Electronic Structure and the Periodic Table Unit 6

Electronic Structure and the Periodic Table Unit 6 Honors Chemistry

Electromagnetic Waves: Electromagnetic waves: progressive, repeating disturbances that come from the movement of electric charges Electromagnetic Waves & Light

Wavelength and Frequency n Wavelength ( , lambda): distance between any two points in a wave n measured in any distance unit (mainly nm or m: 1 nm = 1 x 10 -9 m)

Wavelength Can be Measured in One of Two Ways…

Wavelength and Frequency n. Frequency ( ; pronounced nu): the number of cycles of the wave that pass through a point in a unit of time n. Measured in sec-1 (/sec) n 1 sec-1 = 1 Hertz (Hz)
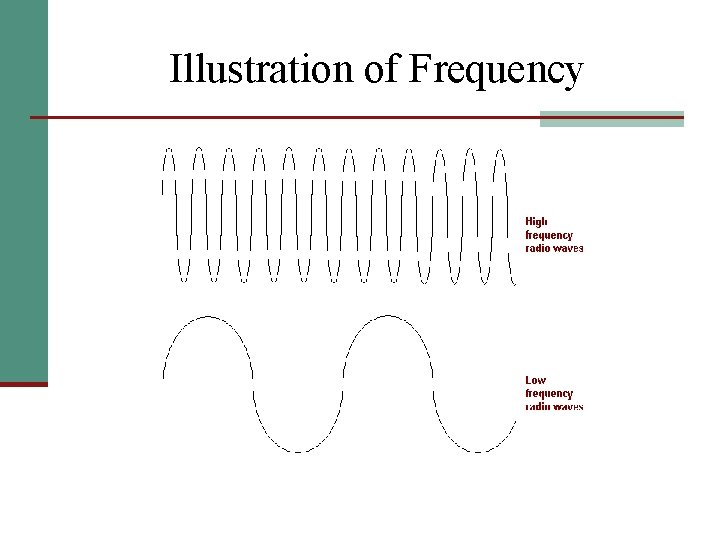
Illustration of Frequency
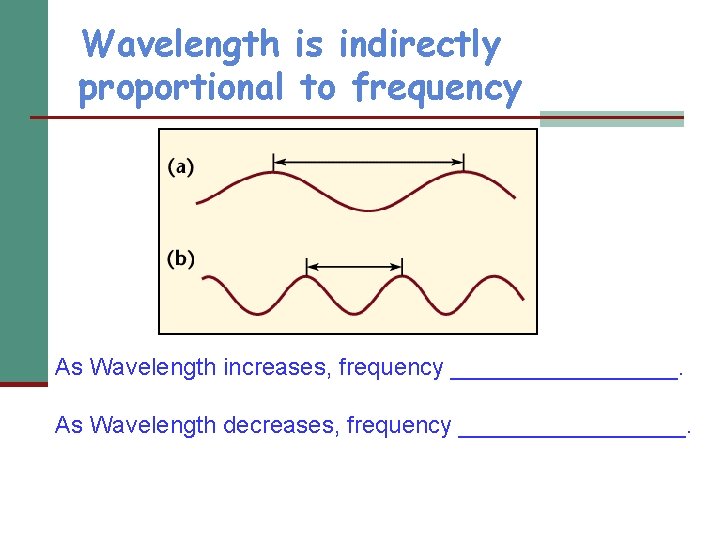
Wavelength is indirectly proportional to frequency As Wavelength increases, frequency _________. As Wavelength decreases, frequency _________.

Amplitude n. Note: height of wave is amplitude (intensity or brightness of wave) n. Amplitude is INDEPENDENT of frequency or wavelength!

Speed n. Speed (c): The speed of light! c = 3. 00 x 108 m/s (rounded to 3 sig figs)

Equation n One equation relates speed, frequency and wavelength: c=

Example n The wavelength of the radiation which produced the yellow color of sodium vapor light is 589. 0 nm. What is the frequency of this radiation?
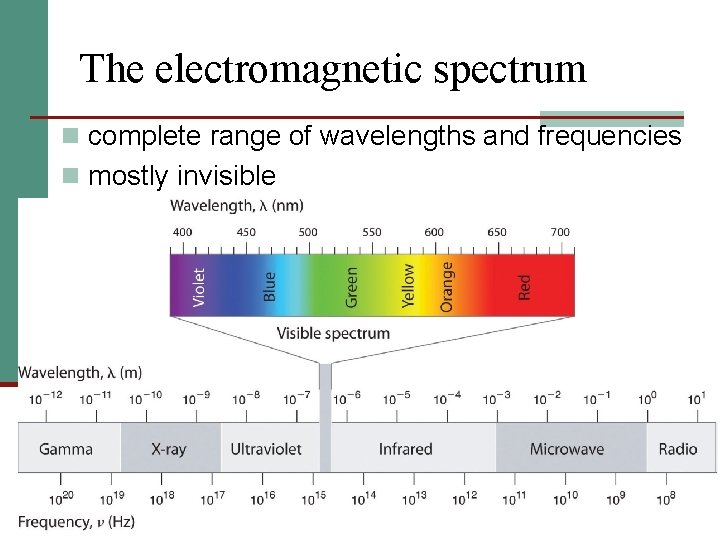
The electromagnetic spectrum n complete range of wavelengths and frequencies n mostly invisible
What is color? TED Talk: What is color?

The visible/continuous spectrum n continuous spectrum: components of white light split into its colors, ROY G BIV n from 390 nm (violet) to 760 nm (red)

Line Spectra n Pattern of lines produced by light emitted by excited atoms of an element n unique for every element n used to identify unknown elements

How do we see color? TED Talk: How we see color

Max Planck n Light is generated as a stream of particles called PHOTONS n Equation: E (Energy of a photon)= h (h =Plank’s constant= 6. 626 x 10 -34 J s)
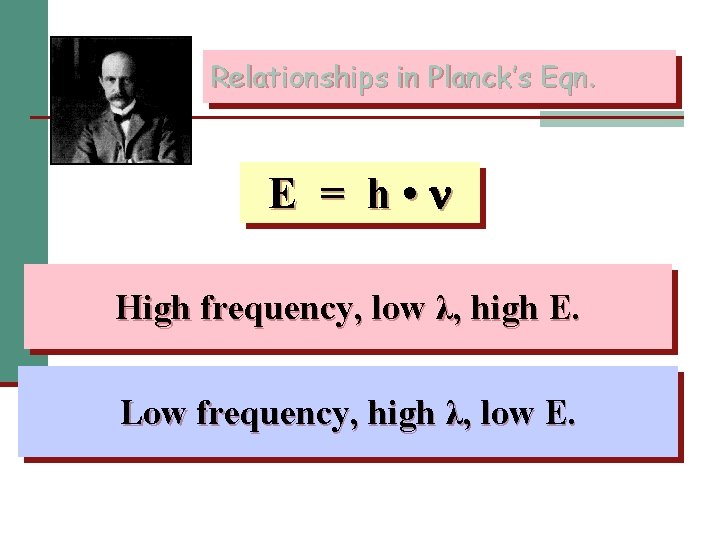
Relationships in Planck’s Eqn. E = h • High frequency, low λ, high E. Low frequency, high λ, low E.

