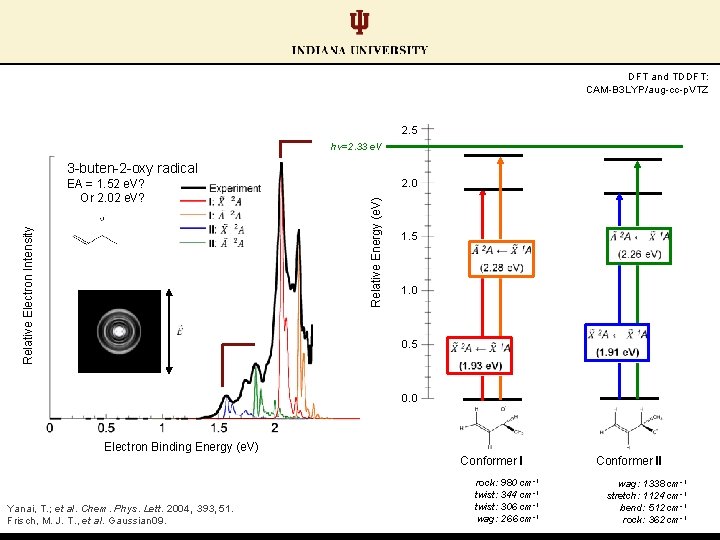
Two competing conformers Conformer I DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical EA = 2. 02 e. V? Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. rock: 980 cm-1 twist: 344 cm-1 twist: 306 cm-1 wag: 266 cm-1 Conformer II

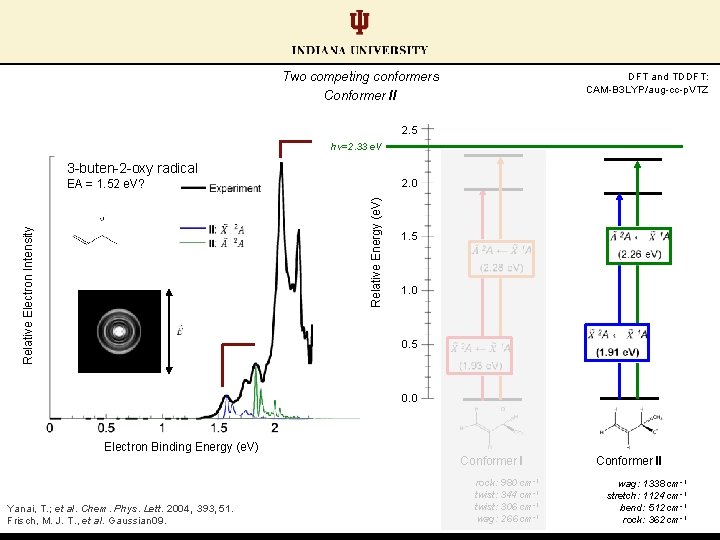
Two competing conformers Conformer II DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical EA = 1. 52 e. V? Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. rock: 980 cm-1 twist: 344 cm-1 twist: 306 cm-1 wag: 266 cm-1 Conformer II wag: 1338 cm-1 stretch: 1124 cm-1 bend: 512 cm-1 rock: 362 cm-1

DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical 2. 0 Relative Energy (e. V) Relative Electron Intensity EA = 1. 52 e. V? Or 2. 02 e. V? 1. 5 1. 0 0. 5 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. rock: 980 cm-1 twist: 344 cm-1 twist: 306 cm-1 wag: 266 cm-1 Conformer II wag: 1338 cm-1 stretch: 1124 cm-1 bend: 512 cm-1 rock: 362 cm-1