Photoelectric effect – Nobel Prize in Physics 1921 to Einstein Occurs when light strikes the surface of a metal and electrons are ejected. Practical uses: Automatic door openers

Photoelectric Effect: Conclusion Light not only has wave properties but also has particle properties. These massless particles, called photons, are packets of energy.

Example 6. 2 Using the frequency calculated in the previous example, calculate the energy, in joules, of a photon emitted by an excited sodium atom. Calculate the energy, in kilojoules, of a mole of excited sodium atoms.

Bohr’s Hydrogen Atom: A Planetary Model Niels Bohr: Proposed planetary model. Electrons “orbit” the nucleus like planets around the sun. NOT current model of atom but used to explain some features of atom.

Ground State vs. Excited State n ground state: all electrons in lowest possible energy levels n excited state: an electron that has absorbed energy and moved to a higher energy level n This is a temporary state!!

Explanation of Line Spectra & Equation Niels Bohr n Energy of an electron is quantized: can only have specific values. n Energy proportional to energy level.
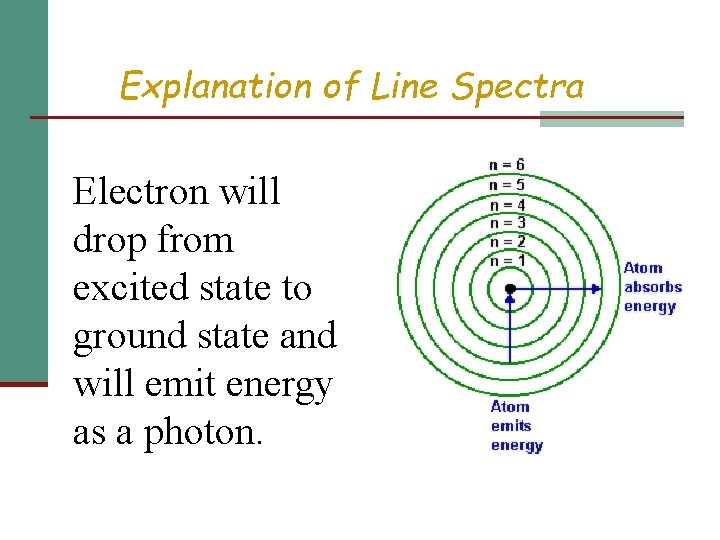
Explanation of Line Spectra Electron will drop from excited state to ground state and will emit energy as a photon.

Explanation of Line Spectra n Type of photon emitted by electron depends on energy difference of energy levels Elevel = -RH 1 – 1 (nhi)2 (nlow)2 AND Elevel = hc/ (h: Planck’s constant, 6. 626 x 10 -34 J sec/photon)

Flaw in Bohr’s Model Only works well for 1 electron species (H atom). Does not explain fine structure of line spectra.

Wave-Particle Duality n. Light has properties of both WAVES and PARTICLES. n most matter has undetectable wavelengths (1000 kg car at 100 km/hr has = 2. 39 x 10 -38 m) n This work led to the development of the electron microscope

Quantum Mechanics n Quantum mechanics: atomic structure based on wavelike properties of the electron n Schrödinger: wave equation that describes hydrogen atom

Heisenberg Uncertainty Principle n The exact location of an electron cannot be determined (if we try to observe it, we interfere with the particle) n You can know either the location or the velocity but not both n Electrons exist in electron clouds and not on specific rings or orbits

Quantum Numbers n Four quantum numbers are a mathematical way to represent the most probable location of an electron in an atom n analogy. . . state = energy level, n city = sublevel, l address = orbital, ml house number = spin, ms

Principal Quantum Number: n n Always a positive integer (1, 2, 3… 7) n Indicates size of orbital, or how far electron is from nucleus n Similar to Bohr’s energy levels or shells n Larger n value = larger orbital or distance from nucleus
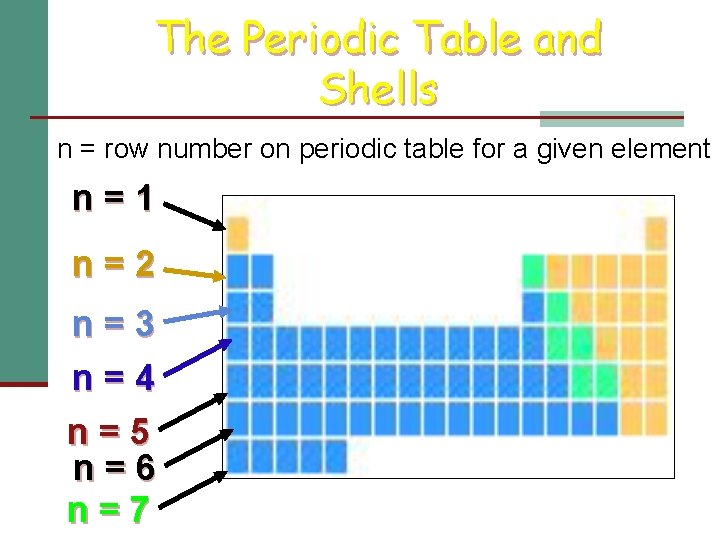
The Periodic Table and Shells n = row number on periodic table for a given element n=1 n=2 n=3 n=4 n=5 n=6 n=7
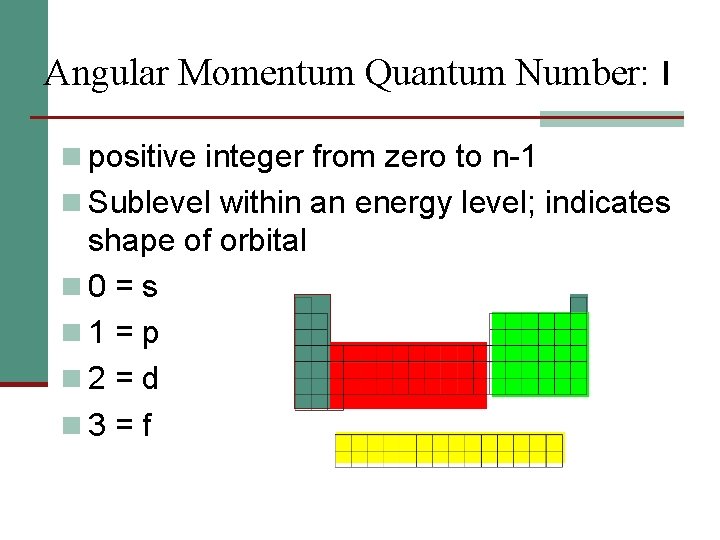
Angular Momentum Quantum Number: l n positive integer from zero to n-1 n Sublevel within an energy level; indicates shape of orbital n 0 = s n 1 = p n 2 = d n 3 = f
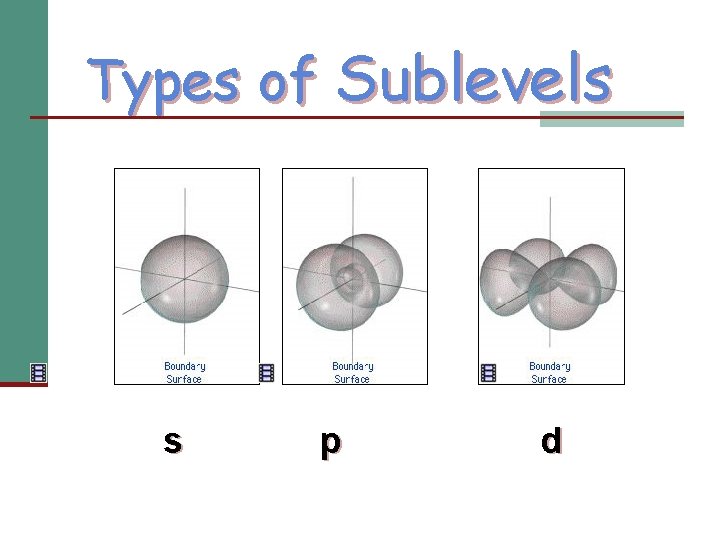
Types of Sublevels s p d

Magnetic Quantum Numbers: ml n integer from -l to +l n Indicates orientation of orbital in space n Orbital = electron containing area

Spin Quantum Number: ms n Two values only: + ½ or -½ n 2 electrons max. allowed in each orbital n (Pauli Exclusion Principle) n Indicates spin of electron; spins of each electron must be opposite

REVIEW: QUANTUM NUMBERS Every Electron has four! n ---> level 1, 2, 3, 4, . . . l ---> sublevel 0, 1, 2, . . . n - 1 ml ---> orbital -l. . . 0. . . +l ms ---> electron spin +½ and -½
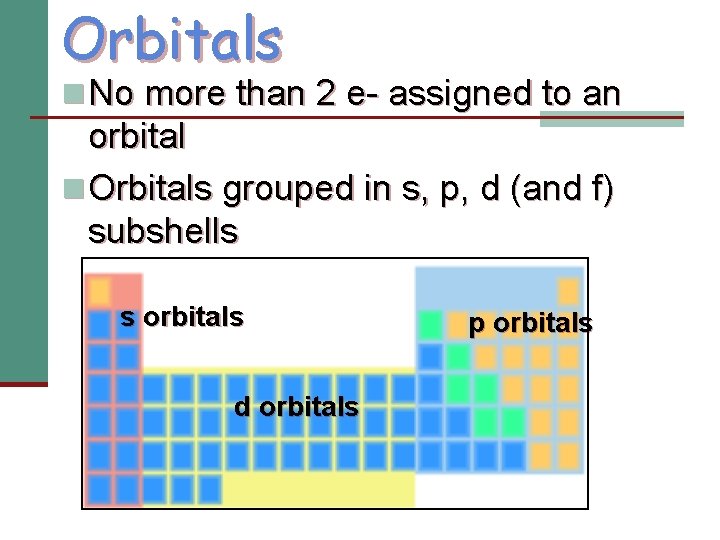
Orbitals n No more than 2 e- assigned to an orbital n Orbitals grouped in s, p, d (and f) subshells s orbitals d orbitals p orbitals
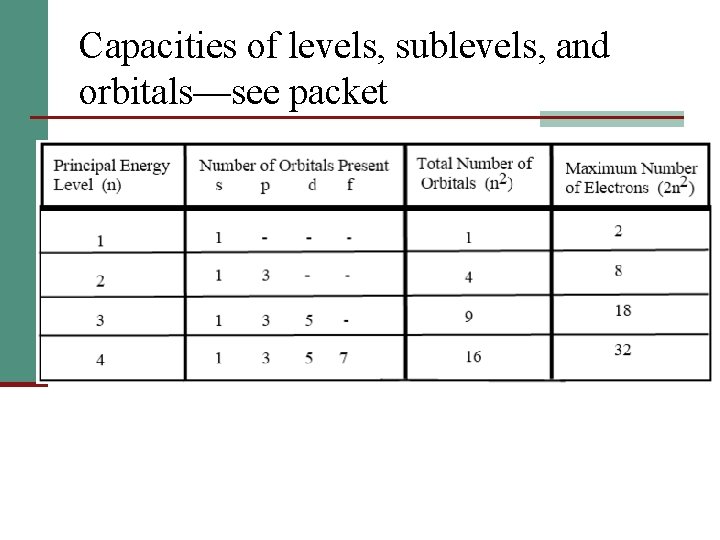
Capacities of levels, sublevels, and orbitals—see packet

Example 6. 6 Give the n and l values for the following orbitals: a. 3 p b. 4 s

Example 6. 8 What are the possible ml values for the following orbitals: a. 3 p b. 4 f
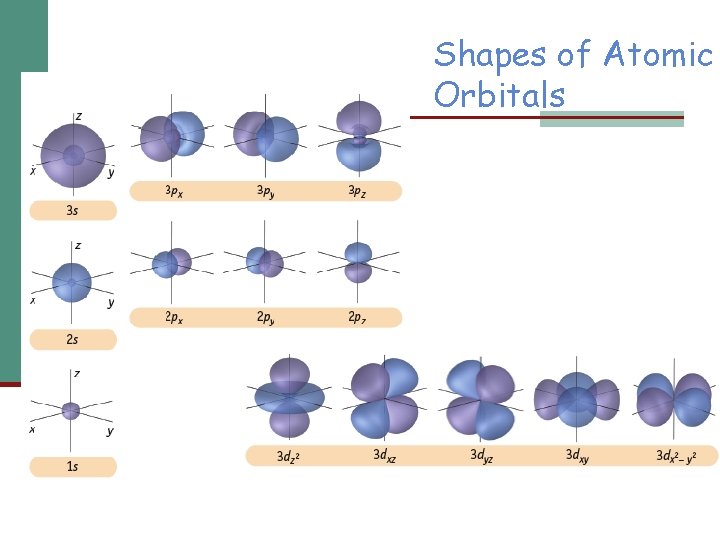
Shapes of Atomic Orbitals

Shapes of Atomic Orbitals s = spherical p = peanut d = dumbbell (clover) f = flower

Multielectron Atoms In the hydrogen atom the subshells (sublevels) of a principal energy level or shell are at the same energy level. Previous Equation: En = –RH /n 2

Multielectron Atoms In a multielectron atom, only the orbitals are at the same energy level: the sublevels are at different energy levels!
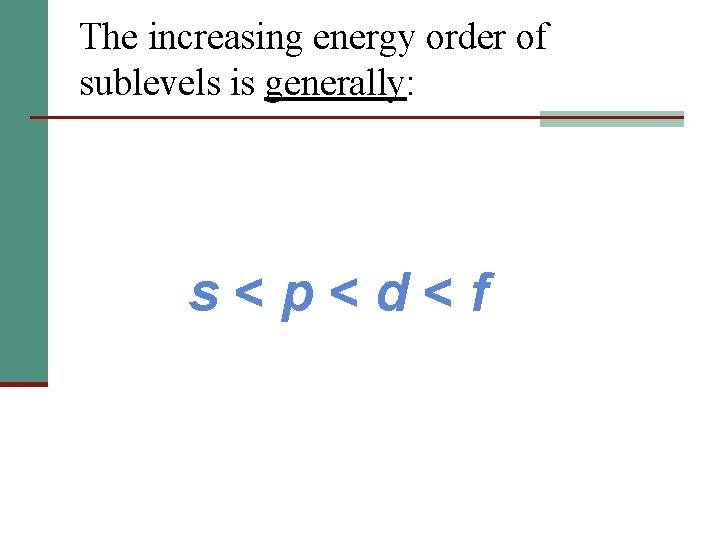
The increasing energy order of sublevels is generally: s<p<d<f
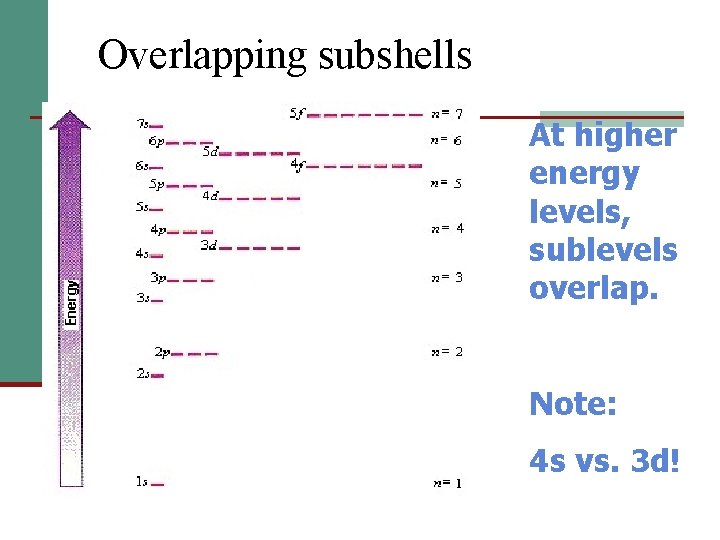
Overlapping subshells At higher energy levels, sublevels overlap. Note: 4 s vs. 3 d!