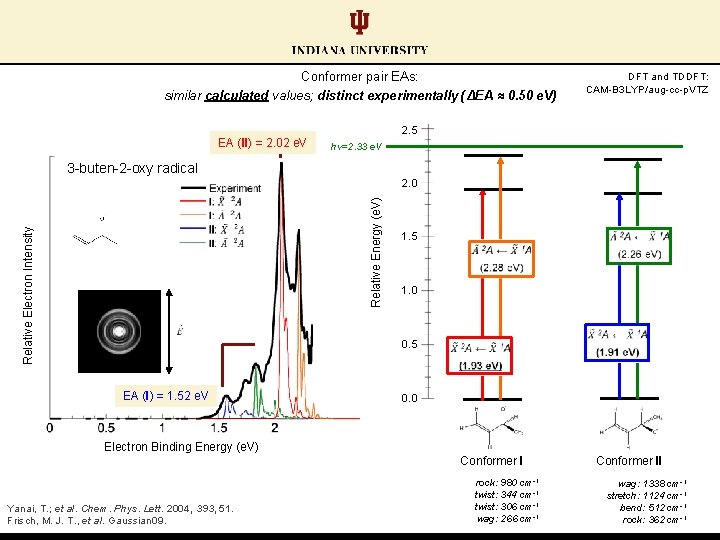
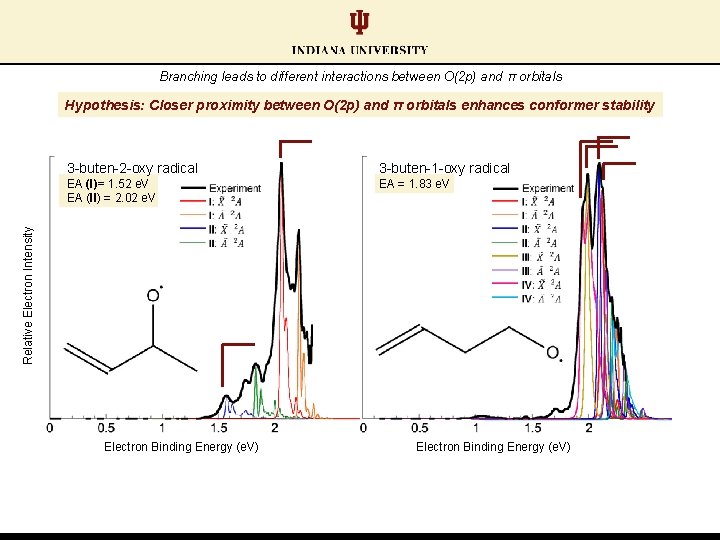
Conformer pair EAs: similar calculated values; distinct experimentally (ΔEA ≈ 0. 50 e. V) EA (II) = 2. 02 e. V DFT and TDDFT: CAM-B 3 LYP/aug-cc-p. VTZ 2. 5 hν=2. 33 e. V 3 -buten-2 -oxy radical Relative Electron Intensity Relative Energy (e. V) 2. 0 1. 5 1. 0 0. 5 EA (I) = 1. 52 e. V 0. 0 Electron Binding Energy (e. V) Conformer I Yanai, T. ; et al. Chem. Phys. Lett. 2004, 393, 51. Frisch, M. J. T. , et al. Gaussian 09. rock: 980 cm-1 twist: 344 cm-1 twist: 306 cm-1 wag: 266 cm-1 Conformer II wag: 1338 cm-1 stretch: 1124 cm-1 bend: 512 cm-1 rock: 362 cm-1

3 -buten-1 -oxy radical EA (I)= 1. 52 e. V EA (II) = 2. 02 e. V EA = 1. 83 e. V Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