Introduction to Electron Configuration Definition: describes the distribution of electrons among the various orbitals in the atom Represents the most probable location of the electron! EOS

Electron Configurations n The system of numbers and letters that designates the location of the electrons n 3 major methods: Full electron configurations n Abbreviated/Noble Gas configurations n Orbital diagram configurations n

Full or Complete Electron Configuration (uses spdf) Uses numbers to designate a principal energy level and the letters to identify a sublevel; a superscript number indicates the number of electrons in a designated sublevel. EOS

Rules for Electron Configurations The Aufbau principle: Electrons fill from the lowest energy level to the highest (they don’t skip around) 1 s 22 p 63 s 23 p 64 s 23 d 10 etc.

Pauli Exclusion Principle No two electrons in the same atom can have the same set of 4 quantum numbers. That is, each electron has a unique “address” In other words, the maximum # of electrons an orbital can hold is 2 e- (one with ms = +1/2 and one with ms = -1/2)

HUND’S RULE Orbitals of equal energy in a sublevel must all have 1 electron before the electrons start pairing up a. k. a “creepy person on the bus rule” *** also electrons in half-filled orbitals have same spin
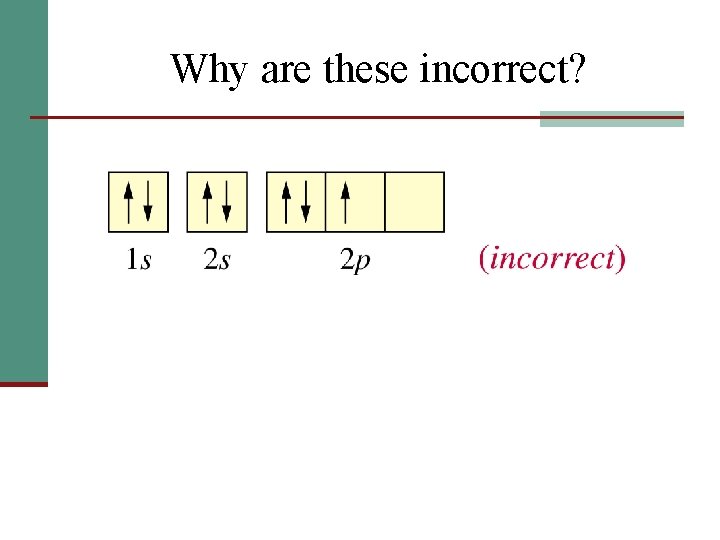
Why are these incorrect?
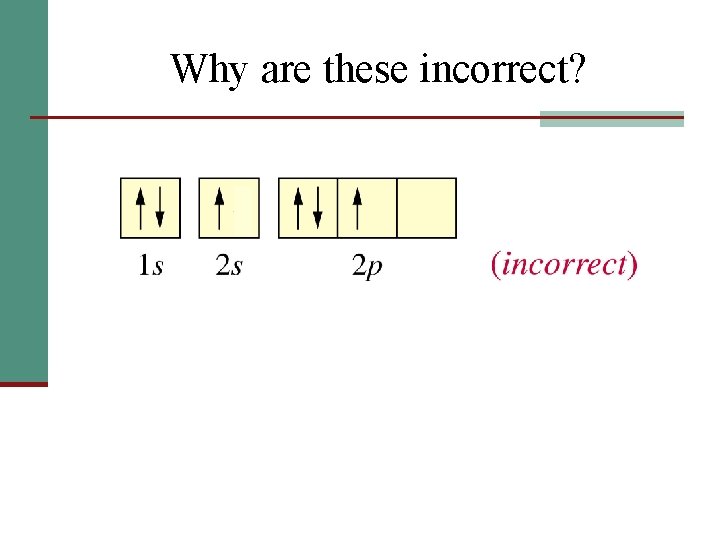
Why are these incorrect?
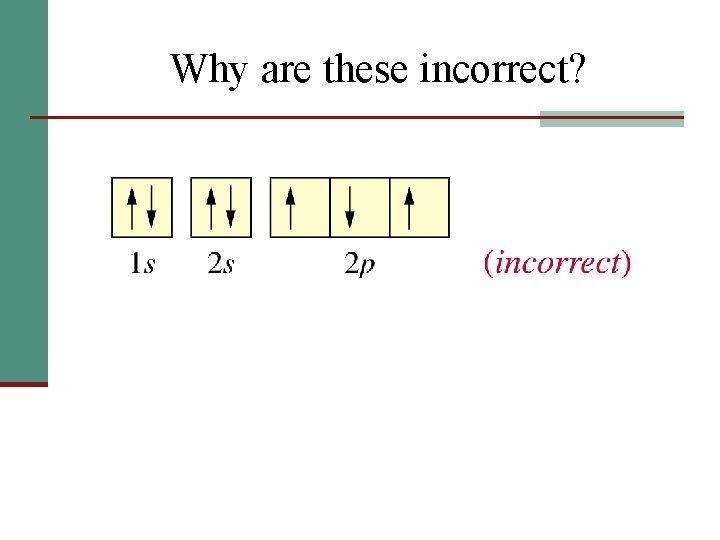
Why are these incorrect?

Full Electron Configuration Example Notation: n 1 s 2 2 s 1 (Pronounced “one-s-two, two-s-one”) A. What does the coefficient mean? Principle energy level B. What does the letter mean? Type of orbital (sublevel) C. What does the exponent mean? # of electrons in that orbital

Steps to Writing Full Electron Configurations 1. Determine the total number of electrons the atom has (for neutral atoms it is equal to the atomic number for the element). Example: F atomic # = # of p+ = # of e- = 2. Fill orbitals in order of increasing energy (see Aufbau Chart). 3. Make sure the total number of electrons in the electron configuration equals the atomic number.

Aufbau Chart (Order of Energy Levels) When writing electron configurations: n d sublevels are n – 1 from the row they appear in n f sublevels are n – 2 from the row they appear in

Writing Electron Configurations Nitrogen: Helium: Phosphorous: Rhodium: Bromine: Cerium:

Abbreviated/Noble Gas Configuration i. Where are the noble gases on the periodic table? ii. Why are the noble gases special? iii. How can we use noble gases to shorten regular electron configurations?

Abbreviated/Noble Gas Configuration Example: Barium 1. Look at the periodic table and find the noble gas in the row above where the element is. 2. Start the configuration with the symbol for that noble gas in brackets, followed by the rest of the electron configuration.

Abbreviated/Noble Gas Configuration Practice! Write Noble Gas Configurations for the following elements: Rubidium: Bismuth: Arsenic: Zirconium:

Writing Electron Configurations Another way of writing configurations is called an orbital diagram. (also called orbital notation or orbital diagrams) One electron has n = 1, l = 0, ms = + ½ Other electron has n = 1, l = 0, ms = - ½
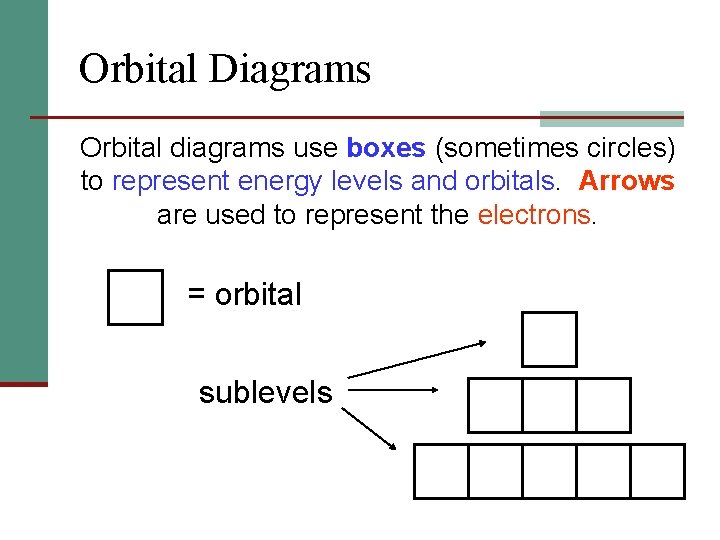
Orbital Diagrams Orbital diagrams use boxes (sometimes circles) to represent energy levels and orbitals. Arrows are used to represent the electrons. = orbital sublevels

Orbital Diagrams Don’t forget - orbitals have a capacity of two electrons!! Two electrons in the same orbital must have opposite spin so draw the arrows pointing in opposite directions. Increasing Energy Example: oxygen 2 p 2 s 1 s 1 s 22 p 4

Drawing Orbital Diagrams 1. First, determine the electron configuration for the element. 2. Next draw boxes for each of the orbitals present in the electron configuration. n Boxes should be drawn in order of increasing energy (see the Aufbau chart). 3. Arrows are drawn in the boxes starting from the lowest energy sublevel and working up. This is known as the Aufbau principle. n Add electrons one at a time to each orbital in a sublevel before pairing them up (Hund’s rule) n The first arrow in an orbital should point up; the second arrow should point down (Pauli exclusion principle) 4. Double check your work to make sure the number of arrows in your diagram is equal to the total number of electrons in the atom. n # of electrons = atomic number for an atom