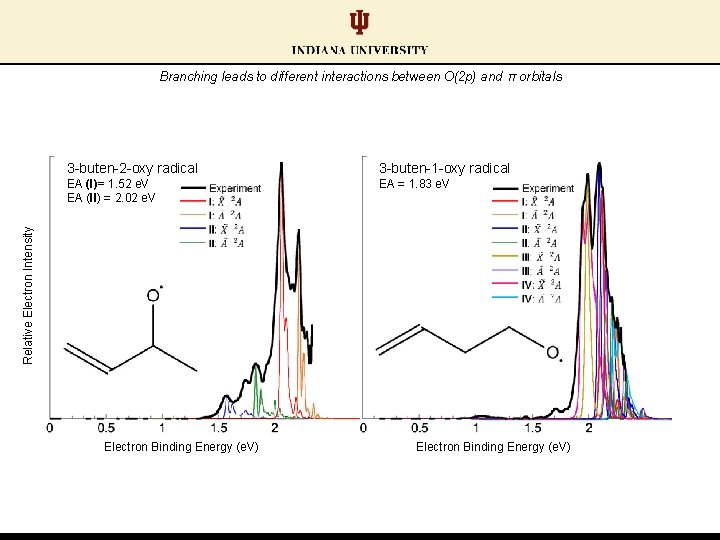
Branching leads to different interactions between O(2 p) and π orbitals 3 -buten-1 -oxy radical EA (I)= 1. 52 e. V EA (II) = 2. 02 e. V EA = 1. 83 e. V Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

Branching leads to different interactions between O(2 p) and π orbitals Hypothesis: Closer proximity between O(2 p) and π orbitals enhances conformer stability 3 -buten-1 -oxy radical EA (I)= 1. 52 e. V EA (II) = 2. 02 e. V EA = 1. 83 e. V Relative Electron Intensity 3 -buten-2 -oxy radical Electron Binding Energy (e. V)

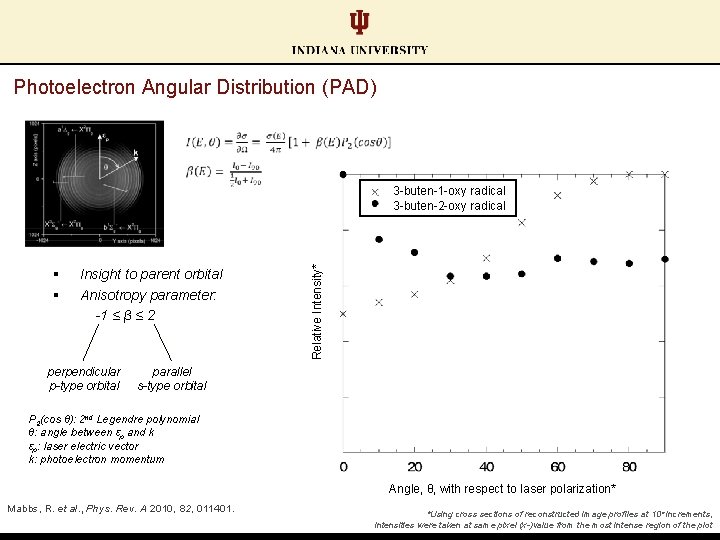
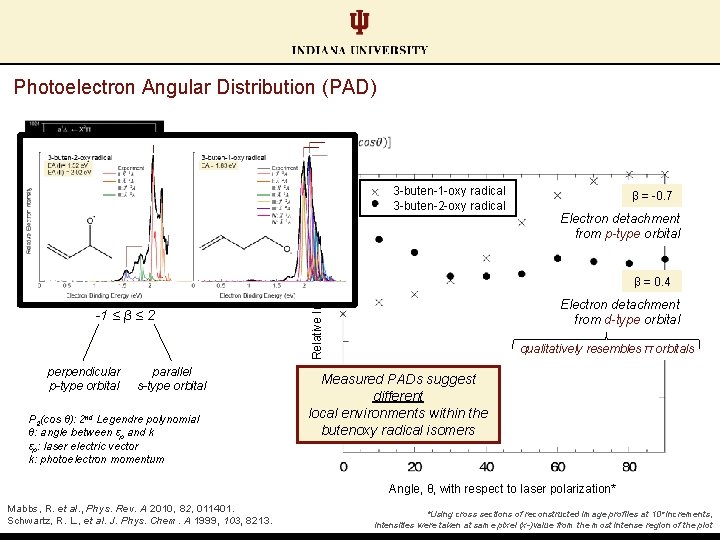
Photoelectron Angular Distribution (PAD) § § Insight to parent orbital Anisotropy parameter: -1 ≤ β ≤ 2 perpendicular p-type orbital parallel s-type orbital P 2(cos θ): 2 nd Legendre polynomial θ: angle between εp and k εp: laser electric vector k: photoelectron momentum Mabbs, R. et al. , Phys. Rev. A 2010, 82, 011401. Schwartz, R. L. , et al. J. Phys. Chem. A 1999, 103, 8213.