Electron Configurations for Nitrogen

Electron Configurations for Nickel
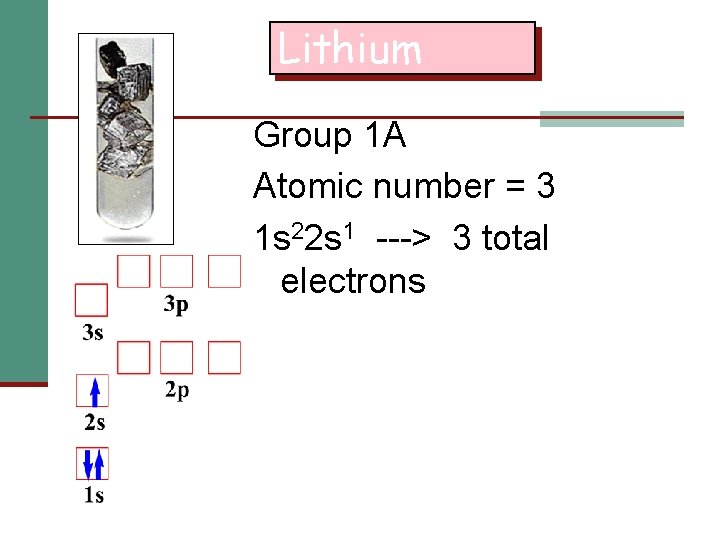
Lithium Group 1 A Atomic number = 3 1 s 22 s 1 ---> 3 total electrons
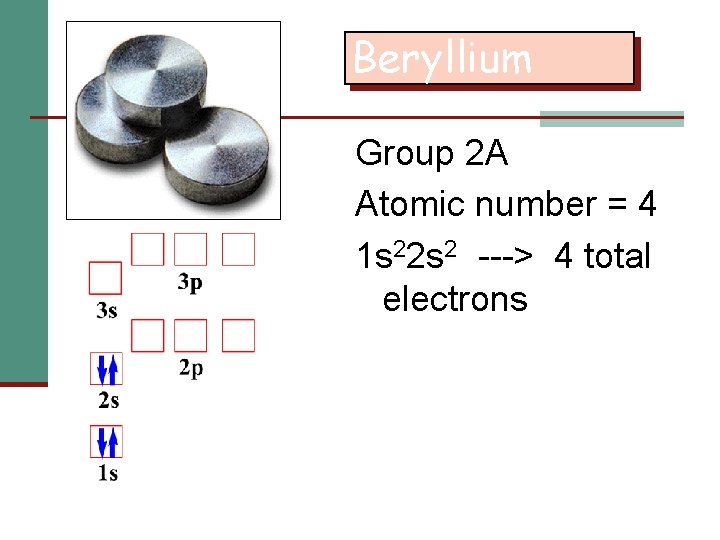
Beryllium Group 2 A Atomic number = 4 1 s 22 s 2 ---> 4 total electrons
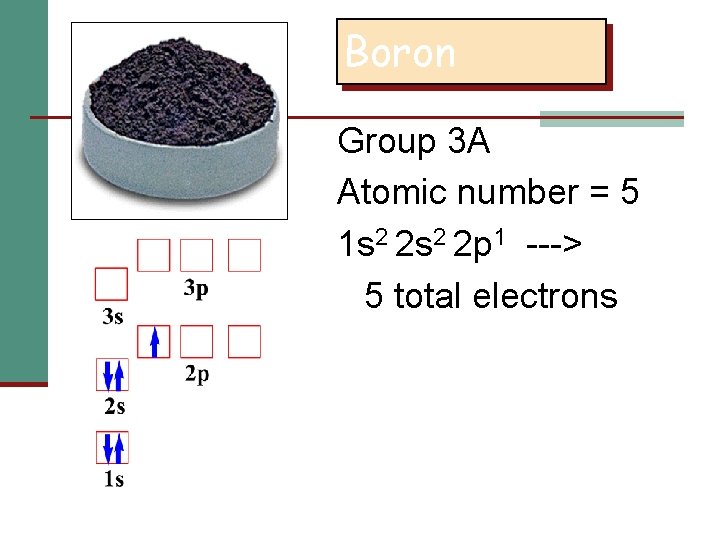
Boron Group 3 A Atomic number = 5 1 s 2 2 p 1 ---> 5 total electrons
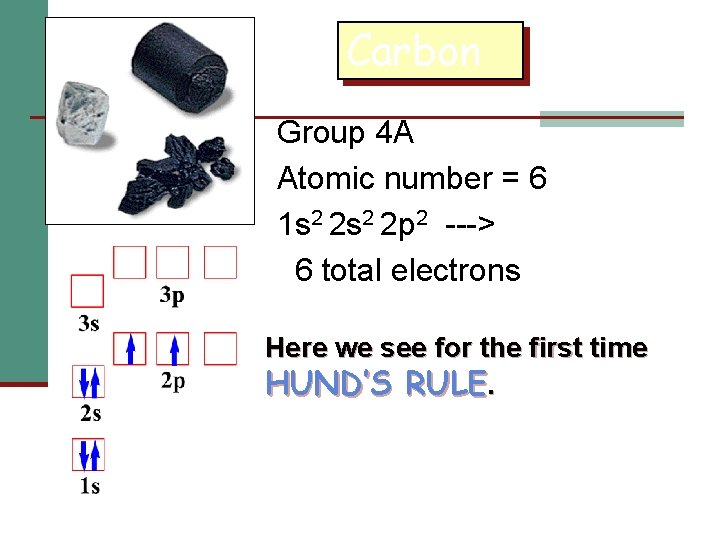
Carbon Group 4 A Atomic number = 6 1 s 2 2 p 2 ---> 6 total electrons Here we see for the first time HUND’S RULE.
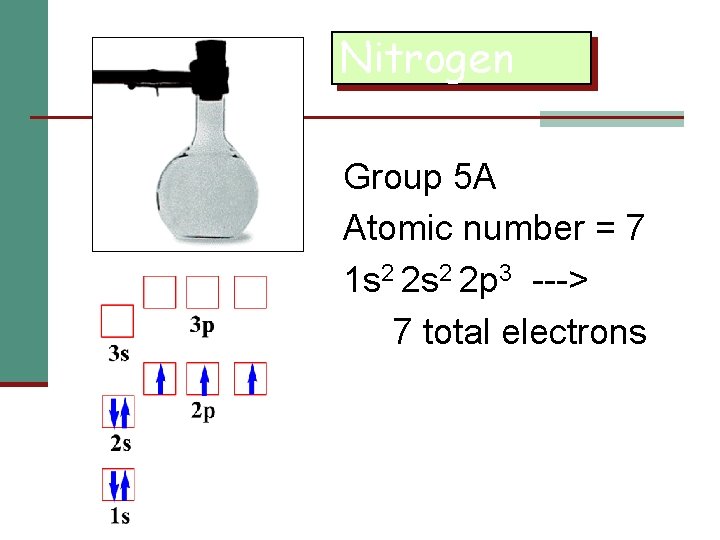
Nitrogen Group 5 A Atomic number = 7 1 s 2 2 p 3 ---> 7 total electrons
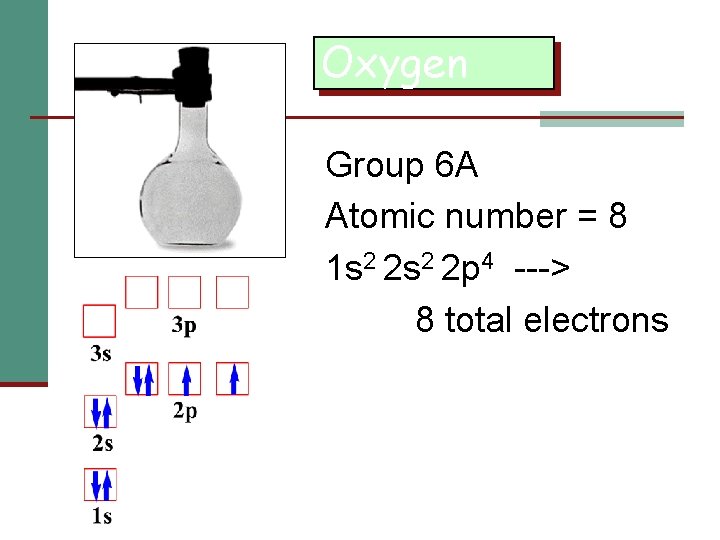
Oxygen Group 6 A Atomic number = 8 1 s 2 2 p 4 ---> 8 total electrons
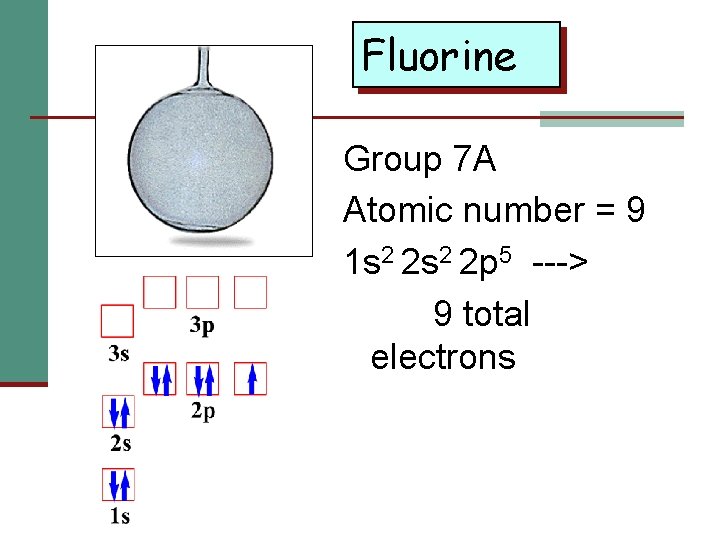
Fluorine Group 7 A Atomic number = 9 1 s 2 2 p 5 ---> 9 total electrons

Neon Group 8 A Atomic number = 10 1 s 2 2 p 6 ---> 10 total electrons Note that we have reached the end of the 2 nd period, and the 2 nd shell is full!
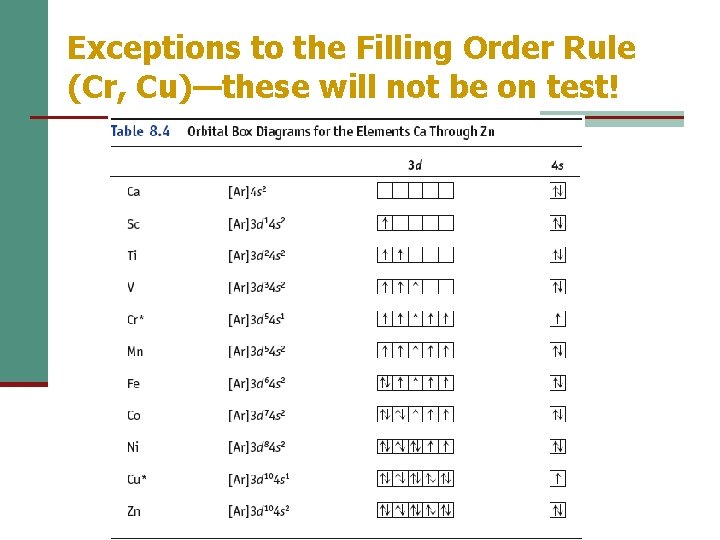
Exceptions to the Filling Order Rule (Cr, Cu)—these will not be on test!