Photoelectron Angular Distribution (PAD) § § Insight to parent orbital Anisotropy parameter: -1 ≤ β ≤ 2 perpendicular p-type orbital Relative Intensity* 3 -buten-1 -oxy radical 3 -buten-2 -oxy radical parallel s-type orbital P 2(cos θ): 2 nd Legendre polynomial θ: angle between εp and k εp: laser electric vector k: photoelectron momentum Angle, θ, with respect to laser polarization* Mabbs, R. et al. , Phys. Rev. A 2010, 82, 011401. Schwartz, R. L. , et al. J. Phys. Chem. A 1999, 103, 8213. *Using cross sections of reconstructed image profiles at 10° increments, intensities were taken at same pixel (x-)value from the most intense region of the plot

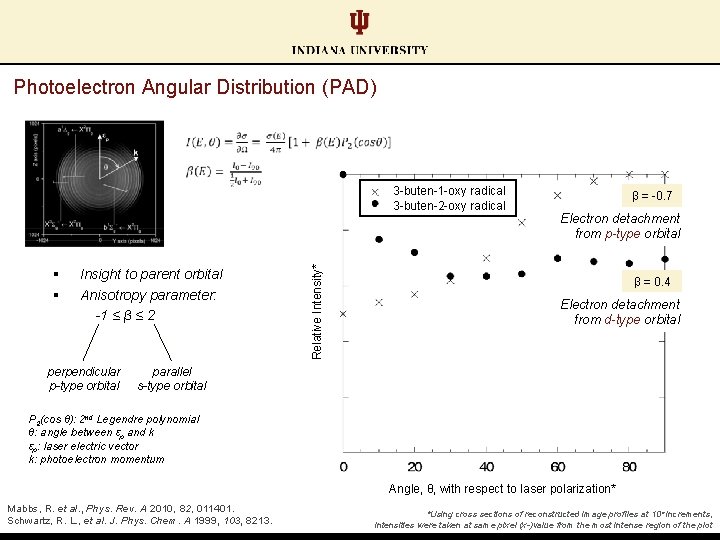
Photoelectron Angular Distribution (PAD) § § Insight to parent orbital Anisotropy parameter: -1 ≤ β ≤ 2 perpendicular p-type orbital Relative Intensity* 3 -buten-1 -oxy radical 3 -buten-2 -oxy radical β = -0. 7 Electron detachment from p-type orbital β = 0. 4 Electron detachment from d-type orbital parallel s-type orbital P 2(cos θ): 2 nd Legendre polynomial θ: angle between εp and k εp: laser electric vector k: photoelectron momentum Angle, θ, with respect to laser polarization* Mabbs, R. et al. , Phys. Rev. A 2010, 82, 011401. Schwartz, R. L. , et al. J. Phys. Chem. A 1999, 103, 8213. *Using cross sections of reconstructed image profiles at 10° increments, intensities were taken at same pixel (x-)value from the most intense region of the plot