Valence electrons Importance and definition: Definition: Electrons in the outermost energy levels; they determine the chemical properties of an element. Write the noble gas configuration. . . the valence electrons are the ones beyond the core. Example: Sulfur

Valence Electrons and Core Configuration (Shorthand) What is the shorthand notation for S? Sulfur has six valence electrons EOS
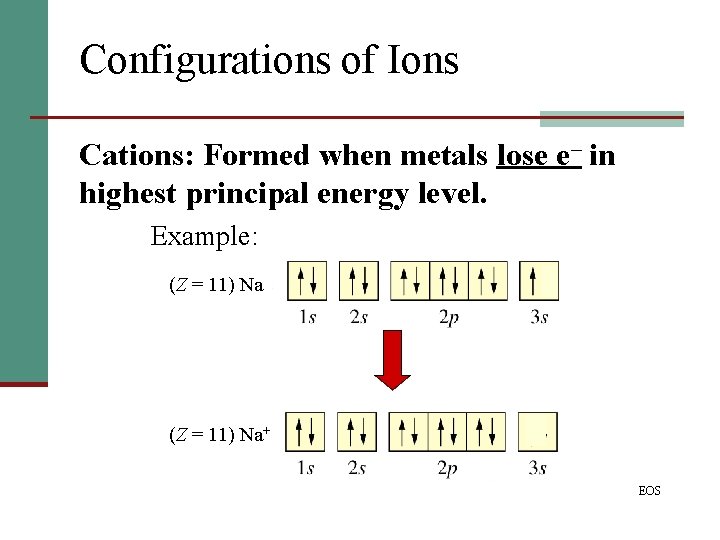
Configurations of Ions Cations: Formed when metals lose e– in highest principal energy level. Example: (Z = 11) Na+ EOS

Configurations of Ions Anions: Formed when non-metals gain e– to complete the p sublevel. Example: Z= 18 Cl EOS

Transition Metals Transition metals (and p block metals) lose e– from the highest principal energy level (n) FIRST, then lose their d electrons! Zr: [Kr] 5 s 24 d 2 Zr+2 : [Kr] 4 d 2 EOS

Isoelectronic Species Definition: Ions or atoms that have the same number of electrons Example: Neon, O 2 -, F-, Na+, Mg 2+, Al 3+ all have the same configuration (1 s 22 p 6) and are isoelectronic

Electron Spin and Magnetism • Diamagnetic: NOT attracted to a magnetic field • Paramagnetic: substance is attracted to a magnetic field. • Substances with unpaired electrons are paramagnetic.

Examples n Mg n Cl Write orbital notation: if it has an unpaired e- it is paramagnetic.

Periodic Properties & Trends n Electronegativity n Ability of an atom to pull e- towards itself n Increases going up and to the right n n Across a period more protons in nucleus = more positive charge to pull electrons closer Down a group more electrons to hold onto = element can’t pull e- as closely

Periodic Properties & Trends Electronegativity n n n Ability of an atom to pull e- towards itself Across a period more protons in nucleus = more positive charge to pull electrons closer Down a group more electrons to hold onto = protons in nucleus can’t pull e- as closely
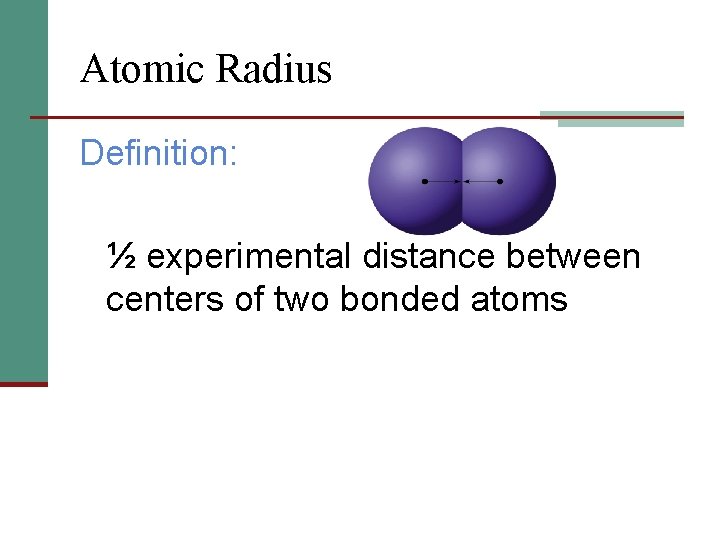
Atomic Radius Definition: ½ experimental distance between centers of two bonded atoms
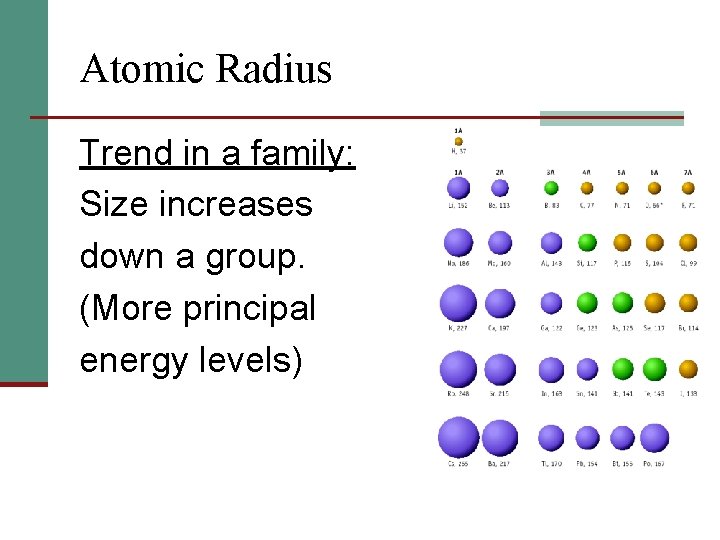
Atomic Radius Trend in a family: Size increases down a group. (More principal energy levels)

Atomic Radius Trend in a period: Size decreases across a period, e- more strongly attracted to nucleus.
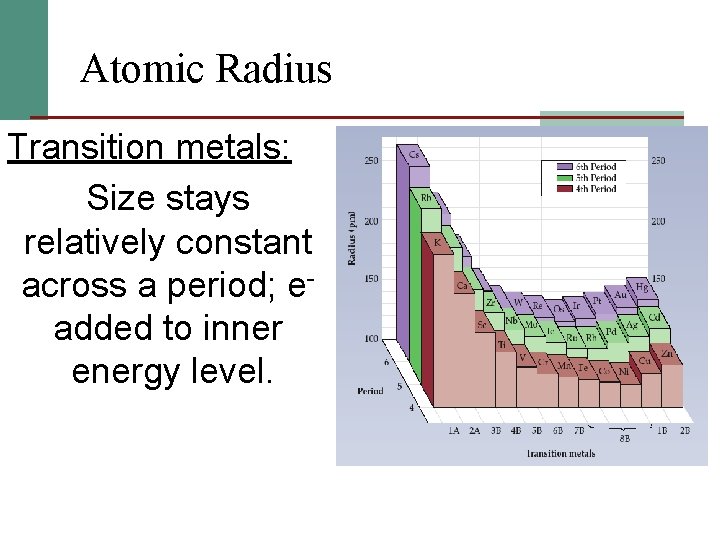
Atomic Radius Transition metals: Size stays relatively constant across a period; eadded to inner energy level.
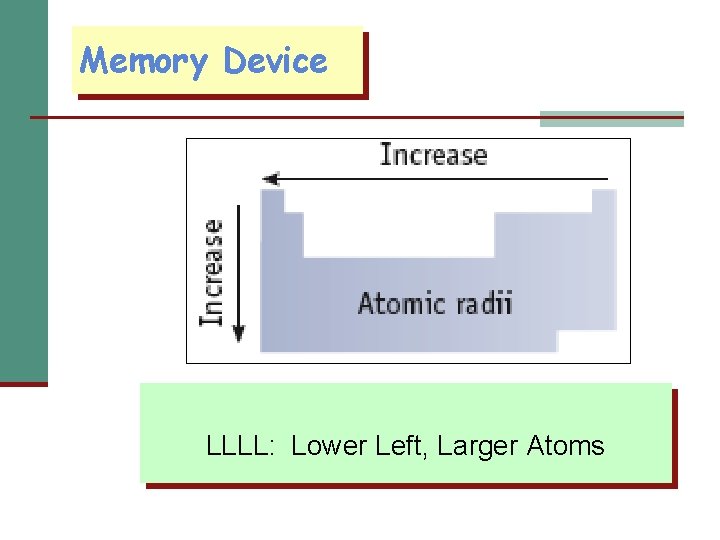
Memory Device LLLL: Lower Left, Larger Atoms

Sizes of Ions + Li, 152 pm 3 e and 3 p Li + , 78 pm 2 e and 3 p n CATIONS are SMALLER than the atoms from which they are formed. n Size decreases due to increasing he electron/proton attraction.

Sizes of Ions F, 71 pm 9 e and 9 p F- , 133 pm 10 e and 9 p n ANIONS are LARGER than the atoms from which they are formed. n Size increases due to more electrons in shell.

Trends in Ion Sizes Trends in ion sizes are the same as atom sizes. Active Figure 8. 15
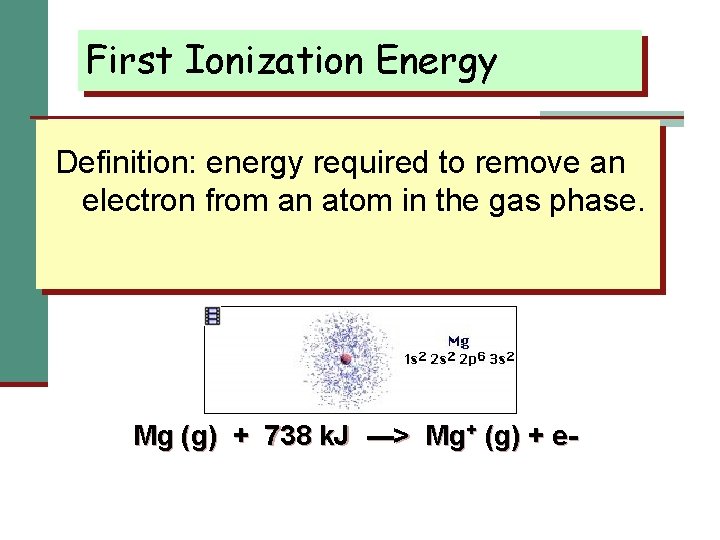
First Ionization Energy Definition: energy required to remove an electron from an atom in the gas phase. Mg (g) + 738 k. J ---> Mg+ (g) + e-
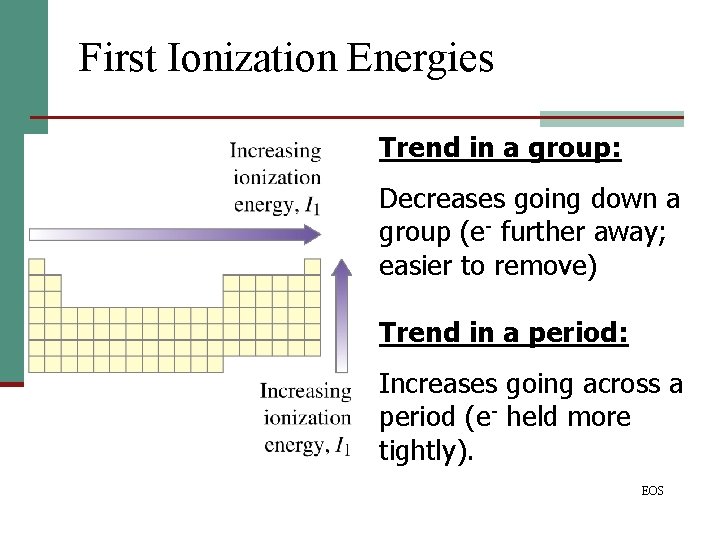
First Ionization Energies Trend in a group: Decreases going down a group (e- further away; easier to remove) Trend in a period: Increases going across a period (e- held more tightly). EOS
Memory Device LLLL: Lower Left, Larger Atoms; Looser electrons

Second Ionization Energy Definition: energy required to remove 2 nd electron from an atom in the gas phase. Takes because e- +ise. Mgmore (g) + energy 738 k. J ---> Mg+ (g) removed from increasingly positive ion. Mg+ (g) + 1451 k. J ---> Mg 2+ (g) + e-

Electron Affinity Some elements GAIN electrons to form anions. Electron affinity is the energy involved when an atom gains an electron to form an anion. A(g) + e- ---> A-(g) E. A. = ∆E
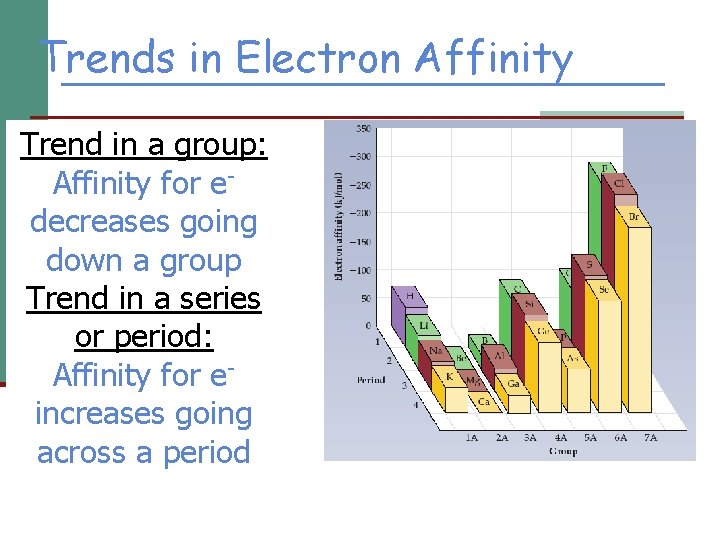
Trends in Electron Affinity Trend in a group: Affinity for edecreases going down a group Trend in a series or period: Affinity for eincreases going across a period

Electron Affinity Note that the trend for E. A. is the SAME as for I. E. !

Trends in Metallic Properties Most metallic means easiest loss of electrons! Metals are on left, nonmetals on right of p. t.
A Summary of Periodic Trends Remember LLLL!!
- Slides: 106