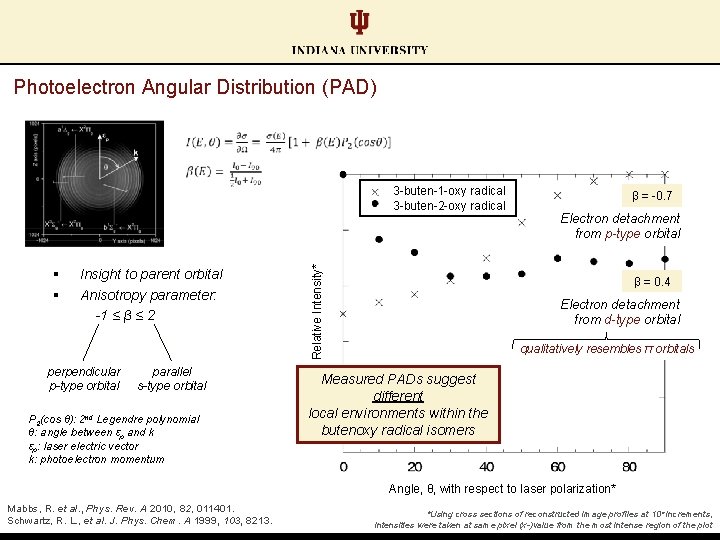
Photoelectron Angular Distribution (PAD) § § Insight to parent orbital Anisotropy parameter: -1 ≤ β ≤ 2 perpendicular p-type orbital parallel s-type orbital P 2(cos θ): 2 nd Legendre polynomial θ: angle between εp and k εp: laser electric vector k: photoelectron momentum Relative Intensity* 3 -buten-1 -oxy radical 3 -buten-2 -oxy radical β = -0. 7 Electron detachment from p-type orbital β = 0. 4 Electron detachment from d-type orbital qualitatively resembles π orbitals Measured PADs suggest different local environments within the butenoxy radical isomers Angle, θ, with respect to laser polarization* Mabbs, R. et al. , Phys. Rev. A 2010, 82, 011401. Schwartz, R. L. , et al. J. Phys. Chem. A 1999, 103, 8213. *Using cross sections of reconstructed image profiles at 10° increments, intensities were taken at same pixel (x-)value from the most intense region of the plot

Photoelectron Angular Distribution (PAD) § § Insight to parent orbital Anisotropy parameter: -1 ≤ β ≤ 2 perpendicular p-type orbital parallel s-type orbital P 2(cos θ): 2 nd Legendre polynomial θ: angle between εp and k εp: laser electric vector k: photoelectron momentum Relative Intensity* 3 -buten-1 -oxy radical 3 -buten-2 -oxy radical β = -0. 7 Electron detachment from p-type orbital β = 0. 4 Electron detachment from d-type orbital qualitatively resembles π orbitals Measured PADs suggest different local environments within the butenoxy radical isomers Angle, θ, with respect to laser polarization* Mabbs, R. et al. , Phys. Rev. A 2010, 82, 011401. Schwartz, R. L. , et al. J. Phys. Chem. A 1999, 103, 8213. *Using cross sections of reconstructed image profiles at 10° increments, intensities were taken at same pixel (x-)value from the most intense region of the plot

Summary Calculations suggest multiple competing conformers with similar EAs Hypothesis: Closer proximity between O(2 p) and π orbitals enhances stability of the 3 -buten -2 -oxy conformers: distinct bands in experimental spectrum PAD indicative of detachment from d-type local orbital Subtle structural changes of isomers can dramatically alter electronic properties [implications for atmospheric models]

Acknowledgements Prof. Caroline Chick Jarrold Dr. Jennifer Mann Marissa Dobulis Josey Topolski Jarrett Mason Richard Schaugaard Brett Williams Joshua Ewigleben Caleb Huizenga Dr. Jared Kafader Dr. Manisha Ray CHEM-1265991 CHE-1664965

Acknowledgements Prof. Caroline Chick Jarrold Dr. Jennifer Mann Marissa Dobulis Josey Topolski Jarrett Mason Richard Schaugaard Brett Williams Joshua Ewigleben Caleb Huizenga Dr. Jared Kafader Dr. Manisha Ray Thank you! CHEM-1265991 CHE-1664965